Research Article
Improving Patient Compliance in Hypertension Treatment: Development of Hydrochlorothiazide ODTs with Enhanced Solubility
- Aman Kumar
- Tushar Sonare
- Krutika Mandloi
- Krutika Mandloi
- Akash Yadav *
- Dinesh Kumar Jain
IPS Academy College of Pharmacy, Rajendra Nagar, A.B. Road, Indore, India.
*Corresponding Author: Akash Yadav, IPS Academy College of Pharmacy, Rajendra Nagar, A.B. Road, Indore, India.
Citation: Kumar A, Sonare T, Mandloi K, Khadsondni K, Yadav A, et, al. (2025). Improving Patient Compliance in Hypertension Treatment: Development of Hydrochlorothiazide ODTs with Enhanced Solubility. Addiction Research and Behavioural Therapies, BRS Publishers. 4(1):1-17. DOI: 10.59657/2837-8032.brs.25.036
Copyright: © 2025 Akash Yadav, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: February 11, 2025 | Accepted: March 08, 2025 | Published: March 15, 2025
Abstract
The development of oral disintegrating tablets (ODTs) for antihypertensive drugs represents a promising approach to improve patient adherence, especially for those with difficulty swallowing traditional tablets. This study focuses on the formulation and optimization of hydrochlorothiazide (HCTZ) ODT using Box-Behnken design (BBD) for process optimization. The research employs excipients such as fenugreek seed powder, Cross-povidone and Locust bean gum as a super disintegrant, Banana powder as binders, corn starch as a glidant, magnesium stearate as a lubricant, microcrystalline cellulose (MCC) as a filler, and aspartame as a sweetener. Additionally, polyethylene glycol 4000 (PEG-4000) is used as a solubility enhancer through solid dispersion techniques. The BBD method investigates the effect of concentrations of fenugreek seeds mucilage, Locust bean gum and PEG-4000 on tablet properties such as disintegration time, dissolution rate, hardness, friability etc. The optimized formulation exhibits rapid disintegration (within 30 seconds), improved dissolution profile and enhanced bioavailability of hydrochlorothiazide due to the solubility-enhancing properties of PEG-4000. The results indicate that the combination of excipients and solid-phase dispersion methods can significantly improve the efficacy of ODT, providing a potential solution to improve therapeutic outcomes in the treatment of Hypertension.
Keywords: hypertension; oral disintegrating tablets (ODTs); hydrochlorothiazide; box-behnken design (BBD); super disintegrants; solid dispersion; solubility enhancement
Introduction
Hypertension is a significant contributor to illness and death globally. It ranks among the top causes of cardiovascular problems, such as ischemic heart disease, stroke, and heart failure, as well as chronic kidney disease and overall mortality rates. The number of individuals affected by hypertension is anticipated to rise as the population ages. About 10% to 15% of patients receiving treatment for hypertension experience resistant hypertension. Without confirmatory tests, resistant hypertension is characterized by blood pressure readings that remain above the target, despite the patient using three or more antihypertensive medications, usually including a diuretic, or needing four or more medications to manage blood pressure effectively [1-4].
Enhancing therapies for managing resistant hypertension is essential since various studies indicate that individuals with this condition face a heightened risk of adverse outcomes. In large retrospective studies involving 200,000 to 400,000 patients, those suffering from resistant hypertension were nearly twice as likely to experience cardiovascular events—such as heart attacks, ischemic heart disease, heart failure—as well as strokes, chronic kidney disease (CKD) or end-stage renal disease, and mortality compared to their counterparts without resistant hypertension. Furthermore, resistant hypertension is linked to poorer outcomes in patients with certain comorbidities, including CKD, ischemic heart disease, obesity, diabetes, and obstructive sleep apnea. As a result, resistant hypertension imposes significant medical and financial challenges due to treatment expenses, disability, and early death. Thus, it is vital to explore more effective approaches for managing patients with resistant hypertension to enhance their overall prognosis [5-6].
Diuretics are a diverse group of medications primarily designed to promote increased urine production. They generally work by altering the movement of ions at various points in the nephron, thus encouraging the kidneys to eliminate water and electrolytes, mainly sodium and chloride. These drugs are classified based on their method of action and location of effect into categories such as carbonic anhydrase inhibitors, osmotic diuretics, thiazides, loop (or high-ceiling) diuretics, and potassium-sparing diuretics. Thiazide diuretics are the most frequently prescribed antihypertensives and are sometimes used in combination with potassium-sparing diuretics. Loop diuretics, on the other hand, are reserved for specific situations [7].
Hydrochlorothiazide
Hydrochlorothiazide (HCTZ) is a thiazide diuretic that has been in clinical use for over fifty years. It functions by blocking the sodium-chloride cotransporter system located in the distal convoluted tubules of the kidneys, leading to its diuretic effect as water follows the increased potassium and sodium within the nephron. HCTZ is widely prescribed and is recognized as a primary treatment option for high blood pressure. Its popularity stems from its affordability, efficacy, and its ability to address additional conditions like heart failure and fluid retention. Despite its common prescription, HCTZ can lead to several typical side effects, including imbalances in electrolytes, elevated blood sugar levels, increased lipid levels, and higher uric acid levels. Nevertheless, health authorities continue to recommend it as the main option for managing hypertension. HCTZ can also result in low potassium levels, low sodium levels, high calcium levels, and low magnesium levels, along with potential kidney issues in patients with compromised renal function [3]. Recommended dosages for HCTZ suggest an intake of 25 mg to 100 mg daily for edema, while 25 mg to 50 mg is advised for those dealing with high blood pressure [8]. Hydrochlorothiazide has the ability to activate calcium-dependent potassium channels, leading to hyperpolarization of vascular smooth muscle cells.
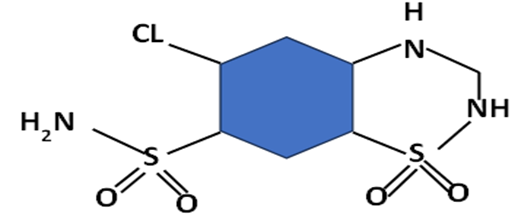
Figure 1: Hydrochlorothiazide
This process results in the closure of calcium channels, ultimately reducing calcium influx and preventing vasoconstriction. Additionally, hydrochlorothiazide inhibits carbonic anhydrase in blood vessels, which may alter the pH within smooth muscle cells and support the mechanism mentioned above. While these effects have been extensively studied in laboratory settings, their specific contribution to the antihypertensive action of thiazides remains uncertain. It is clear, however, that the primary effect targets the sodium-chloride cotransporter, as thiazides exhibit a diminished antihypertensive response in patients with kidney dysfunction. Furthermore, individuals with functional mutations in the SLC12A3 gene tend to have lower blood pressure compared to those without such mutations [9-10].
