Research Article
Effects of Concurrent Administration of Lead and Dioxin on The Liver of Adult Female Wistar Rat
1Department of Biological Science, School of Science and Technology, The Federal Polytechnic Offa.
2Department of Biochemical Science, School of Science and Technology, The Federal Polytechnic Offa.
3Department of Biological Science, Faculty of Science University of Lagos Goodfella.
*Corresponding Author: Olushola Adefila, Department of Biological Science, School of Science and Technology, The Federal Polytechnic Offa.
Citation: Adefila. O, Ibitomi. O, Dada. O. (2025). Effects Of Concurrent Administration of Lead and Dioxin on The Liver of Adult Female Wistar Rat. Journal of BioMed Research and Reports, BioRes Scientia Publishers. 7(2):1-16. DOI: 10.59657/2837-4681.brs.25.132
Copyright: © 2025 Dr. Olushola Adefila, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: January 21, 2025 | Accepted: February 05, 2025 | Published: February 12, 2025
Abstract
Lead (Pb) and dioxin have the potential to cause damage to the liver by exposure. The current study was therefore designed to investigate the histopathological effects of lead (Pb) and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) (singly and in combination) on the liver of adult female rats. Thirty-two (32) female wistar rats were randomly divided into four groups (A – D) of eight (8) rats each. Group A rats served as control and were given distilled water, group B fed 7.0 mg/kg body weight of lead (Pb) only, group C fed 30 μg/kg body weight of dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) only, group D fed 7.0 mg/kg body weight of lead (Pb) and 30 μg/kg body weight of dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) for twentyeight (28) consecutive days. Experimental Animals were sacrificed on the 29th day by cervical dislocation. The liver was collected after sacrifice and fixed in 10 % formol calcium and subsequently processed for histological observation using Hematoxylin and Eosin staining principle. In co‑exposed rats, hepatic tissue damages were significantly increased when compared to the individual toxic effects of lead (Pb) and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin). These toxicants exhibited synergism which was supported by histopathological studies. Result revealed that when the animals were co-exposed to lead (Pb) and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin), the hepatic tissues became more vulnerable. Thus, the chances of hepatotoxicity increase when the subjects are being exposed to both lead (Pb) and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) than either one of these toxicants alone. The chances of hepatotoxicity increase when the subjects are being exposed to both lead (Pb) and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) than either one of these toxicants alone. The distortions of the liver morphology may adversely affect liver functions and subsequently predispose to liver failure.
Keywords: lead; liver; wistar rat
Introduction
Lead is the most toxic heavy metal in the environment (Henretig, 2016). Lead exposure is widespread and has an impact on public health (Dart et al., 2014). It is used to produce many products, including lead crystal tools, printing pigments, hair coloring pigments, and cosmetics (Patrick, 2016). Exposure to lead occurs through lead-related professions and in industrial processes such as lead smelting, combustion, pottery making, paint, battery recycling dyes, book printing, house dust, and polluted soil (Levin et al., 2018). Occupational exposure to lead occurs in smelters, mines, and battery factories, as well as welding of lead-coated metal. The magnitude of the response to lead toxicity depends on the dose, age of the person exposed, duration of exposure, nutritional status and occupational exposure (Kosnett, 2015). Lead spreads to all organs and systems in the body, but the prime target of lead toxicity is the central nervous system (Ekong et al., 2016). It can affect the neurological and biological functions of the animal body. It causes several problems in various organs, such as mental retardation, behavioral problems, violence, and growth delay, damage to the central nervous system and learning abilities, damage to sensory nerves, and impairment in cognitive function, hearing and vision (Patrick, 2016). Lead can cause adverse health effects in three ways inhalation, ingestion and skin contact (Mycyk et al., 2015). It also causes toxic effects on genes, especially in the brain, bone marrow, lungs, and liver (Mahaffey, 2016). Dioxins are permanent environmental pollutants that pose a potential risk to human health (Guo et al., 2019). Among all dioxin compounds, 2,3,7,8-tetrachlorodibenzo-p-dioxin is a highly toxic environmental contaminant (Knutsen et al., 2018). In the experimental animals exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin, various biochemical changes and diseases like dermal toxicity, immunotoxicity, hepatotoxicity, carcinogenesis, teratogenicity, behavioral and endocrine changes are identified (Li et al., 2021). In addition to such diseases, exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin is determined to cause oxidative stress (Ishiniwa et al., 2013). Oxidative stress is defined as defects in the oxidant-antioxidant balance (Devito 2012). In an early study, oxidative stress is identified in various tissues exposed acutely to high doses of 2,3,7,8-tetrachlorodibenzo-p-dioxin (Mikolajczyk et al., 2021). It is reported that free oxygen species, deoxyribonucleic acid damage and lipid peroxidation increase in experimental animals exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin (Guo et al., 2019). Most of the toxicological studies on water contaminating of permanent environmental pollutants deal with the single metal exposure. Little is known about the interaction among these metals due to their co-exposure through drinking water and their combined toxic effects on animals and humans. Therefore, the present study was to address interaction of metals by assessing the effects of exposure to combination of lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) on liver in rats.
The Liver
The liver is a reddish-brown gland located immediately inferior to the diaphragm, filling most of the right hypochondriac and epigastric regions. It is the body’s largest gland. The liver has numerous functions. From their variety and importance, it is not hard to understand why such liver diseases as cirrhosis, hepatitis, and liver cancer are so serious and often fatal. Only one of these functions contributes to digestion; the secretion of bile (Saladin, 2018). Bile is a yellow-green fluid containing minerals, cholesterol, neutral fats, phospholipids, bile pigments, and bile acids. The principal pigment is bilirubin, derived from the decomposition of hemoglobin. Bacteria of the large intestine metabolize bilirubin to urobilinogen, which is responsible for the brown color of feces. In the absence of bile secretion, the feces are grayish white and marked with streaks of undigested fat (acholic feces). Bile acids (bile salts) are steroids synthesized from cholesterol. Bile acids and lecithin, a phospholipid, emulsify fat; breaking globules of dietary fat into smaller droplets with more surface area exposed to enzyme action. Emulsification greatly enhances the efficiency of fat digestion (Lan and Muralitharan, 2015).
