Review Article
Protocol and Guidelines for Ketamine Use in Psychiatric Disorders
- Hesham Maged Mohamed Abdelfatah *
International Scholars, Neuropsychiatry Doctor, Cairo, Egypt.
*Corresponding Author: Hesham Maged Mohamed Abdelfatah, International Scholars, Neuropsychiatry Doctor, Cairo, Egypt.
Citation: Abdelfatah HMM. (2025). Protocol and Guidelines for Ketamine Use in Psychiatric Disorders, Clinical Case Reports and Studies, BioRes Scientia Publishers. 10(5):1-5. DOI: 10.59657/2837-2565.brs.25.273
Copyright: © 2025 Hesham Maged Mohamed Abdelfatah, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: August 18, 2025 | Accepted: September 01, 2025 | Published: September 08, 2025
Abstract
Ketamine has anti depressive characters and approved by (FDA) for the induction and maintenance of anesthesia, Ketamine has been incorporated into the management of psychiatric disorders, such as major depressive disorder (MDD) especially with suicidal ideation or attempting, and of label in management of Bipolar Disorder, Post-Traumatic Stress Disorder (PTSD), obsessive-compulsive disorder (OCD), substance use disorders (SUD). Intravenous ketamine regimen is not a first-line management for psychiatric disorders and considered by the patient’s interdisciplinary team work after failure of standard treatment. Ketamine mechanism of action is different from existing treatments and has demonstrated efficacy in improving people with Major Depression Disorder where conventional first line management as antidepressants, psychotherapy, have not be fully effective.in these papers we discussed all aspects of ketamine Protocol and guidelines usages in psychiatric field.
Keywords: ketamine infusion; suicide ideation; depression; PTSD; OCD; substance abuse; bipolar disorder; alcohol abused
Introduction: What is Ketamine?
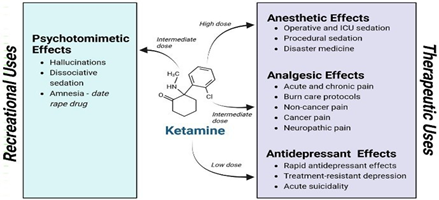
Ketamine is a dissociative anesthetic agent, a compound that induces a state of dissociation between the mind and body of patient. It provides multiple levels of anesthesia, from decreased consciousness but awake to fully unconsciousness, hence why ketamine is used as anesthetic agent. The stage between awake and deeply anesthetic, known as dissociation results in a level of awareness like when we are dreaming.
Figure 1: Ketamine Structure and its Effects.
Background
Ketamine is an NMDA receptor antagonist with a power anesthetic effect. It was developed in 1963 as a replacement for phencyclidine (PCP) by Calvin Stevens at Parke Davis Laboratories. Ketamine started being used for veterinary aspects in Belgium and in 1964 was founded that compared to PCP, it produced minor hallucinogenic characters and shorter psychotomimetic actions. It was approved by FDA in 1970, and from there, it has been used as an anesthetic for children or patients undergoing minor operation.
Ketamine Mechanism of Action
In depression and at low doses ketamine is preferentially bind to and inhibit N-methyl-d-aspartate receptors (NMDARs) on γ- aminobutyric acid (GABA) ergic interneurons. This leading to reduced excitability of these inhibitory interneurons, which causes disinhibition of glutamatergic neurons. With increased depolarization of the presynaptic neuron leads to a surge of glutamate release.
Figure 2: Ketamine Mechanism of Action.
Indications of Treatment with Ketamine
- Patients must be suffering from Resistant depression disorder not response to antidepressants drugs and psychotherapy.
- Patients Have given at least 3 different types of antidepressants drugs for at least 6 weeks with maximum doses.
- Patients Have given at least one type of psychological treatment, e.g., CBT, CAT, Mindfulness. In addition, have either received been considered for ECT or augmentation of antidepressant medications.
- Patients be referred to the regimen by their treating psychiatrist.
- Patients be able to understand the aim of the that treatment regimen, its benefits and possible side effects and can give informed consent for this treatment.
Contraindications of Ketamine Therapy
- Patients not given informed consent.
- Patients lacks of mental capacity.
- Current or recent symptoms of psychosis.
- Significant cardiovascular disease, thyroid disease, eye glaucoma, increased intracranial pressure, epilepsy.
- Pregnant or breast-feeding women or undergoing IVF therapy.
- History of drug induced manic attack.
- Patients can't stop alcohol use for at least 3 days.
Frequency of Treatment of Ketamine Therapy
weekly regimen within the Ketamine with a maximum of 6 sessions for the acute phase of treatment. Then this will continue at an extended time for maintenance, weekly, fortnightly etc. The frequency may be Adjustment with patient as needs, Response rates in controlled trials have ranged from (25% to 85% at 24 hours post infusion) and from (14% to 70%) at 72 hours post infusion).
A weekly assessment and evaluation are done with a planned further infusion fortnightly extended to 3 weekly that depending upon patient responses.
Obtaining Consent
Before starting treatment with Ketamine obtaining informed consent is the responsibility of the Consultant Psychiatrist for Ketamine therapy, and the patients known the all details of guidance of ketamine therapy and he approved and accepted.
Advice to Patients
- For women's that's pains may be felt due to uterine contractions, that depending on the stage of menstrual cycle.