Oral Disintegrating tablets (ODTs)
These fast-dissolving tablets provide an effective solution for patients who struggle with taking medications orally, including children, individuals receiving medical therapies, or those dealing with mental health issues. Created from a soft molded matrix that has low porosity and compressive strength, these tablets quickly dissolve in saliva, negating the need for water and offering an easy method of delivery for those facing oral intake challenges. They greatly improve the overall experience for patients who have trouble swallowing, ultimately enhancing treatment results while remaining easy to use [11-12].
Orodispersible tablets (ODTs) are solid medication forms that dissolve in the mouth within seconds, without needing to be chewed or swallowed. They offer a potential way to enhance patient adherence and make medication administration easier for elderly individuals, children, and those with upper gastrointestinal tract issues. A variety of ODTs are already on the market, including options like pain relievers, blood pressure medications, and antidepressants. Recently, there has been increased interest in creating ODTs with active ingredients such as naloxone, hydramine, metformin, and probiotics.
Figure 2: Oral disintegrating tablet
ODTs must meet high standards, particularly regarding their disintegration speed. The European Pharmacopoeia stipulates that ODTs should disintegrate in no more than 3 minutes. In contrast, the requirements set by the USP and FDA are more stringent, allowing a maximum disintegration time of just 30 seconds. Consequently, these tablets need to have adequate porosity or incorporate an efficient super disintegrant [13-14].
Box-Behnken Experimental Design
Response Surface Methodology (RSM) investigates the connection between control and response variables. It is crucial to optimize the test design to lower costs, necessitating careful thought regarding the mix and levels involved. Techniques like full factorial, Box-Behnken Design (BBD), and central composite designs play a vital role in this optimization. The Box-Behnken Design, utilizing Design Expert® software (version 7.0, Stat-Ease Inc.), allows for three levels and ensures reliability across 15 runs with five repetitions. A second-order polynomial model was created to represent the measured variables. This approach speeds up the development of pharmaceutical data and enhances research efficiency by delivering powerful, straightforward, and optimized research solutions [15-16].
Materials and Methods
Hydrochlorothiazide is given as gift sample by (Unichem Laboratories Ltd, District Kolhapur Maharashtra–416 236), microcrystalline cellulose (Maple Biotech Pvt Ltd., Pune, India), Superdisintegrants (G. M. Herbal medicine store in Indore). Talc and magnesium stearate were purchased from S. D. Fine Chem Ltd., Mumbai, India. All other solvents and reagents were purchased from the market.
Extraction
Locust Bean Gum: Carob pods are crushed to separate the seeds from the pulp. The steps involved in processing carob seeds include dehusking (using acid or thermo-mechanical methods), splitting, milling, sifting, clarification, and drying. This processing can be quite challenging due to the hard and tough outer layer of the seeds. To remove the outer shell of carob kernels, one can either treat them with dilute sulfuric acid or use thermo-mechanical techniques known as acid peeling and thermal peeling, respectively. In the acid peeling method, carob seeds are exposed to sulfuric acid at high temperatures to break down the seed coat. The remnants of the seed coat are then washed away from the endosperm through an effective washing and brushing process. The cleaned kernels are dried and cracked, causing the brittle germ to be crushed. These germ pieces are separated from the unbroken halves of the endosperm, referred to as splits.
Figure 3: Locust bean gum
The carob bean gum produced from these splits is light in color and has a higher viscosity. In the thermo-mechanical peeling method, carob kernels are roasted in a rotating furnace, causing the seed coat to pop off from the inner parts. After mechanical processing, the endosperm halves or carob splits are collected from the roasted outer layer or husk and the crushed germ.
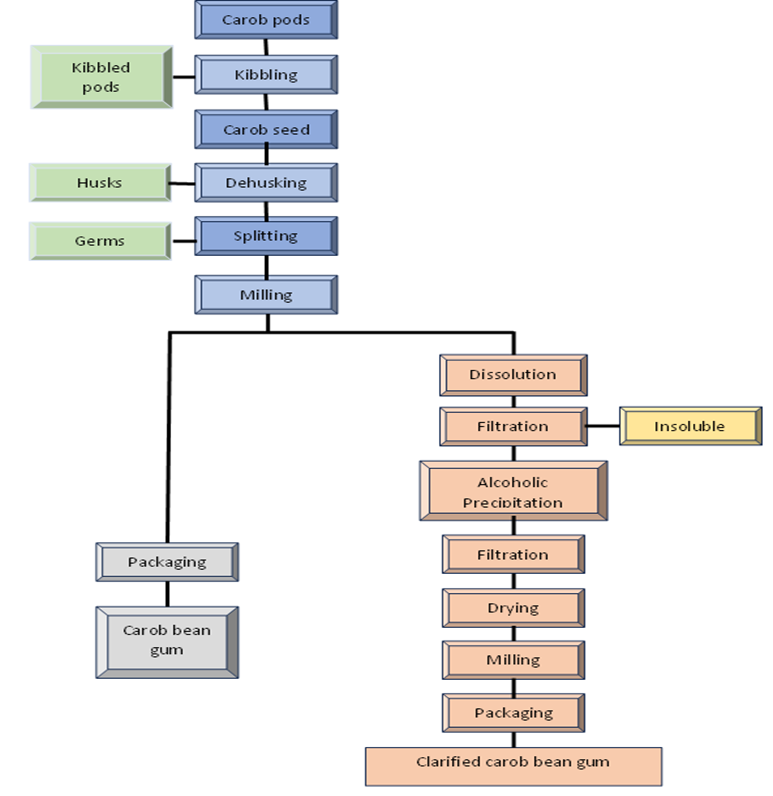
Figure 4: Flow diagram for manufacturing of locust bean gum
These isolated endosperm halves are then milled and screened to create a fine powdered form of native carob bean gum or locust bean gum (Figure 4). The resulting product tends to be darker due to the heating or roasting process. This thermo-mechanical method produces no waste since it does not use sulfuric acid during processing. To clarify the native locust bean gum, it can be dispersed in water and dissolved through heating. This solution is then filtered to remove any insoluble materials, resulting in a clear liquid. Locust bean gum is recovered by using isopropanol or ethanol for precipitation, followed by filtering, drying, and milling to create a fine powder of clarified or purified carob bean gum [17-19].
Fenugreek seed mucilage: FSM was extracted from fenugreek seeds following established procedures found in the literature. Two hundred grams of fenugreek seeds were immersed in 1.5 liters of distilled water at room temperature for 12 hours. The mixture was then heated in a water bath until a slurry was formed. Once it cooled, the slurry was placed in the refrigerator overnight to allow undissolved particles to settle.
Figure 5: Fenugreek seed mucilage
The clear liquid on top was decanted and concentrated in a water bath at 60 °C to one-third of its original volume. After cooling to room temperature, this solution was mixed with three times its volume of acetone while stirring continuously. The resulting precipitate was washed several times with acetone and allowed to dry at room temperature for 24 hours. The isolated substance was then passed through an 80-mesh sieve and stored in desiccators for future use [20].