Anatomy Of the Liver
The liver weighs about 1.36 kilograms (kg) and is located in the right upper quadrant of the abdomen, tucked against the inferior surface of the diaphragm (Elaine, 2016). The posterior surface of the liver is in contact with the right ribs 5–12. It is divided into two major lobes, the right lobe and the left lobe, which are separated by a connective tissue septum, the falciform ligament. Two smaller lobes, the caudatelobeand the quadratelobe, can be seen from an inferior view. Also seen from the inferior view is the porta, which is the “gate” through which blood vessels, ducts, and nerves enter or exit the liver (Lan and Muralitharan, 2015).
Functions Of the Liver
The liver performs important digestive and excretory functions, stores and processes nutrients, detoxifies harmful chemicals, and synthesizes new molecules (Saladin, 2018). The liver produces and secretes about 600–1000 mL of bile each day. Bile (bı̄l) contains no digestive enzymes, but it plays an important role in digestion by diluting and neutralizing stomach acid and by dramatically increasing the efficiency of fat digestion and absorption. Digestive enzymes cannot act efficiently on large fat globules. Bile salts emulsify fats, breaking the fat globules into smaller droplets, much like the action of detergents in dishwater. The small droplets are more easily digested by digestive enzymes. Bile also contains excretory products, such as cholesterol, fats, and bile pigments, including bilirubin; a bile pigment that results from the breakdown of hemoglobin. Gallstones may form if the amount of cholesterol secreted by the liver becomes excessive and is not able to be dissolved by the bile salts (Saladin, 2018). Neural and hormonal stimuli regulate the secretion and release of bile. Bile secretion by the liver is stimulated by parasympathetic stimulation through the vagus nerve. Secretin, which is released from the duodenum, also stimulates bile secretion and release. Cholecystokinin stimulates the gallbladder to contract and release bile into the duodenum. In addition, most (90% of) bile salts are reabsorbed in the ileum. The blood carries the bile salts back to the liver, where they stimulate additional bile salt secretion and are once again secreted into the bile. The loss of bile salts in the feces is reduced by this recycling process (Lan and Muralitharan, 2015). The liver can remove sugar from the blood and store it in the form of glycogen. It can also store fat, vitamins, copper, and iron. This storage function is usually short-term. Foods are not always ingested in the proportion needed by the tissues. If this is the case, the liver can convert some nutrients into others. For example, if a person eats a meal that is very high in protein, a large number of amino acids and only a small amount of lipids and carbohydrates are delivered to the liver (Saladin, 2018).
The liver can break down the amino acids and cycle many of them through metabolic pathways to produce adenosine triphosphate and to synthesize lipids and glucose. The liver also transforms some nutrients into more readily usable substances. Ingested fats, for example, can be combined with choline and phosphorus in the liver to produce phospholipids, which are essential components of cell membranes (Saladin, 2018). Many ingested substances are harmful to body cells. In addition, the body itself produces many by-products of metabolism that, if accumulated, are toxic. The liver is an important line of defense against many of those harmful substances. It detoxifies them by altering their structure, which makes their excretion easier. For example, the liver removes ammonia, a toxic by-product of amino acid metabolism, from the circulation and converts it to urea, which is then secreted into the circulation and eliminated by the kidneys in the urine. The liver also removes other substances from the circulation and excretes them into the bile (Lan and Muralitharan, 2015).
Literature Review
Literature ReviewDioxins
Dioxins, polychlorinated dibenzodioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs), are the most toxic group of environmental pollutants that have been described to date (Gotz et al., 2017). Chemically, dioxins consist of two aromatic rings linked via either one or two atoms of oxygen, and give rise, respectively, to polychlorinated dibenzofurans or polychlorinated dibenzodioxins. This extremely stable structure contains one to eight positions that can be chlorinated, which confers both high structural stability and extreme hydrophobicity. Depending on the number and position of chlorination (P = 1–8), the dioxin group includes 75 polychlorinated dibenzodioxins and 135 polychlorinated dibenzofurans congeners that vary significantly in terms of their overall toxicity (Ishiniwa et al., 2013). The number and position of chlorines in dioxin molecules affect their toxicity by modifying their shape, determining thus their binding ability to the AhR receptor (Pajurek et al., 2023). Typically, congeners with chlorine atoms substituted in the lateral 2, 3, 7 and 8 positions of the aromatic rings are considered as the most toxic. Of these, 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), with a toxic equivalency factor (TEF) of 1.0, is the most toxic congener of all the dioxins (Pius et al., 2020). Dioxins are formed as a by-product from incomplete thermic reactions, including industrial processes, combustion processes such as burning of household-, municipal- and medical waste, or burning fuels. Dioxins often occur as a mixture of chemical congeners in the environment and can act both synergistically, inducing a stronger toxic effect, or antagonize the effect of another (Fromme et al., 2016). Dioxins are formed as a by-product from incomplete thermic reactions, including industrial processes, combustion processes such as burning of household-, municipal- and medical waste, or burning fuels (Sindiku et al., 2015). Dioxins released in the environment because of improper disposal are extremely persistent to degradation and can remain in both water and soils for years. Dietary intake of dioxins is the main exposure route for humans and animals, because of their lipophilic properties and the high accumulation potential. Dioxins bioaccumulate in marine foodwebs and seafood is the main source of exposure for humans (Pajurek et al., 2022). Dioxins are emitted into the environment from two main sources. The first one, which causes emission of only a small proportion of these chemical contaminants, is due to natural combustion-related process such as forest fires and volcanoes (Goto et al., 2017). Over the last decade, the incidence of forest fires has increased resulting in higher dioxin releases from this source (Wikoff et al., 2012). Secondly, dioxins are released into the environment from industrial sources, such as processes involving melting of metals, production of polyvinyl chloride plastics, treatment of white paper pulp with chlorine, the combustion of coal, natural gas and petroleum products and wastes, and the manufacture of chlorinated pesticides and herbicides for agriculture. The incineration of municipal and medical wastes as wastes from fossil fuels and fly ash storage are also important sources of dioxins (Zhang et al., 2016). Polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans are also released from plastic bottles and containers exposed to microwave radiation or freezing, and from the vast and ever-increasing amounts of electronic wastes that are estimated to reach 52.2 million tonnes by 2021 (Sindiku et al., 2015). Ironically, recycling methods in processing of so-called e-waste, such as manual disassembly, roasting, acid leaching and open burning also result in the formation of polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans (Sindiku et al., 2015). Finally, road transport emissions due to the combustion of carbon-based fuels are an important source of several halogenated organic pollutant, like polychlorinated dibenzo-p-dioxins/polychlorinated dibenzofurans, polychlorinated biphenyls and polybrominated diphenyl ethers (PBDEs) (Goto et al., 2017).
