Case Report
Urinary Tract Infection and Tick Infestation in a Dog: Zoonotic and One Health Insights in Niger State, Nigeria
- Fatima Kudu Muhammed
- Fatima Muhammad Sonfada
- Attahiru Baba
- Joel Jagaba
- Abubakar Raji
- Sani Bello Nma
- Mohammed Katun Yakubu
- Haruna Evuti Aliyu *
Bosso Veterinary Hospital Complex, Minna, Ministry of Livestock and Fisheries, Niger State, Nigeria.
*Corresponding Author: Haruna Evuti Aliyu, Bosso Veterinary Hospital Complex, Minna, Ministry of Livestock and Fisheries, Niger State, Nigeria.
Citation: Fatima K. Muhammed, Fatima M. Sonfada, Baba A., Jagaba J, Haruna E. Aliyu, et al. (2025). Urinary Tract Infection and Tick Infestation in a Dog: Zoonotic and One Health Insights in Niger State, Nigeria, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 8(5):1-7. DOI: 10.59657/2837-4681.brs.25.203
Copyright: © 2025 Haruna Evuti Aliyu, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: September 05, 2025 | Accepted: September 19, 2025 | Published: September 26, 2025
Abstract
Urinary tract infections (UTIs) and prostatitis are common clinical conditions in dogs, often associated with bacterial pathogens of zoonotic significance. This case report describes the clinical management of a 5-year-old male Lhasa Apso dog presenting with urinary tract infection, suspected prostatitis, and ectoparasitism. Laboratory analysis revealed Staphylococcus aureus infection, with culture sensitivity indicating susceptibility to gentamicin, streptomycin, and ciprofloxacin, but resistance to several commonly used antimicrobials. The dog was also infested with Rhipicephalus sanguineus ticks, a known vector of zoonotic pathogens. Treatment involved a combination of antimicrobial therapy, anti-inflammatory medication, ectoparasite control, and deworming. This case underscores the significance of companion animals as potential reservoirs of multidrug-resistant bacteria and ectoparasites with zoonotic potential. A One Health perspective is essential to mitigate public health risks posed by antimicrobial resistance (AMR) and tick-borne pathogens at the human–animal–environment interface.
Keywords: dog; urinary tract infection; staphylococcus aureus; antimicrobial resistance; ectoparasitism; one health; zoonosis
Introduction: Signalment
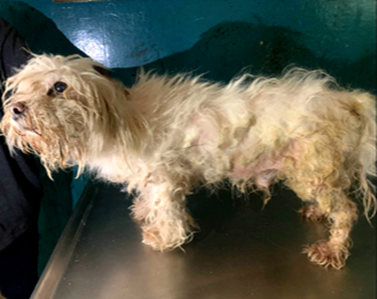
A 5-year-old intact male Lhasa Apso weighing 10 kg (Figure 1) was presented on 11th August 2025 with frequent urination, urinary incontinence, straining during urination, and difficulty in defecation requiring owner assistance. The dog, which was fed on homemade diets with occasional commercial feeds and had never been mated, was examined on 4th September 2025 and had a respiratory rate of 32 c/m, pulse 80 b/m, and temperature 38.8°C. as shown in (Table 1). Clinical examination revealed a soiled perineum, alopecia, matted ventral fur, protruding penile head, engorged bulbus glandis, and the presence of ticks. Based on history and findings, differential diagnoses included urinary tract infection (Bartges, 2004), prostatitis (Smith, 2008), prostatic carcinoma (Cornell, 2000), urolithiasis (Ling et al., 1998), ectoparasitism (Wall & Shearer, 2001), and helminthosis (Bowman, 2014). The case illustrates the diagnostic complexity of differentiating urinary and prostatic disorders in intact male small breeds where parasitic infestations and poor perineal hygiene further complicate the clinical picture. Prostatitis, often secondary to ascending bacterial urinary tract infections (Lobetti, 2014), remained a strong consideration, while ticks increased the risk of vector-borne infections (Irwin, 2002) and helminths likely contributed to gastrointestinal straining. A holistic diagnostic and management approach involving urinalysis, urine culture, ultrasonography, parasite screening, antimicrobial therapy, acaricidal treatment, deworming, grooming, and improved nutrition is recommended to address the multifactorial nature of this presentation.