Banana powder: The ripe banana fruits peeled off, sliced transversely (2mm thickness), incubated in 0.5 percentage (w/v) sodium metabisulphite solution for 5 minutes and dried in oven at 60°C for 18 hrs the dried slices were grinded in mill and passed through sieve (mesh size 60) to obtain CBP [21].
Figure 6: Ripe banana and powder
Solid Dispersion Technique: A solid dispersion method called solvent evaporation is used to improve the solubility, rate of dissolution, and bioavailability of medications that are not very soluble in water, such as hydrochlorothiazide. By dispersing the medication within a hydrophilic polymer, it improves wettability, decreases particle size, and inhibits recrystallization. This approach is perfect for fast-release formulations since it is affordable, scalable, and appropriate for medications that are heat-sensitive. HCTZ was made into a solid dispersion by employing the fusion–solvent process. Accurate weights of PEG-4000 and HCTZ were obtained. PEG-4000 was melted in a water bath, and HCTZ was dissolved in methanol. The molten PEG-4000 was vigorously stirred while the medication solution was gradually added. Prior to being granulated through a mesh no. 20, the residue was dried in a desiccator for 24 hours after the solvent was evaporated over a water bath.
Box-Behnken Experimental Design: Using Design Expert® software (version 7.0, Stat-Ease Inc.), the Box-Behnken Experimental Design was utilized to create hydrochlorothiazide tablets that were optimized for quick drug release. The study examined the effects of three factors at three distinct levels, ranging from low to high: locust bean gum, fenugreek seed mucilage, and Crospovidone. This comprehensive approach allowed for the identification of the optimal combination of ingredients to achieve fast-dissolving tablets with optimal efficacy. 15 experimental runs were carried out using the three-factor and three-level design concepts, with three central points to evaluate experimental error and design accuracy. As shown in Table 1, mean tablet hardness, in vitro dissolving time, and in vitro disintegration time were the main responses or dependent variables evaluated.
Selected Independent variables and Dependent variables
The selected independent variables are displayed in Table 1.
Table 1: Independent variables with levels
| Factor | Name | Unit | Minimum | Maximum |
| A | Locust bean gum | % | 0 | 10 |
| B | Fenugreek seed mucilage | % | 0 | 4 |
| C | Crospovidone | % | 0 | 5 |
The selected Dependent variables are displayed in Table 2.
Table 2: Dependent variables
| Response | Variables | Unit |
| 1. | Hardness | kg/cm² |
| 2. | Disintegration time | Seconds |
| 3. | In vitro drug release | % |
Table 3: Composition of experimental batches of tablets
| Std | Runs | Independent Variables | Dependent Variables | ||||
| Locust bean gum % | Fenugreek seed mucilage % | Crospovidone % | Hardness (Kg/cm2) | Disintegration time (Second) | In vitro drug release % | ||
| 10 | 1 | 5 | 4 | 0 | 2.76 | 15.7 | 96.21 |
| 4 | 2 | 10 | 4 | 2.5 | 2.57 | 13.2 | 94.2 |
| 15 | 3 | 5 | 2 | 2.5 | 2.43 | 20.5 | 95.64 |
| 12 | 4 | 5 | 4 | 5 | 2.51 | 14.6 | 94.74 |
| 6 | 5 | 10 | 2 | 0 | 2.89 | 16.5 | 96.34 |
| 9 | 6 | 5 | 0 | 0 | 2.67 | 26.3 | 93.78 |
| 8 | 7 | 10 | 2 | 5 | 2.41 | 12.2 | 98.67 |
| 1 | 8 | 0 | 0 | 2.5 | 2.56 | 24.3 | 94.89 |
| 14 | 9 | 5 | 2 | 2.5 | 2.46 | 18.5 | 97.45 |
| 13 | 10 | 5 | 2 | 2.5 | 2.49 | 19.2 | 95.23 |
| 5 | 11 | 0 | 2 | 0 | 2.6 | 31.2 | 93.98 |
| 11 | 12 | 5 | 0 | 5 | 2.56 | 14.5 | 98.56 |
| 2 | 13 | 10 | 0 | 2.5 | 2.72 | 12.3 | 98.78 |
| 3 | 14 | 0 | 4 | 2.5 | 2.59 | 15.6 | 97.45 |
| 7 | 15 | 0 | 2 | 5 | 2.5 | 15.3 | 97.34 |
Formulation of Oral Disintegrating tablets by using Direct Compression Method
Preparation of Powder Blend: The solid dispersion of Hydrochlorothiazide (HCTZ) and PEG-4000 is thoroughly mixed with microcrystalline cellulose (MCC), mannitol, corn starch, aspartame, and banana powder until a uniform mixture is obtained. At this stage, the lubricant is not added to prevent interference with the blending process.
Addition of Polymers and Super disintegrants: The locust bean gum and fenugreek seed mucilage are directly incorporated into the powder mixture before further mixing to ensure even distribution. Crospovidone, a super disintegrant, is then added, and the mixture is blended until a homogeneous dispersion is achieved. Finally, the lubricant (magnesium stearate) is added to ensure proper tablet compression without adhesion to the die and punches.
Tablet Compression: The final blend is compressed into Oro dispersible tablets (ODTs) using a suitable tablet compression machine. This process ensures uniform tablet weight, consistent thickness, and adequate mechanical strength for proper handling. The optimized compression parameters help maintain tablet integrity while allowing rapid disintegration in oral conditions, ensuring effective drug release and improved patient compliance.
Quality Control and Evaluation: Quality control tests are crucial for ensuring that formulated tablets adhere to pharmaceutical standards. The hardness test gauges the mechanical strength, ensuring that tablets can endure handling without breaking. The friability test checks for brittleness and overall durability, assessing resistance to wear and tear. The disintegration test measures how quickly a tablet dissolves in oral conditions, allowing for swift drug release and absorption. Uniformity tests ensure consistent weight and size across production batches, avoiding variations that might affect effectiveness. The assay test confirms the exact drug content in each tablet, ensuring the correct dosage is delivered for effective treatment. Other important tests include dissolution studies, which evaluate the drug's release characteristics in simulated biological environments, and moisture analysis, which examines stability and shelf life. These thorough assessments are essential for maintaining quality, safety, and effectiveness, guaranteeing that each tablet functions as designed. Compliance with regulatory standards ensures that patients receive dependable and effective medication with each dose [22-24].