Health Effects of Dioxins
Chlorinated dioxins are considered as one of the most toxic man-made substances, because of the low dose required to cause lethality (even though not leading to an instant death) (Pajurek et al., 2023). The 2,3,7,8-tetrachlorodibenzo-p-dioxin congener is used as a model compound since it is the most toxic congener (Devito, 2012). Exposure to dioxins can cause several serious health outcomes such as severe wasting, thymic atrophy, teratogenesis, reproductive effects, chloracne, immunotoxicity, enzyme induction, decreases in T4 and vitamin A, and increased hepatic porphyrins (Fromme et al., 2016). One effect of severe dioxin exposure is chloracne, a condition that is extremely persistent and easily identified. There are examples of chloracne which lasted for more than 30 years (Wikoff et al., 2012). According to Mikolajczyk et al. (2021), the biochemical effects of 2,3,7,8-2,3,7,8-tetrachlorodibenzo-p-dioxin can be grouped into three classes, Altered metabolism resulting from changes in enzyme levels. Altered homeostasis resulting from changes in hormones and their receptors. Altered growth and differentiation resulting from changes in growth factors and their receptors (Mikolajczyk et al., 2021).
The various symptoms of the diseases caused by exposure to chlorinated dioxins are mediated through the Aryl hydrocarbon (Ah) receptor pathway (Kannan et al., 2012). The Ah receptor is a ubiquitous, high affinity protein that responds to planar aromatic ligands (e.g. 2,3,7,8-tetrachlorodibenzo-p-dioxin) by forming an active complex. The activated complex binds to a specific position on deoxyribonucleic acid, and probably functions as transcriptional enhancers (Bi et al., 2020). Several parts of the normal growth, development and differentiation, as well as hypoxia, aging and circadian rhythms are regulated by the Ah receptor (Mikolajczyk et al., 2021). Dioxin is absorbed in the gastrointestinal tract and transported into the systematic circulation. It then binds into lipid rich tissue, accumulates and/or is eliminated. The elimination of dioxin is affected by the amount of body fat (the less body fat the faster elimination) and metabolism. Degradation rate varies among species and even within species (Kulkarni, 2019). Chlorinated dioxins and brominated dioxins have many similar characteristics, and this also applies to their affinity to the Ah receptor (Fromme et al., 2016). Although the studies and knowledge of the toxic properties of polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans are limited, the toxic properties shown e.g. lethality, wasting, thymic atrophy, teratogenesis, reproductive effects, chloracne, immunotoxicity, enzyme induction, decreases in T4 and vitamin A, and increased hepatic porphyrins – are the same as for chlorinated dioxins (Vanden-Berg et al., 2013).There are major challenges in assessing risks of chemicals, and usually there are a number of uncertainties involved. It is also complex to estimate the exposure of a substance, as it may involve different pathways, such as dietary, dermal, inhalation and the rate of this exposure (Devito, 2012). To be able to conduct risk evaluations of polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans, the method of toxic equivalence factors was developed (Wikoff et al., 2012). The model is based on the relationship between congeners and their ability to mediate biological and toxic effects in various in vitro and in vivo test systems (Kulkarni, 2019). In order to grade the toxicity of polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans, relative potency factors were determined for individual polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans congeners. The relative potency factors estimate a relative potency compared with 2,3,7,8-tetrachlorodibenzo-p-dioxin, the prototypical congener for this class (Rovira et al., 2010). According to Kannan et al. (2012), the toxic equivalence factor values are estimated according to the following guidelines
Toxic equivalence factor values have been calculated by comparing the ratio of the molar dose of 2,3,7,8-tetrachlorodibenzo-p-dioxin to produce a 50
Materials and Methods
Experimental animals
Female Rattus norvegicus (albino rats) were chosen for this study. They were kept at the animal house section of Department of Biological Sciences, Federal Polytechnic Offa, Kwara State, Nigeria. Randomized block design was used in this study. Thirty-two (32) appearancely healthy adult female albino rats were incorporated in the study. They were kept to acclimatize for fourteen (14) days before treatment and weigh about 179–184 g.
Housing Conditions
The animal house was free of pathogens and other extraneous factors with restricted access. The rats were kept in cages and marked respectively for identification. The animal room’s temperature was maintained at about 22 ◦C (± 3°C) and humidity of at least 30 %. With respect to lightning, the pattern was 12 hours light and 12 hours dark. They were fed with a conventional laboratory diet and water ad libitum. There was adherence with the existing protocols for the use of lab animals strictly.
Drug Source
Lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) were purchased from Yomi-Esthony Enterprise, Nigeria. All other chemicals were obtained from Sigma Chemical Company (Poole, Dorset, United Kingdom). Other chemicals and solvents used in this study were of analytical grade.
Animal Grouping and Treatment
Female albino wistar rats were randomly divided into four (four) groups (A-D) of eight (8) rats each and the treatment protocol was for twenty-eight (28) consecutive days in a single dose as follows;
Group A: The control groups were given ad libitum access to mineral water in 100 ml bottle/day for 28 consecutive days.