- No drive or operate machines for 24 hours.
- No alcoholism for 24 hours posts treatment.
Prescribing Ketamine
This will be the responsibility of the Anesthetist in consultation with Consultant Psychiatrist.
Ketamine will be prescribed on the inpatient Hospitals not in private clinics.
Administration of Ketamine
Ketamine will be used as a single sub-anaesthetic intravenous dose at (500 micrograms per kilogram taking over 40 minutes) with continuous vital sign monitoring, the dose can be altered according to response and the side effects, tolerability.
Pre-treatment with Ketamine
A full physical examination must be done, and the results made available to the Anesthetist. (CBC, FBC, U&Es, LFTs ECG), are indicated.
Day of Treatment with Ketamine
- Patient must be Fasting from consuming food for at least 4 hours before the setting of ketamine therapy,
- Fluids may be taken up to 2 hours prior to the session.
Administration of Ketamine Infusion
- Cannulation and Ketamine dose calculation. Dose as (500 micrograms per kg).
- Product: Ketamine injection vial 10 mg/ml.
- Diluent: Sodium chloride infusion.
- Infusion Rate: Over 40 minutes.
- The patient will be observed for 2 hours after session and recorder every 30 minutes post infusion until the vital signs are stable and the Patient became full conscious.
- The patient given a light snack and drink.
Figure 3: Ketamine Therapy for Depression.
Side Effects of Ketamine Therapy
The side effects of using ketamine depend on factors such as the amount used, the method of intakes.
- Psychiatric side effects of ketamine include: Anxiety, Agitation or irritation, Frustration or anger, Drowsiness, Confusion, Dizziness, Amnesia, Difficulties concentrating, Disorientation, Delusions, Hallucinations, Dissociation.
- GIT side effects of ketamine include: Stomach Upset, Nausea, Vomiting.
- Cardiovascular side effects of ketamine include: Decreased heart rate, Irregular heartbeat, Increased blood pressure, 4- Respiratory side effects of ketamine include:
- Slowed breathing.
- Ophthalmologic side effects of ketamine include: Involuntary eye movements, Double vision.
- Neurologic side effects of ketamine include: Decreased motor function, Seizures.
Figure 4: Ketamine Side Effects.
Termination of Treatment with Ketamine Therapy
Will be done if there is no response to the number of agreed sessions or if there a consistent nonresponse for 3 consecutive sessions or patient noncompliance by Ketamine treatment protocol.
Off-Label Usages of Ketamine in Psychiatric Field
Mood disorders, post-traumatic stress disorder (PTSD), Bipolar Disorder, Obsessive- Compulsive Disorder (OCD), Substance abuse management.
These psychiatric disorders not approved till now but research papers concluded a positive impact with Ketamine treatment.
Conclusion
Ketamine treatment for Resistant Depression Disorder includes pharmacotherapy, psychotherapy Currently oral antidepressants are the mainstay form of pharmacotherapy there is an overwhelming need to address the obstacles in treating Resistant Depression Disorder due to the delayed treatment response with oral antidepressants and the poor response rate new modalities and drugs can be explored to alleviate the severity of symptoms while ketamine was initially approved as a general anesthetic, it has been repurposed to treat Resistant Depression Disorder, suicidal ideation, and other off-label uses such as substance-use disorders and Mood Disorders, PTSD, Bipolar Disorder, OCD.
Following FDA approval of intranasal (S)-ketamine used in conjunction with an oral antidepressant, other potential routes of administration, including intravenous is currently being researched and explored. An advantage of IV administration is the rapid antidepressant response observed the effects of ketamine in these patients can be observed within 24 hours of the initial dose and last 4-7 days after one intravenous infusion. Ketamine therapy response rates in controlled trials have ranged from 25% to 85% at 24 hours post infusion and from 14% to 70% at 72 hours post infusion.
Disadvantage of IV ketamine administration Was its side effects, the high cost, special precautions and Commitment of the protocol and guidelines of Ketamine therapy. Ketamine been associated with the potential for abuse due to its ability to cause psychotropic side effects. The costs and benefits must be considered before treatment with ketamine begins.
Declarations
Conflict of Interest
The author has declared that no competing interests exist.
Financial support and sponsorship
Not found.
Data Sharing and Data Accessibility Policy
I Agree.
References
- Oxford Health Ketamine Service.
Publisher | Google Scholor - Royal College Statement on ketamine to treat depression.
Publisher | Google Scholor - Ketamine Infusion Therapy Checklist. Ketamine Infusion Therapy, AANA, (2016).
Publisher | Google Scholor - Turner Mason, Loretta Wilson. (2017). Very Special K: A Little Goes A Long Way: Changing the Conversation about Mental Health Treatment. Kaiser Permanente.
Publisher | Google Scholor - Sanacora, G., Frye, M. A., McDonald, W., Mathew, S. J., Turner, M. S., et al. (2017). A Consensus Statement on The Use of Ketamine in The Treatment of Mood Disorders. JAMA Psychiatry, 74(4):399-405.
Publisher | Google Scholor - Parikh, S. V., Lopez, D., Vande Voort, J. L., Rico, J., Achtyes, E., et al. (2021). Developing an IV Ketamine Clinic for Treatment-Resistant Depression: A Primer. Psychopharmacology Bulletin, 51(3):109.
Publisher | Google Scholor