Figure 7: Formulation of Oral Disintegrating Tablets by using Direct Compression Method
Table 4: Formulation Batches
| Formula-tion Code | S.D. of HCTZ (mg) | Locust Bean Gum (mg) | Fenugreek Seed Mucilage (mg) | Crospovidone (mg) | Banana Powder (mg) | Corn Starch (mg) | Mg Stearate (mg) | Aspartame (mg) | MCC (mg) |
| ODT 1 | 50 | 5 | 4 | 0 | 4 | 2 | 1 | 2 | 32 |
| ODT 2 | 50 | 10 | 4 | 2.5 | 4 | 2 | 1 | 2 | 24.5 |
| ODT 3 | 50 | 5 | 2 | 2.5 | 4 | 2 | 1 | 2 | 22.5 |
| ODT 4 | 50 | 5 | 4 | 5 | 4 | 2 | 1 | 2 | 27 |
| ODT 5 | 50 | 10 | 2 | 0 | 4 | 2 | 1 | 2 | 29 |
| ODT 6 | 50 | 5 | 0 | 0 | 4 | 2 | 1 | 2 | 35 |
| ODT 7 | 50 | 10 | 2 | 5 | 4 | 2 | 1 | 2 | 24 |
| ODT 8 | 50 | 0 | 0 | 2.5 | 4 | 2 | 1 | 2 | 28.5 |
| ODT 9 | 50 | 5 | 2 | 2.5 | 4 | 2 | 1 | 2 | 21.5 |
| ODT 10 | 50 | 5 | 2 | 2.5 | 4 | 2 | 1 | 2 | 32.5 |
| ODT 11 | 50 | 0 | 2 | 0 | 4 | 2 | 1 | 2 | 39 |
| ODT 12 | 50 | 5 | 0 | 5 | 4 | 2 | 1 | 2 | 29 |
| ODT 13 | 50 | 10 | 0 | 2.5 | 4 | 2 | 1 | 2 | 28.5 |
| ODT 14 | 50 | 0 | 4 | 2.5 | 4 | 2 | 1 | 2 | 34.5 |
| ODT 15 | 50 | 0 | 2 | 5 | 4 | 2 | 1 | 2 | 34 |
Precompression Studies
Angle of repose: The maximum angle at which a buildup of loose powder remains stable without collapsing is known as the angle of repose. It acts as a gauge of the cohesiveness and flow properties of the powder. Better flow is indicated by lower angles, and greater angles indicate worse flow. A mound of powder is progressively built up on a level surface until it starts to slump in order to be measured.

Bulk Density: The mass of a powder divided by its whole volume, which includes the empty spaces between particles, is known as its bulk density. It is commonly given in quantities such as g/cm³ or kg/L and indicates how well the powder packs together. Using tools like a graduated cylinder, a known volume of the powder—including its vacant spaces—must be weighed in order to make a determination.

Tapped Density: The maximum bulk density attained when a powder is tapped or vibrated to remove interparticle gaps is known as "tapped density." The powder is tapped repeatedly in a container until no more volume loss takes place, indicating the powder's packing capabilities under compaction.

Hausner's Ratio: Hausner's ratio is the ratio of tapped density to bulk density, calculated using the formula:

It offers information about the packing efficiency and flow behaviour of the powder. Good flowability is indicated by a ratio near 1, and poor flowability is indicated by values greater than 1.
Carr's Index: Carr's index, or compressibility index, quantifies the powder's compressibility and is calculated as follows:
 00%
00%
It shows the extent of volume reduction brought about by compression or tapping. Superior flowability is indicated by lower Carr's index values, whereas reduced flow or enhanced cohesion are implied by higher values.
Partition Coefficient: A partition coefficient study was carried out using 10 mg of hydrochlorothiazide dissolved in 50 ml of n-octanol, which had previously been saturated with water. The solution was mixed thoroughly and shaken vigorously before adding 50 ml of distilled water that had been saturated with n-octanol. This mixture was then subjected to mechanical shaking for 24 hours to enable the drug to distribute between the two immiscible phases. Once equilibrium was achieved, the phases were carefully separated, and the concentrations of hydrochlorothiazide in each phase were measured using UV absorbance readings. The partition coefficient (
Result and Discussion
Organoleptic properties: Organoleptic properties of the drug sample were found to be as given in table below.
Table 5: Organoleptic properties of Hydrochlorothiazide
| S. No. | Organoleptic Characteristics | Result |
| 1. | Color | White to off-white crystalline compound |
| 2. | Nature | Crystalline powder |
| 3. | Odour | Odorless |
| 4. | Taste | Bitter |
Melting point of Hydrochlorothiazide: The final reading was obtained by averaging the results of three independent experiments, resulting in a temperature of 268–270°C.
Partition Coefficients: The partition coefficients of Hydrochlorothiazide found to be -0.07.