Group B: Given lead acetate in a dose of 7 mg/kg body weight by orogastric tube once daily for 28 days.
Group C: Given dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) in a dose of 30 μg/kg body weight by orogastric tube once daily for 28 consecutive days.
Group D: lead acetate in a dose of 7 mg/kg body weight and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) in a dose of 30 μg/kg body weight by orogastric tube once daily for 28 consecutive days.
Termination Of Treatment
Twenty-four hours after the last day of administration, the animals were weighed and were sacrificed using cervical dislocation. After dissection of the abdomen, the liver was removed and fixed in 10 % formol calcium for 24 hours.
Preparations Of Chemicals
Formol calcium: 100 ml of 40 % formaldehyde, 9 g Calcium chloride and 100 ml of distilled water.
Xylene
Graded alcohol
50 % alcohol: 50 ml of absolute alcohol + 50 ml of distilled water.
70 % alcohol: 70 ml of absolute alcohol + 30 ml of distilled water.
90 % alcohol: 90 ml of absolute alcohol + 10 ml of distilled water.
100 % alcohol: Absolute alcohol.
Glycerin
Fixation
The liver tissues were fixed in formol calcium. All tissues were fixed for 24 hours at room temperature. This prevents dehydration and decomposition of the liver tissues.
Dehydration
The recommended procedure of Pattanayak and Majumder (2009) was adopted. All tissues were dehydrated through ascending grades of alcohol by immersion.
Procedure
50% alcohol 1 hour
70% alcohol 1 hour
90% alcohol 1 hour
Absolute alcohol 1 1 hour
Absolute alcohol 2 1 hour
Clearing
The dehydrated tissues were cleared in xylene as follows;
Absolute alcohol and xylene 1 hour
Xylene I 1 hour
Xylene II 1 hour
Impregnation And Infiltration
Molten paraffin wax (melting point 56 °C) was used for infiltration. The wax was placed in an electrically heated, thermostatically regulated oven. The temperature of the oven was maintained at 60 °C. Infiltration was done twice for 1 hour each to remove the clearing agent prior to embedding.
Embedding
Thermostatically fixed (melting point 60°C) wax with 2 Lenkers (L-metal pieces) metal, each of appropriate size were used to form blocks of molten wax. The tissues were then immersed in the block and oriented to desired planes before the solidification in the wax on it.
Trimming
The blocks of tissues were trimmed to a suitable size and shape with a knife, leaving 1-2 mm of embedding medium on both sides. Thereafter the blocks were mounted on wooden blocks that can be held by the Olympus rotary microtome clamps during sectioning.
Sectioning
Each block was clamped on the microtome chuck and was brought a few microns to the knife edge by manual control. Ribbons of tissues were obtained using Olympus rotary microtome. The microtome was set at a thickness of 5 um while the knife was placed at an angle of 45 °C to the block of wax containing the tissue. Ice blocks were applied to the surface of the tissue blocks before and during sectioning to reduce the heat generated. After the trimming, a knife was used for the sectioning proper and ribbons were produced.
Floating Of Sections
The sections were allowed to flatten out in warm water bath to enhance the spreading out of sections.
Mounting Of Sections
To lift out the sections at an angle to the plane of the water, the glass slides were dipped vertically into the warm water.
Dewaxing
The slides were placed on a hot plate for a short period of time, this was to dry the slides. The slides were then stored in slide racks before staining.
Histochemical Techniques
Haematoxylin And Eosin
Technique: Cut sections were;
Dewaxed in two changes of xylene for 3 minutes each.
Hydrated in descending grades of alcohol.
Rinsed in distilled water.
Stained in Haematoxylin for 10 minutes.
Washed in running tap water for 3 minutes.
Differentiated in 1
Results
Table 1: Table Showing the Body Weights of the Animals
| Parameters | Group A | Group B | Group C | Group D |
| Initial weight | 182.3 ± 0.52 | 178.4 ± 0.02 | 181.4 ± 0.05 | 176.1 ± 0.03 |
| Final weight | 224.5 ± 0.81 | 186.2 ± 0.08 | 192.2 ± 0.07 | 184.6 ± 0.07 |
Mean ± SEM; SEM = Standard Error of Mean; Key Group A = Control rats given distilled water throughout the study period. Group B = Experimental rats treated with lead acetate in a dose of 7.0 mg/kg body weight. Group C = Experimental rats treated with dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) in a dose of 30 μg/kg body weight. Group D = Experimental rats treated with lead acetate in a dose of 7.0 mg/kg body weight and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) in a dose of 30 μg/kg body weight
Microscopic Plates of The Liver
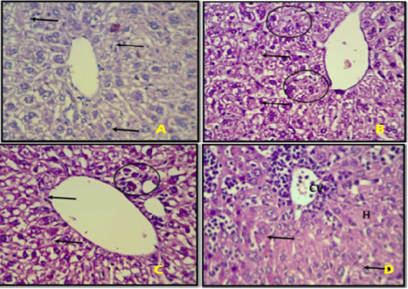
Figure 1: Photomicrography of the Experimental Rats.
(A) Photomicrograph of a Liver section of adult albino rats in control group (Group A), showing normal central vein (CV). The sinusoids are not occluded (S); the hepatocytes appear normal (H), and the central portal vein is devoid of any occlusion (CV). (B) Photomicrograph of a Liver section of rats treated with lead acetate in a dose of 7 mg/kg body weight (Group B), showing mild degenerative changes of the hepatocyte and dilation of the central vein. (C) Photomicrograph of a Liver section of rats treated with dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) in a dose of 30 μg/kg body weight (Group C), showing dilation of the central vein (arrows). (D) Photomicrograph of a Liver section rats treated with lead acetate in a dose of 7 mg/kg body weight and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) in a dose of 30 μg/kg body weight showing marked degeneration of hepatocytes (H) and occlusion of the central portal vein (CV) (H & E X400).
Discussion
Environmental pollution is the presence of a pollutant in an environment such as air, water, soil, and consequently in food which may be toxic and will cause harm to living things in the polluted environment. Lead and dioxin in the environment are considered the most dangerous toxins and pollutants that cause neurological disorders (Rubin and Strayer, 2018). In addition, dioxin and lead acetate caused liver toxicity in albino rat (Nault et al., 2021; Aldahmash and El-Nagar, 2016). The present study investigates toxicity of lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) in the histology of the liver of adult female albino rats.Body weight is important indicators of the toxic effect of any exogenous compound on the body. In the present study, lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) exposed rats registered less gain in body weight as compared with the control. This decrease in gain of body weight might be associated with lack of food consumption occurring due to loss of appetite or gastrointestinal disturbances (Bhattacharjee et al., 2016). Similar decrease in body weight has also been observed by Ciftci and Ozdemir (2013) in rats exposed to dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin). Bhattacharjee et al. (2016) registered decrease in body weight in rats fed lead acetate. Dardouri et al. (2016) reported decrease in body weight of rats due to combined effects of Cadmium and Mercury in wistar rats.
Light microscopic examination of control group (group A) revealed normal hepatic architecture. Haematoxylin and Eosin (H & E) stained sections of liver of the control animals (group A) revealed normal characteristic of hepatic architecture; the hepatic lobules appeared to be made up of hepatocytes arranged in cords radiating from the central veins. The histological outline of the liver from the rats in the control group was well preserved. The liver sections of group B which were treated with lead acetate showed degenerative changes of the hepatocyte and dilation of the central vein as shown in Figure 4.1B. Mild swelling of the hepatocytes in places with areas of necrosis, inflammatory leukocytic infiltration, and moderate congestion of central vein were found in rats administered with dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) (group C). The liver cells were degenerated with necrosis, severe congestion and hemorrhage hypertrophy of hepatocytes, hepatocyte swelling, was evident in rats fed with combination of lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) (group D).Histopathological alterations in the liver due to lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) administration causes liver damage as evidenced by the structural changes, i.e., disruption of the normal architecture, necrosis, cellular infiltration, and leading to pathological damage. Similar histopathological alterations in liver of rats after ingestion of lead acetate have been reported by Aldahmash and El-Nagar (2016). Nault et al. (2021) have also reported that dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) causes liver damage including changes in the normal histology, increased permeability and necrosis of hepatocytes. The liver is well-known target organ of the toxic impact regarding its function in biotransformation and excretion of xenobiotics. After entering uptake, liver is the first organ to be exposed by portal circulation. Hepatotoxicity is toxicity to the liver, bile duct, and gall bladder. The liver is particularly susceptible to xenobiotics due to a large blood supply and its role in metabolism. In this study, after oral administration of lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin), histopathological changes were observed in the liver of rats in all treatment groups compared with control group, most especially, in the rats co-exposed to lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) showed severe histopathological changes in the liver.The most pronounced effect of these metals was observed in the groups of rats co-exposed to lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin). The combined effects of different metals on the liver of experimental animals were studied by many investigators. These results are in accordance with the study of Bhattacharjee et al. (2016) who revealed that necrotic lesions, congestion areas, and occlusion of central vein were more prominent in the rat liver treated with combination of Lead, Cadmium and Arsenic than when exposed to single metal. Dardouri et al. (2016) reported that liver tissue was completely damaged and qualitative degenerative and necrotic changes were observed in almost all structures of the liver tissues after Cadmium and Mercury co-exposure in wistar rats. Ezedom et al. (2020) reported that wistar rat’s liver co-exposed to Cadmium and Arsenic showed changes in hepatic trabecular structure and increased hepatocytes with cytoplasmatic vacuoles. Results showed that lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) co-exposure induced drastic histopathological effects on the liver of rats. Lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) co-exposure causes liver damage which includes dilation of sinusoidal spaces with irregular nuclear shape, degenerative changes include binucleated cells, hypertrophy of hepatocytes and lymphocytic infiltration in the central vein.
Conclusion
The present study demonstrates the effects of lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) on the histology of the rat liver. It may be concluded from the study that exposure to combination of metals produced more deleterious effects than when expose to single metal. The study revealed that lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) co-exposure caused deleterious effects on rat’s liver which was evidenced in histological changes.
Recommendations
This study shows that lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) exposure is associated with deleterious effects in the liver. Thus, people should avoid exposure to lead acetate and dioxin (2,3,7,8-tetrachlorodibenzo-p-dioxin) because it may have adverse effect on the histology of the liver.
References
- Aldahmash, B. A. and El-Nagar, D. M. (2016). Antioxidant Effects of Captopril against Lead Acetate-induced Hepatic and Splenic Tissue Toxicity in Swiss Albino Mice. Saudi Journal of Biological Science, 23(6):667-673.
Publisher | Google Scholor - Barone, G., Storelli, A., Busco, A., Mallamaci, R. and Storelli, M. M. (2019). Polychlorinated Dioxins, Furans and Dioxin–like Polychlorinated Biphenyls in Food from Italy: Estimates of Dietary Intake and Assessment. Journal of Food Science, 86:4741-4753.
Publisher | Google Scholor - Bas, H., Kalender, Y., Pandir, D. and Kalender, S. (2015). Effects of Lead Nitrate and Sodium Selenite on Deoxyribonucleic Acid Damage and Oxidative Stress in Diabetic and Non-diabetic Rat Erythrocytes and Leucocytes. Toxicology Pharmacology, 9:1019-1026.
Publisher | Google Scholor - Bellinger, D., Leviton, A., Waternaux, C., Needleman, H. and Rabinowitz, M. (2017). Longitudinal Analyses of Prenatal and Postnatal Lead Exposure and Early Cognitive Development. England Journal Medicine; 316:1037-1043.
Publisher | Google Scholor - Bellinger, D. C. (2014). Lead. Pediatrics, 113 (4 SuPagel):1016-1022.