pH determination of Hydrochlorothiazide: The pH range of Hydrochlorothiazide is adjusted to 5.5–7.0 during preparation to ensure that it is stable and effective in treating hypertension.
Table 6: Flow properties of powder blend
| Formulation batch | Angle of Repose (θ) | Bulk Density (g/cm3) | Tapped Density (g/cm3) | Hausner’s Ratio | Carr’s Ratio (%) |
| ODT 1 | 29.81 ± 1.29 | 0.457 ± 0.0131 | 0.508 ± 0.004 | 1.2548 ± 0.0217 | 25.79 ± 1.0134 |
| ODT2 | 27.96 ± 1.69 | 0.505 ± 0.0119 | 0.578 ± 0.018 | 1.3148 ± 0.0233 | 21.87 ± 1.1245 |
| ODT3 | 24.96 ± 1.66 | 0.464 ± 0.0116 | 0.490 ± 0.023 | 1.2768 ± 0.0127 | 22.32 ± 1.0043 |
| ODT4 | 28.61 ± 1.17 | 0.498 ± 0.0114 | 0.547 ± 0.024 | 1.2878 ± 0.0323 | 25.27 ± 1.0021 |
| ODT5 | 29.61 ± 1.27 | 0.478 ± 0.0126 | 0.506 ± 0.019 | 1.3218 ±0.02317 | 22.27 ± 1.1425 |
| ODT6 | 25.40 ± 1.02 | 0.453 ± 0.0116 | 0.498 ± 0.026 | 1.2341 ± 0.0212 | 27.83 ± 1.2571 |
| ODT7 | 26.85 ± 1.78 | 0.449 ± 0.0132 | 0.501 ± 0.032 | 1.3412 ± 0.0301 | 26.89 ± 1.1121 |
| ODT 8 | 29.17 ± 1.54 | 0.510 ± 0.0134 | 0.574 ± 0.012 | 1.3032 ± 0.0123 | 27.14 ± 1.2372 |
| ODT 9 | 27.64 ± 0.66 | 0.468 ± 0.0129 | 0.528 ± 0.028 | 1.2431 ± 0.0732 | 26.68 ± 0.8414 |
| ODT 10 | 28.95 ± 0.99 | 0.479 ± 0.0131 | 0.521 ± 0.023 | 1.2842 ± 0.0345 | 27.12 ± 1.1571 |
| ODT 11 | 26.45 ± 0.92 | 0.480 ± 0.0152 | 0.536 ± 0.055 | 1.2989 ± 0.0312 | 27.85 ± 1.1571 |
| ODT 12 | 29.05 ± 0.45 | 0.557 ± 0.0194 | 0.593 ± 0.012 | 1.2965 ±0.02987 | 28.11 ± 1.1522 |
| ODT 13 | 27.22 ± 0.35 | 0.447 ± 0.0192 | 0.483 ± 0.011 | 1.2714± 0.0312 | 27.71 ± 1.2578 |
| ODT 14 | 28.35 ± 0.15 | 0.489 ± 0.0192 | 0.514 ± 0.015 | 1.3192 ± 0.0297 | 27.29 ± 1.2278 |
| ODT 15 | 30.32 ± 0.41 | 0.560 ± 0.0219 | 0.596 ± 0.025 | 1.2954 ± 0.0231 | 26.34 ± 1.1578 |
Post-compression Studies
Organoleptic characters
Table 7: Organoleptic properties of Hydrochlorothiazide Tablets
| S. No. | Organoleptic Characteristics | Result |
| 1. | Color | White to off-white |
| 2. | Shape | Circular |
| 3. | Odour | Odorless |
| 4. | Taste | Appreciably sweet |
Identification of Hydrochlorothiazide by UV spectrophotometry
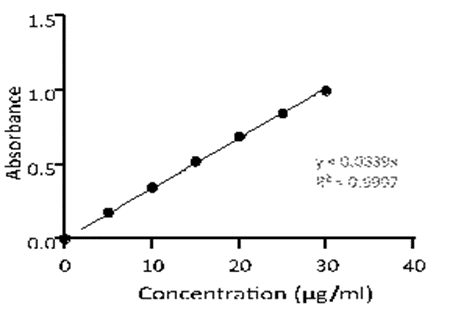
Derivation of drug spectrum: The stock solution of Hydrochlorothide, prepared with NaOH** and a concentration of10 µg/ml displayed a peak absorption (**λmax of **273 nm. At this specific, the solution absorbance was recorded at 0.3455. This absorbance reading was utilized for additional analysis and to determine the drug's concentration in the solution. Understanding the absorption properties is essential for quantitative analysis in various analytical methodologies, allowing for precise evaluation of Hydrochlorothiazide levels in formulations and contributing to the creation of trustworthy pharmaceutical tests.
Preparation of calibration curve of Hydrochlorothiazide in NaOH: The calibration curve for Hydrochlorothiazide dissolved in 0.1M NaOH was plotted using a UV spectrophotometer, with measurements taken at a wavelength of 273 nm. The curve was constructed from six dilutions of the stock solution, each prepared at a concentration of 5 µg/ml. This calibration method is essential for quantitative analysis, providing a relationship between absorbance and concentration of Hydrochlorothiazide. By measuring absorbance at the specified wavelength, this curve can be used to determine the concentration of Hydrochlorothiazide in unknown samples. The results of the calibration curve are summarized in the accompanying table.
Table 8: Calibration curve data of Hydrochlorothiazide in NaOH
| S. No. | Concentration (μg/ml) | Absorbance at 273 nm |
| 1. | 5 | 0.1765 |
| 2. | 10 | 0.3455 |
| 3. | 15 | 0.5223 |
| 4. | 20 | 0.6905 |
| 5. | 25 | 0.8464 |
| 6. | 30 | 0.9967 |
Calibration curve of Hydrochlorothiazide in 0.1M NaOH at λmax 273nm.
Figure 8: Calibration curve of Hydrochlorothiazide
Table 9: Post-compression parameters of tablets
| Formulation Batch | Thickness (mm) | Hardness (Kg/cm2) | Friability (%) | Disintegration time (Sec) | Weight (mg) |
| ODT 1 | 1.85±0.07 | 2.76 | 0.53±0.12 | 15.7 | 99.23±0.12 |
| ODT2 | 1.88±0.04 | 2.57 | 0.52±0.13 | 13.2 | 101.25±0.13 |
| ODT3 | 1.89±0.02 | 2.43 | 0.57±0.12 | 20.5 | 98.67±0.12 |