Publisher | Google Scholor - Benjelloun, M., Tarrass, F., Hachim, K., Medkouri, G., Benghanem, M. G. and Ramdani, B. (2017): Chronic Lead Poisoning: A “Forgotten” Cause of Renal Disease Saudi. Journal of Kidney Disease Transplant, 18:83-86.
Publisher | Google Scholor - Bergeson, L. L. (2018). The Proposed Lead National Ambient Quality Standard: Is Consideration of Cost in the Clean Air Act ‘s Future? Environmental Quality Management 18:79-84.
Publisher | Google Scholor - Bhattacharjee, T., Bhattacharjee, S. and Choudhuri, D. (2016). Hepatotoxic and Nephrotoxic Effects of Chronic Low Dose Exposure to a Mixture of Heavy Metals – Lead, Cadmium and Arsenic. International Journal of Pharmaceutical, Chemical and Biological Sciences, 6(1):39-47.
Publisher | Google Scholor - Bi, C., Chen, Y., Zhao, Z., Li, Q., Zhou, Q., Ye, Z. and Ge, X. (2020). Characteristics, Sources and Health Risks of Toxic Species (Polychlorinated Dibenzo-Pdioxins/Dibenzofurans, Polycyclic Aromatic Hydrocarbons and Heavy Metals) in Particulate Matter 2. 5 During Fall and Winter in an Industrial Area. Chemosphere, 238:124-620.
Publisher | Google Scholor - Breeher, L., Mikulski, M. A., Czeczok, T., Leinenkugel, K. and Fuortes, L. J. (2015). A Cluster of Lead Poisoning among Consumers of Ayurvedic Medicine. Intentional Journal of Occupational Environmental Health, 82-98.
Publisher | Google Scholor - Brunton, L. L., Goodman, L. S., Buxton, I. and Parker, K. L. (2017). Goodman and Gillmans Manual of Pharmocology and Theraupetics. Mcgraw Hill Professional, 216-225.
Publisher | Google Scholor - Busselberg D., Evans, M. L., Haas, H. L. and Carpenter, D. O. (2013). Blockade of Mammalian and Invertebrate Calcium Channels by Lead. Neurotoxicology; 14:249-258.
Publisher | Google Scholor - Chisolm, J. and Harrison, H. (2016). The Exposure of Children to Lead. Journal of American Academic Pediatrics; 18:943-958.
Publisher | Google Scholor - Ciftci, O., Vardi, N. and Ozdemir, I. (2013). Effects of Quercetin and Chrysin on 2,3,7,8-tetrachlorodibenzo-p-dioxin Induced Hepatotoxicity in Rats. Environmental Toxicology; 28(3):146-154.
Publisher | Google Scholor - Cleveland, L. M., Minter, M. L., Cobb, K. A., Scott, A. A. and German, V. F. (2018). Lead Hazards for Pregnant Women and Children. American Journal of Nursing, 108:40-49.
Publisher | Google Scholor - Cohen, A. R., Trotzky, M. S. and Pincus D. (2015): Reassessment of the Microcytic Anemia of Lead Poisoning. Pediatrics, 67:904-906.
Publisher | Google Scholor - Dardouri, K., Haouem, S., Gharbi, I., Sriha, B., Haouas, Z., El-Hani, A. and Hammami, M. (2016). Combined Effects of Cadmium and Mercury on Liver and Kidney Histology and Function in Wistar Rats. Journal of Agricultural Chemistry and Environment; 5:159-169.
Publisher | Google Scholor - Dart, R. C., Hurlbut, K. M. and Boyer-Hassen, L. V. (2014). Lead in Medical Toxicology (3rd Edition. Lippincott Williams and Wilkins, 312-323.
Publisher | Google Scholor - Devito M. (2012). Toxicity Equivalence Factors for Dioxin and Related Compounds. Dioxins and Health. John Wiley & Sons. Incorporation, 37-52.
Publisher | Google Scholor - Ekong, E. B., Jaar, B. G. and Weaver, V. M. (2016). Lead-related Nephrotoxicity: A Review of the Epidemiologic Evidence. Kidney International, 70:2074-2084.
Publisher | Google Scholor - Ezedom, T., Asagba, S. and Tonukari, N. J. (2020). Toxicological Effects of the Concurrent Administration of Cadmium and Arsenic through the Food Chain on the Liver and Kidney of Rats. The Journal of Basic and Applied Zoology, 81(16):1-12.
Publisher | Google Scholor - Fromme, H., Hilger, B., Albrecht, M., Gries, W., Leng, G. and Volkel, W. (2016). Occurrence of Chlorinated and Brominated Dioxins/Furans, Polybominated Biphenyl and Brominated Flame Retardants in Blood of German Adults, International Journal of Hygiene and Environmental Health. 219:380-388.
Publisher | Google Scholor - Gonzalez, N. and Domingo, J. L. (2021). Polychlorinated dibenzo–p–dioxins and Dibenzofurans (PCDD/Fs) in Food and Human Dietary Intake: An Update of the Scientific Literature. Food Chemistry Toxicology, 157:112-585.
Publisher | Google Scholor - Goto, A., Tue, N. M., Someya, M., Isobe, T., Takahashi, S. and Tanabe, S. (2017). Spatiotemporal Trends of Polybrominated dibenzo-p-dioxins and Dibenzofurans in Sediments from Tokyo Bay, Japan. Science of the Total Environment; 52:340-347.
Publisher | Google Scholor - Gotz, R., Bergemann, M., Stachel, B. and Umlauf, G. (2017). Dioxin in the River Elbe. Chemosphere, 183:229-241.
Publisher | Google Scholor - Guo, W., Pan, B., Sakkiah, S., Yavas, G., Ge, W., Zou, W., Tong, W. and Hong, H. (2019). Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods. Journal of Environmental Research Public Health, 16 4361.
Publisher | Google Scholor - He, Y., Von-Lampe, K., Wood, L. and Kurti, M. (2015). Investigation of Lead and Cadmium in Counterfeit Cigarettes Seized in the United States. Food Chemistry Toxicology; 81:40-45.