| ODT4 | 1.86±0.04 | 2.51 | 0.53±0.04 | 14.6 | 97.54±0.04 |
| ODT5 | 1.87±0.04 | 2.89 | 0.52±0.08 | 16.5 | 99.67±0.08 |
| ODT6 | 1.85±0.08 | 2.67 | 0.55±0.50 | 26.3 | 97.34±0.50 |
| ODT7 | 1.87±0.09 | 2.41 | 0.51±0.09 | 12.2 | 100.67±0.09 |
| ODT 8 | 1.89±0.01 | 2.56 | 0.57±0.07 | 24.3 | 98.24±0.07 |
| ODT 9 | 1.86±0.05 | 2.46 | 0.54±0.01 | 18.5 | 99.80±0.01 |
| ODT 10 | 1.87±0.04 | 2.49 | 0.51±0.13 | 19.2 | 102.65±0.13 |
| ODT 11 | 1.85±0.09 | 2.6 | 0.56±0.50 | 31.2 | 99.67±0.50 |
| ODT 12 | 1.86±0.06 | 2.56 | 0.57±0.50 | 14.5 | 101.24±0.07 |
| ODT 13 | 1.89±0.02 | 2.72 | 0.52±0.11 | 12.3 | 99.80±0.01 |
| ODT 14 | 1.88±0.04 | 2.59 | 0.59±0.08 | 15.6 | 104.65±0.13 |
| ODT 15 | 1.87±0.04 | 2.5 | 0.51±0.04 | 15.3 | 97.67±0.50 |
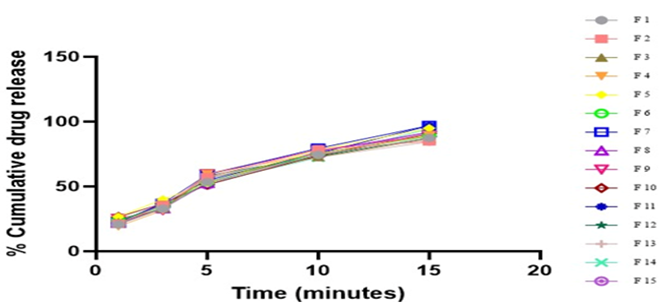
In-vitro dissolution studies
Table 10: Cumulative drug release of Hydrochlorothiazide Tablets formulations (F1-F15)
| Cumulative Drug Release (%) | |||||
| Formulation code | Time (Minutes) | ||||
| 1 | 3 | 5 | 10 | 15 | |
| ODT 1 | 20.67±0.23 | 32.98±0.53 | 53.47±0.62 | 74.39±0.57 | 87.41±1.32 |
| ODT2 | 21.28±0.40 | 35.76±0.62 | 57.22±0.32 | 78.54±1.12 | 84.87±0.47 |
| ODT3 | 22.43±0.43 | 33.12±0.67 | 56.63±1.00 | 72.65±0.62 | 86.85±0.63 |
| ODT4 | 19.34±0.54 | 31.72±0.62 | 59.65±0.09 | 75.32±0.43 | 89.41±1.32 |
| ODT5 | 26.72±1.23 | 39.98±0.76 | 52.50±0.68 | 78.39±0.57 | 94.92±0.44 |
| ODT6 | 23.63±1.12 | 34.80±0.74 | 53.47±0.62 | 75.65±0.71 | 88.78±0.81 |
| ODT7 | 22.43±0.43 | 36.98±0.53 | 59.12±0.81 | 79.43±0.95 | 96.90±1.00 |
| ODT 8 | 21.67±0.23 | 33.12±0.67 | 52.54±0.65 | 76.78±0.78 | 91.78±1.15 |
| ODT 9 | 25.19±0.34 | 31.78±1.45 | 59.63±1.00 | 78.20±1.32 | 89.20±0.78 |
| ODT 10 | 26.46±0.87 | 36.87±1.15 | 51.40±0.37 | 73.65±0.62 | 90.87±0.47 |
| ODT 11 | 23.23±0.76 | 35.98±0.43 | 54.80±1.32 | 75.54±1.12 | 96.85±0.63 |
| ODT 12 | 21.28±0.73 | 33.32±0.54 | 58.22±0.32 | 73.29±0.65 | 87.65±1.34 |
| ODT 13 | 25.13±1.00 | 32.76±0.62 | 53.98±0.91 | 72.50±0.47 | 84.43±1.27 |
| ODT 14 | 23.28±0.40 | 34.32±0.67 | 52.65±1.00 | 72.22±0.37 | 94.67±0.78 |
| ODT 15 | 21.86±0.51 | 37.19±1.32 | 51.32±0.78 | 74.63±0.23 | 92.20±0.28 |
Figure 9: Cumulative drug release of Hydrochlorothiazide Tablets (ODT 1-ODT 15)
Response data for all 15 batches by using Box-Behnken Experimental Design (FI–F15)
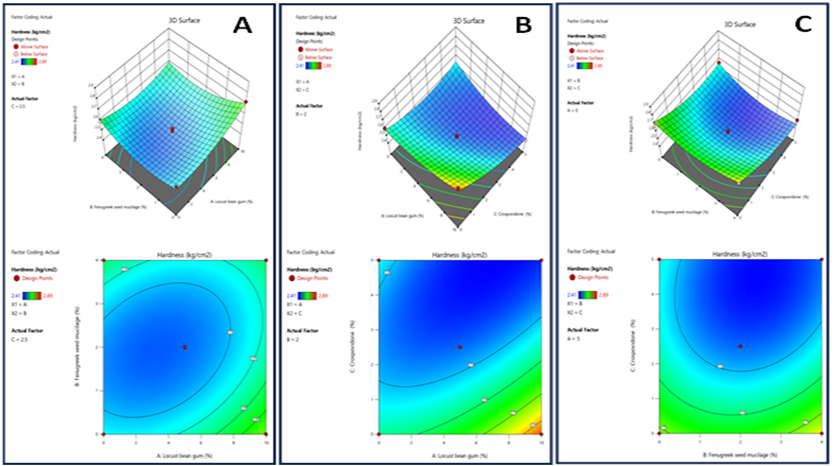
Response 1: hardness
Figure 10: The 2D contour plot and 3D surface graph show that how Hardness is affected by factors X1 (Hardness), X2, (Disintegrating time), X3 (In vitro release)
ANOVA for Quadratic model Response 1: Hardness
Table 11: R1(Hardness)
| Source | Sum of Squares | df | Mean Square | F-value | p-value | |
| Model | 0.2313 | 9 | 0.0257 | 11.17 | 0.0081 | significant |
| A-Locust bean gum | 0.0145 | 1 | 0.0145 | 6.28 | 0.0541 | |
| B-Fenugreek seed mucilage | 0.0008 | 1 | 0.0008 | 0.3478 | 0.5810 | |
| C-Crospovidone | 0.1104 | 1 | 0.1104 | 48.02 | 0.0010 | |
| AB | 0.0081 | 1 | 0.0081 | 3.52 | 0.1194 | |
| AC | 0.0361 | 1 | 0.0361 | 15.70 | 0.0107 | |
| BC | 0.0049 | 1 | 0.0049 | 2.13 | 0.2042 | |
| A² | 0.0144 | 1 | 0.0144 | 6.27 | 0.0542 | |
| B² | 0.0283 | 1 | 0.0283 | 12.29 | 0.0172 | |
| C² | 0.0222 | 1 | 0.0222 | 9.64 | 0.0267 | |
| Residual | 0.0115 | 5 | 0.0023 | |||
| Lack of Fit | 0.0097 | 3 | 0.0032 | 3.59 | 0.2253 | not significant |
| Pure Error | 0.0018 | 2 | 0.0009 | |||
| Cor Total | 0.2428 | 14 |
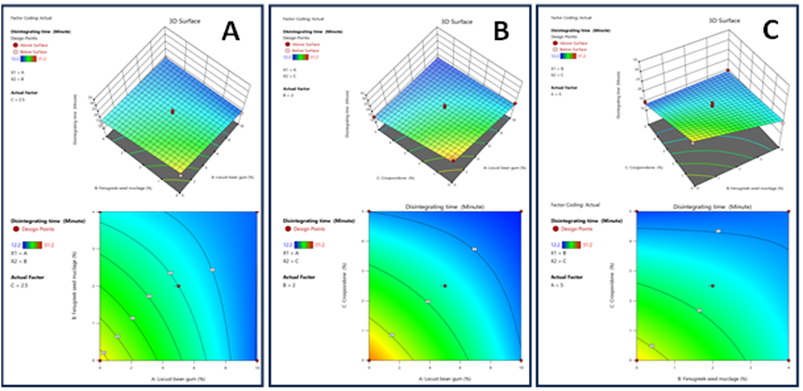
Response 2: Disintegrating time
Figure 11: The 2D contour plot and 3D surface graph show that how Disintegrating time is affected by factors X1, X2, X3
ANOVA for 2FI model Response 2: Disintegrating time
Table 12: R2 (disintegrating time)
| Source | Sum of Squares | df | Mean Square | F-value | p-value | |
| Model | 393.72 | 6 | 65.62 | 17.15 | 0.0004 | significant |
| A-Locust bean gum | 129.60 | 1 | 129.60 | 33.87 | 0.0004 | |
| B-Fenugreek seed mucilage | 41.86 | 1 | 41.86 | 10.94 | 0.0107 | |
| C-Crospovidone | 136.95 | 1 | 136.95 | 35.79 | 0.0003 | |
| AB | 23.04 | 1 | 23.04 | 6.02 | 0.0397 | |
| AC | 33.64 | 1 | 33.64 | 8.79 | 0.0180 | |
| BC | 28.62 | 1 | 28.62 | 7.48 | 0.0256 | |
| Residual | 30.61 | 8 | 3.83 | |||
| Lack of Fit | 28.55 | 6 | 4.76 | 4.62 | 0.1886 | not significant |
| Pure Error | 2.06 | 2 | 1.03 | |||
| Cor Total |
Response 3: In vitro drug release
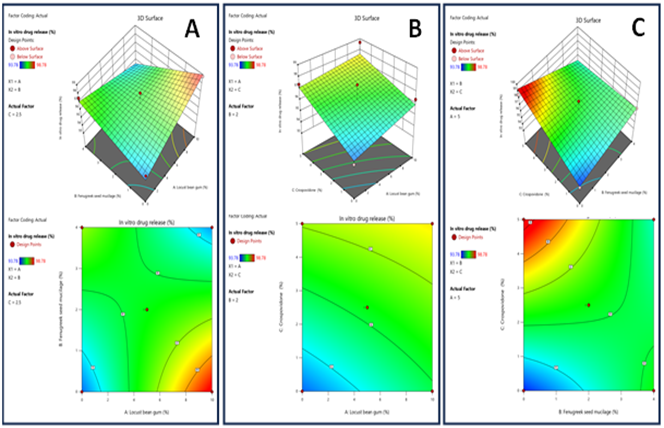
Figure 12: The 2D contour plot and 3D surface graph shows that how In vitro drug release is affected by factors X1, X2, X3
ANOVA for 2FI model Response 3: In vitro drug release
Table 13: R3 (In vitro drug release)
| Source | Sum of Squares | df | Mean Square | F-value | p-value | |
| Model | 36.70 | 6 | 6.12 | 8.22 | 0.0045 | significant |
| A-Locust bean gum | 2.34 | 1 | 2.34 | 3.15 | 0.1139 | |
| B-Fenugreek seed mucilage | 1.45 | 1 | 1.45 | 1.95 | 0.1999 | |
| C-Crospovidone | 10.13 | 1 | 10.13 | 13.60 | 0.0061 | |
| AB | 12.74 | 1 | 12.74 | 17.12 | 0.0033 | |
| AC | 0.2652 | 1 | 0.2652 | 0.3563 | 0.5671 | |
| BC | 9.77 | 1 | 9.77 | 13.12 | 0.0068 | |
| Residual | 5.96 | 8 | 0.7445 | |||
| Lack of Fit | 3.16 | 6 | 0.5275 | 0.3780 | 0.8499 | not significant |
| Pure Error | 2.79 | 2 | 1.40 | |||
| Cor Total | 42.65 | 14 |
Table 14: formulation of optimized batch from BBD (ODT 16)
| S. No. | Ingredients | Quantities (mg) |
| 1 | Solid dispersion of HCTZ and PEG-4000 (mg) | 50 |
| 2 | Locust bean gum (mg) | 7.9 |
| 3 | Fenugreek seed mucilage (mg) | 1.6 |
| 4 | Crospovidone (mg) | 2.85 |
| 5 | Banana powder (mg) | 4 |
| 6 | Corn Starch (mg) | 2 |
| 7 | Mg Stearates (mg) | 1 |
| 8 | Aspartame (mg) | 2 |
| 9 | MCC (mg) | 28.35 |
Table 15: pre compressional data of optimized batch (ODT 16)
| S. No. | Pre-compressional evaluation parameter | Results |
| 1 | Bulk density | 0.501 ± 0.0131 |
| 2 | Tapped density | 0.523 ± 0.0119 |
| 3 | Hausner’s ratio | 1.274 ± 0.0217 |
| 4 | Carss index | 23.79 ± 1.0134 |
| 5 | Angle of repose | 26.96 ± 1.069 |
Table 16: post compressional data of optimized batch (ODT 16)
| S. No. | Post-compression evaluation parameter | Results |
| 1 | Weight (mg) | 102 |
| 2 | Thickness (mm) | 1.86 |
| 3 | Hardness (kg/cm2) | 2.76 |
| 4 | Disintegration time (second) | 16 |
| 5 | Friability (%) | 0.52 |
Table 17: In vitro drug release for optimized batch (ODT 16)
| Cumulative Drug Release (%) | |||||
| Formulation Batch | Time (Minutes) | ||||
| 1 | 3 | 5 | 10 | 15 | |
| ODT 16 | 21.67±0.23 | 34.98±0.53 | 53.47±0.62 | 74.39±0.57 | 97.41±1.32 |
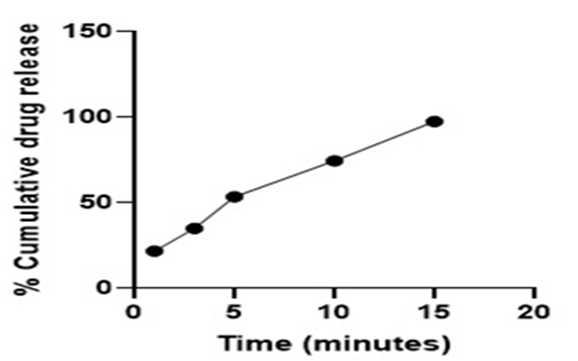
Figure 10: Percent drug release of batch ODT 16 (optimised batch)
The obtained Percent drug release of batch ODT 16 (optimised batch) has 95.981 % of drug release in 15 minutes, and the optimized batch was evaluated for all the pre and post compressional parameters required for quality control of dosage form and the obtained results was found between satisfied range.
Stability Studies: The stability studies of fast-dissolving tablets of Hydrochlorothiazide at a 25mg dose indicated that the best formulation remained stable even after storing at 40±20°C / 75±5 percentage RH for 3 months. The tablets were visually examined for any physical changes, evaluated for drug content, and in vitro drug release at monthly intervals. The results showed that the formulation maintained its drug release profile within the specified limits, demonstrating stability over the study period.
Conclusion
In conclusion, this study successfully employed a Box-Behnken design and direct compression to develop fast dissolving Hydrochlorothiazide tablets. Natural (Locust bean gum and Fenugreek seed mucilage) and synthetic (Crospovidone) superdisintegrants were evaluated for optimal disintegration and drug release. Pre-formulation studies ensured compatibility. This approach presents a promising patient-centric formulation with improved compliance, especially for Hypertension with swallowing difficulties. To confirm clinical efficacy, further research is needed to assess large-scale production and conduct in-vivo studies. Acknowledgement: I would like to show my sincere gratitude towards IPS Academy College of Pharmacy for providing the necessary requirements and facilities throughout the study. Lastly, I would like to express my appreciation to all my colleagues and peers for their helpful discussions and moral support, which contributed to the success of this work.