Publisher | Google Scholor - Henretig, F. M. (2016). Lead in Goldfrank’s Toxicoliogic Emergencies. 8th Edition. McGraw Hill Professional, 212-18.
Publisher | Google Scholor - Ishiniwa, H., Sakai, M., Tohma, S., Matsuki, H., Takahashi, Y. and Kajiwara, H. (2013). Dioxin Pollution Disrupts Reproduction in Female Japanese Field Mice. Ecotoxicology, 22: 1335-1347.
Publisher | Google Scholor - Jurdziak, M., Gac, P., Martynowicz, H. and Poręba, R. (2015). Function of Respiratory System Evaluated using Selected Spirometry Parameters in Persons Occupationally Exposed to Lead without Evident Health Problems. Journal of Environmental Toxicology Pharmacology, 39:1034-1040.
Publisher | Google Scholor - Kannan, K., Liao, C. and Moon, H. B. (2012). Polybrominated Dibenzo-P-Dioxins and Dibenzofurans. John Wiley and Sons, Incorporation, 255-302.
Publisher | Google Scholor - Karri, S. K., Saper, R. B. and Kales, S. N. (2018). Lead Encephalopathy Due to Traditional Medicines. Journal of Environmental Health Perspective, 3:54-59.
Publisher | Google Scholor - Knutsen, H. K., Alexander, J., Barregård, L., Bignami, M., Bruschweiler, B., Ceccatelli, S., Cottrill, B., Dinovi, M. and Edler, L. (2018). Risk for Animal and Human Health Related to the presence of Dioxins and Dioxin–like Polychlorinated Biphenyls in Feed and Food. European Food Safety Authority Journal,16:53-58.
Publisher | Google Scholor - Kulkarni, P. (2019). Dioxins. Encyclopedia Environmental Health,2:125-134.
Publisher | Google Scholor - Kosnett, M. J. (2015). Lead. In Brent, J. Critical Care Toxicology: Diagnosis and Management of the Critically Poisoned Patient. Gulf Professional Publishing, 357-362.
Publisher | Google Scholor - Lan, P. and Muralitharan, N. (2015). Anatomy and Physiology for Nurses at a Glance. Wiley-Blackwell Company, 320-324.
Publisher | Google Scholor - Landrigan, P. J., Schechter, C. B., Lipton, J. M., Fahs, M. C. and Schwartz, J. (2012). Environmental Pollutants and Disease in American Children. Journal of Environmental Health Perspective,110:721-728.
Publisher | Google Scholor - Levin, R., Brown, M. J., Kashtock, M. E., Jacobs, D. E., Whelan, E. A. and Rodman, J. (2018). Lead Exposures in U.S. Children, 2018: Implications for Prevention. Journal of Environmental Health Perspective, 116:1285-1293.
Publisher | Google Scholor - Li, M., Zhou, Y., Wang, G., Zhu, G., Zhou, X., Gong, H., Sun, J., Wang, L. and Jinsong, L. (2021). Evaluation of Atmospheric Sources of Polychlorinated dibenzo–p–dioxins and Dibenzofurans around a Municipal Solid Waste Incineration Plant Using Active and Passive Air Samplers. Chemosphere, 274:129-685.
Publisher | Google Scholor - Lv, C., Xu, Y., Wang, J., Shao, X., Ouyang, J. and Li, J. (2015). Dysplastic Changes in Erythroid Precursors as a Manifestation of Lead Poisoning: Report of a Case and Review of Literature. International Journal of Clinical Experimental Pathology; 8:818-823.
Publisher | Google Scholor - Mahaffey, K. R. (2016). Environmental Lead Toxicity: Nutrition as a Component of Intervention. Journal of Environmental Health Perspective, 89:75-78.
Publisher | Google Scholor - Mai, F. M. (2016). Beethoven’s Terminal Illness and Death. Journal of Royal College Physicians. 36:258-263.
Publisher | Google Scholor - Marino, P. E., Landrigan, P. J., Graef, J., Nussbaum, A., Bayan, G., Boch, K. and Boch, S. (2016): A Case Report of Lead Paint Poisoning during Renovation of a Victorian Farmhouse. American Journal of Public Health, 80:1183-1185.
Publisher | Google Scholor - Merill, J. C., Morton, J. J. P. and Soileau, S. D. (2017). Metals in Principles and Methods of Toxicology (5th Edition) Chemical and Rubber Company Press, 21-30.
Publisher | Google Scholor - Meyer, P. A., McGeehin, M. A. and Falk, H. (2013): A Global Approach to Childhood Lead Poisoning Prevention. Internal Journal of Environmental Health; 206: 363–369.
Publisher | Google Scholor - Mikolajczyk, S., Warenik-Bany, M. and Pajurek, M. (2021). Polychlorinated Dibenzo-Pdioxins/Dibenzofurans and Polychlorinated Biphenyls in Baltic Fish – Recent Data, Risk for Consumers. Marine Pollution Bulletin, 171:112-763.
Publisher | Google Scholor - Mycyk, M., Hryhorcu, D. and Amitai, Y. (2015). Lead. Paediatric Toxicology: Diagnostic and Management of the Poisoned Child. Mcgraw Hill Professional, 112-130.
Publisher | Google Scholor - Nault, R., Fader, K. A., Bhattacharya, S. and Zacharewski, T. R. (2021). Single-Nuclei RNA Sequencing Assessment of the Hepatic Effects of 2,3,7,8-Tetrachlorodibenzo-p-dioxin. Cell Molecular Gastroenterological Hepatology, 11(1):147-159.
Publisher | Google Scholor - Navas-Acien, A., Guallar, E., Silbergeld, E. K. and Rothenberg, S. J. (2017). Lead Exposure and Cardiovascular Disease - A Systematic Review. Environmental Health Perspective, 115: 472-482.
Publisher | Google Scholor - Needlemann, H. L., Schell, A., Bellinger, D., Leviton, A. and Allred, E. N. (2016). The long-term Effects of Exposure to Low Doses of Lead in Childhood - An 11-Year Follow-up Report. England Journal of Medicine, 322:83-88.
Publisher | Google Scholor - Roy, A., Queirolo, E., Martinez, G. and Kordas, K. (2015). Association of Blood Lead Levels with Urinary F2-8α Isoprostane and 8-Hydroxy-2-deoxy-guanosine Concentrations in First-grade Uruguayan Children. Environmental Research, 140:127-135.
Publisher | Google Scholor - Rubin, R. and Strayer, D. S. (2018). Environmental and Nutritional Pathology in Rubinspathology, Clinicopathologic Foundations of Medicine. Lippincott Williams & Wilkins, 112-118.
Publisher | Google Scholor - Pajurek, M., Warenik–Bany, M. and Mikolajczyk, S. (2023). Dioxin Transfer Simulation from Feed to Animal Tissues and Risk Assessment. Chemosphere, 313:137-379.
Publisher | Google Scholor - Pajurek, M., Warenik–Bany, M. and Mikolajczyk, S. (2022): Feed as a Source of Dioxins and Polychlorinated Biphenyls. Chemosphere308:136-243.
Publisher | Google Scholor - Patrick, L. (2016): Lead Toxicity, A Review of the Literature. Part 1: Exposure, Evaluation, and Treatment. Alternative Medicine Review, 11:2-22.
Publisher | Google Scholor - Park, S. K., O’Neill, M. S., Vokonas, P. S., Sparrow, D., Wright, R. O., Coull, B., Nie, H., Hu, H., and Schwartz, J. (2018): Air Pollution and Heart Rate Variability: Effect Modification by Chronic Lead Exposure. Epidemiology, 19:111-120.
Publisher | Google Scholor - Pearce, J. M. S. (2017). Burton’s Line in Lead Poisoning. European Neurology; 57: 118–119.
Publisher | Google Scholor - Pearson, H. A. and Schonfeld, D. J. (2013). Lead in Rudolph’s Pediatrics (21st Edition) McGraw Hill professional, 118-125.
Publisher | Google Scholor - Pius, C., Koosaletse-Mswela, P., Sichilongo, K. and Dikinya, O. (2020). Mapping Polychlorinated Dibenzo-Pdioxins/Dibenzofurans in Soils around Pugu Municipal Dump Site in Dares Salaam, Tanzania: Implications on Dermal and Soil Ingestion Exposure for People in the Peripheral. Environmental Pollution, 258:113-125.
Publisher | Google Scholor - Rovira, J., Mari, M., Nadal, M., Schuhmacher, M. and Domingo, J. L. (2010). Environmental Monitoring of Metals, Polychlorinated Dibenzo-Pdioxins/Dibenzofurans and Polychlorinated Biphenyls as a Complementary Tool of Biological Surveillance to Assess Human Health Risks. Chemosphere, 80(10):1183-1189.
Publisher | Google Scholor - Saladin, K. S. (2018): Anatomy and Physiology - The Unity of Form and Function, 7th Edition. Dubuque. IA: McGraw-Hill Higher Education, 543-579.
Publisher | Google Scholor - Schoeters, G., Hond, E. D., Dhoogem, W., Larebekem N, V. and Leijs, M. (2018): Endocrine Disruptors and Abnormalities of Pubertal Development. Basic & Clinical Pharmacology & Toxicology, 102:168-175.
Publisher | Google Scholor - Sindiku, O., Babayemi, J. O., Tysklind, M., Osibanjo, O., Weber, R. and Watson, A. (2015): Polybrominated Dibenzo-p-dioxins and Dibenzofurans (PBDD/Fs) in E-waste Plastic in Nigeria. Environmental Science and Pollution Research, 22:14515-14529.
Publisher | Google Scholor - Sokol, R. Z. and Berman, N. (2015): The Effect of Age of Exposure on Lead-induced Testicular Toxicity. Toxicology, 69:269-278.
Publisher | Google Scholor - Timbrell, J. A. (2018): Biochemical Mechanisms of Toxicity: Specific Examples. Principles of Biochemical Toxicology (4th Edition): Information Health Care.
Publisher | Google Scholor - Vanden-Berg, M., Denison, M. S., Birnbaum, L. S., DeVito, M. J., Fiedler, H. and Falandysz, J. (2013). Polybrominated dibenzo-p-dioxins, Dibenzofurans, And Biphenyls: Inclusion in the Toxicity Equivalency Factor Concept for Dioxin-like Compounds. Toxicological Sciences, 133:197-208.
Publisher | Google Scholor - Wadi, S. and Ahmad, G. (2019). Effects of Lead on the Female Reproductive System in Mice. Journal of Toxicology Environmental Health, 56:513-521.
Publisher | Google Scholor - Wedeen, R. P., Maesaka, J. K., Weiner, B., Vitale, L. F. and Joselow, M. M. (2015). Occupational Lead Nephropathy. American Journal of Medicine, 59:630-641.
Publisher | Google Scholor - Wikoff, D., Fitzgerald, L. and Birnbaum, L. (2012). Persistent Organic Pollutants: An Overview. Dioxins and Health. John Wiley & Sons, Incorporation, 1-35.
Publisher | Google Scholor - World Health Organization (WHO). (2010). Preventing Disease through Healthy Environments - Exposure to Dioxins and Dioxin–Like Substances: Public Health Concern.
Publisher | Google Scholor - Yu, C. C., Lin, J. L. and Lin-Tan, D. T. (2014). Environmental Exposure to Lead and Progression of Chronic Renal Diseases: A Four-Year Prospective Longitudinal Study. Journal of American Social Nephrology, 15:1016-1022.
Publisher | Google Scholor - Zhang, M., Buekens, A. and Li, X. (2016). Brominated Flame Retardants and the Formation of Dioxins and Furans in Fires and Combustion. Hazardous Materials, 304:26-39.
Publisher | Google Scholor