Conflict of interest
The authors declare no conflict of interest.
References
- Forouzanfar MH, Liu P, Roth GA, Ng M, Biryukov S, Marczak L, Alexander L, Estep K, Abate KH, Akinyemiju TF, Ali R. (2017). Global burden of hypertension and systolic blood pressure of at least 110 to 115 mm Hg, 1990-2015. Jama, 317(2):165-182.
Publisher | Google Scholor - Carey RM, Calhoun DA, Bakris GL, Brook RD, Daugherty SL, Dennison-Himmelfarb CR, Egan BM, Flack JM, Gidding SS, Judd E, Lackland DT. (2018). Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension, 72(5): e53-90.
Publisher | Google Scholor - Mills KT, Xu Y, Zhang W, Bundy JD, Chen CS, Kelly TN, Chen J, He J. (2015). A systematic analysis of worldwide population-based data on the global burden of chronic kidney disease in 2010. Kidney international, 88(5):950-957.
Publisher | Google Scholor - Carey RM, Sakhuja S, Calhoun DA, Whelton PK, Muntner P. (2019). Prevalence of apparent treatment-resistant hypertension in the United States: comparison of the 2008 and 2018 American Heart Association scientific statements on resistant hypertension. Hypertension, 73(2):424-431.
Publisher | Google Scholor - Lamirault G, Artifoni M, Daniel M, Barber-Chamoux N. (2020). Resistant hypertension: novel insights. Current hypertension reviews, 16(1):61-72.
Publisher | Google Scholor - Kario K, Hoshide S, Narita K, Okawara Y, Kanegae H. (2021). Cardiovascular prognosis in drug-resistant hypertension stratified by 24-hour ambulatory blood pressure: the JAMP study. Hypertension, 78(6):1781-1790.
Publisher | Google Scholor - Morales-Olivas FJ. (2024). Diuretics use in the management of hypertension. Hipertensión y Riesgo Vascular.
Publisher | Google Scholor - Kumar A, Khatri G, Swed S, Hasan MM. (2022). Hydrochlorothiazide: A savior to the heart or a foe to the skin? Annals of Medicine and Surgery, 79.
Publisher | Google Scholor - An C, Yang L, Han T, Song H, Li Z, Zhang J, Zhang K. (2022). Kidney ion handling genes and their interaction in blood pressure control. Bioscience Reports, 42(11): BSR20220977.
Publisher | Google Scholor - Russo A, Di Gaetano C, Cugliari G, Matullo G. (2018). Advances in the genetics of hypertension: the effect of rare variants. International journal of molecular sciences, 19(3):688.
Publisher | Google Scholor - Nandy BC, Mazumder B, Pathak K, Saxena N, Jain S, Sharma S, Amishaben R, Shrivastava A, Saxena P. (2011). An overview on fast dissolving drug delivery system. Asian Journal of pharmaceutical sciences and research, 1(2):1-30.
Publisher | Google Scholor - Sharma AN, Upadhyay PK, Bajpai M, Easwari TS, Bhadauria P, Chaudhary R, Garg A, Jha MK, Dewangan HK. (2023). Formulation and evaluation of fast dissolving tablets of Nimesulide and Paracetamol. Materials Today: Proceedings.
Publisher | Google Scholor - Ciurba A, Rédai E, Pop I, Antonoaea P, Todoran N. Kinetics and Mechanism of Drug Release from Loratadine Orodispersible Tablets Developed without Lactose. Acta Marisiensis-Seria Medica, 63(1):23-26.
Publisher | Google Scholor - Brniak W, Jachowicz R, Krupa A, Skorka T, Niwinski K. (2013). Evaluation of co-processed excipients used for direct compression of orally disintegrating tablets (ODT) using novel disintegration apparatus. Pharmaceutical development and technology, 18(2):464-474.
Publisher | Google Scholor - Roy H. Box-Behnken design for optimization of formulation variables for fast dissolving tablet of Urapidil. Asian Journal of Pharmaceutics (AJP), 12(03).
Publisher | Google Scholor - Roy H, Nandi S, Pavani U, Lakshmi U, Reddy TS, Gayatri DV. (2020). Optimization and quality by design approach for piroxicam fast dissolving tablet formulations using box-behnken design. Current Drug Therapy, 15(2):152-165.
Publisher | Google Scholor - Dionísio M, Grenha A. (2012). Locust bean gum: exploring its potential for biopharmaceutical applications. Journal of Pharmacy and Bioallied Sciences, 4(3):175-185.
Publisher | Google Scholor - Barak S, Mudgil D. Locust bean gum: Processing, properties and food applications—A review. International journal of biological macromolecules, 66:74-80.
Publisher | Google Scholor - Prajapati VD, Jani GK, Moradiya NG, Randeria NP, Nagar BJ. Locust bean gum: A versatile biopolymer. Carbohydrate polymers, 94(2):814-821.
Publisher | Google Scholor - Nayak AK, Pal D, Das S. (2013). Calcium pectinate-fenugreek seed mucilage mucoadhesive beads for controlled delivery of metformin HCl. Carbohydrate polymers, 96(1):349-357.
Publisher | Google Scholor - Babalola OC, Odeku OA. (2014). Disintegrant properties of banana starch obtained from the unripe fruits of Musa sapientum L. Journal of applied pharmaceutical science, 4(9):083-088.
Publisher | Google Scholor - Sallam AA, Omari DM. Preformulation considerations in pharmaceutical formulation process. In Dosage Forms, Formulation Developments and Regulations. Academic Press, 395-441.
Publisher | Google Scholor - Noman MA, Alburyhi MM, Alqubati MA. (2024). Preformulation and Characterization Studies of Clopidogrel Active Ingredient for Orodispersible Tablets Development. World Journal of Pharmacy and Pharmaceutical Sciences, 13(3):996-1015.
Publisher | Google Scholor - Singh K, Kumar A, Singh S. A Research on Formulation Design, Development and Evaluation of Post-Compressional Parameters of Fast Disintegrating Tablets of Losartan Potassium by Using Locast Bean Gum as Super Disintegrants.
Publisher | Google Scholor - Sallam AA, Omari DM. Preformulation considerations in pharmaceutical formulation process. In Dosage Forms, Formulation Developments and Regulations. Academic Press, 395-441.
Publisher | Google Scholor - Noman MA, Alburyhi MM, Alqubati MA. (2024). Preformulation and Characterization Studies of Clopidogrel Active Ingredient for Orodispersible Tablets Development. World Journal of Pharmacy and Pharmaceutical Sciences, 13(3):996-1015.
Publisher | Google Scholor - Singh K, Dadabhau GD, Singh S. (2023). Formulation Design and Development of Fast Disintegrating Tablets of Losartan Potassium by Using Locast Bean Gum as Super disintigrants. International Journal, 10(5):1084-1096.
Publisher | Google Scholor