Figure 1: A 5-year-old intact male Lhasa Apso
Background
Companion animals have become integral to human society, predominantly serving as sources of companionship, security, and emotional support. However, their role presents significant public health implications, particularly in relation to zoonotic diseases. These pets can occasionally act as reservoirs for various pathogens, including multidrug-resistant (MDR) bacteria and vector-borne parasites, which can be transmitted to humans through both direct contact and indirect avenues.
Research indicates that the interaction between humans and their companion animals is a key factor in this transmission dynamic. The frequency of close contact and the nature of such interactions can substantially enhance the risk of transmitting zoonotic pathogens from animals, such as bacteria or viruses, to humans (Joosten et al., 2020; (Johnson et al., 2022). A notable concern arises from findings that various multidrug-resistant bacteria, such as the extended-spectrum beta-lactamase (ESBL)-producing Escherichia coli, have been detected in companion dogs, providing a potential link for transmission (Johnson et al., 2022). This underscores a vital aspect of pet ownership, where the health of animals can directly impact public health through pathogen transmission.
Moreover, vector-borne diseases, such as those transmitted by ticks and mosquitoes, pose significant threats. These vectors can harbor numerous pathogens, and their interactions within shared environments with companion animals complicate the epidemiology of these diseases. Animals infested with these vectors might become infected and provide a transmission route to humans, thus contributing to the spread of diseases such as Lyme disease or other infections (Noden et al., 2021; Haynes et al., 2024).
Urinary tract infections (UTIs) in dogs are commonly attributed to bacterial pathogens, particularly Escherichia coli, Staphylococcus species, and Proteus species. Among these, E. coli remains the leading causative agent, responsible for a significant proportion of canine UTIs, as evidenced by studies indicating its prevalence in both dogs and cats with UTIs (Ataya et al., 2023). Notably, the emergence of Staphylococcus aureus as a causative agent poses clinical implications. Historically considered a minor player in canine UTIs, S. aureus has been increasingly recognized in veterinary medicine, with studies reporting its presence in about 0.5% to 6% of UTI cases in dogs (Alshomrani et al., 2023; Paudel et al., 2023). This shift in prevalence raises concerns not only for veterinary health but also for zoonotic transmission and potential public health risks (Yudhanto et al., 2022).
The relationship between urinary tract conditions and antimicrobial resistance highlights the urgent need for vigilant monitoring. Reports indicate a troubling trend of multidrug-resistant (MDR) strains among bacteria isolated from canine urinary samples, elevating the risk of treatment failures and complicating therapeutic management (Yudhanto et al., 2022) Akmal et al., 2023). This resistance is exacerbated by the zoonotic transmission pathways, which can affect vulnerable populations, suggesting significant implications for public health (Yudhanto et al., 2022).
Beyond urinary infections, tick infestations, particularly those caused by Rhipicephalus sanguineus, have significant public and veterinary health consequences. These ticks are known vectors for pathogens such as Ehrlichia canis and Babesia spp., which can infect both dogs and humans, causing serious health issues (Charles et al., 2024).
This case report documents the clinical presentation, diagnosis, treatment, and public health implications of a Lhasa Apso dog with urinary tract infection, suspected prostatitis, and ectoparasitism. The findings are discussed within a One Health framework, highlighting zoonotic potential and antimicrobial resistance concerns.
Figure 2: Rhipicephalus sanguineus.
Plan of action
Laboratory investigation (urine culture and sensitivity, blood smear, fecal examination, tick identification), symptomatic treatment, hair trimming, and follow-up monitoring.
Table 1: Vital Parameters of Dog
| VITALS | VALUE | REFERENCE RANGE |
| Respiratory Rate (c/m) | 32 | 15 – 30 |
| Pulse Rate (b/m) | 80 | 65 – 90 |
| Temperature (Coc) | 38.8 | 37.5 - 39.4 |
Table 2
| Parameters | Obtained values | Absolute values | Reference Range | |
| PCV | 45% | 37 - 55% | ||
| Hb | 15.0g/dl | 12 - 18g/dl | ||
| WBC | 5.6 X 109/l | (6 - 17.0) x 109/l | ||
| MCV | 82fl | 66 - 77fl | ||
| MCHC | 28/dl | 32.0 - 36.3/dl | ||
| Platelets | 205 | 211 – 621 | ||
| Differential Count | ||||
| Neutrophils | 68% | 3.8 x 109/l | (3.6-13.1) x 109/l | |
| Lymphocytes | 25% | 1.4 x 109/l | (0.7-5.1) x 109/l | |
| Monocytes | 04% | 0.22 x 109/l | (0.18-1.7) x 109/l | |
| Eosinophils | 03% | 0.1 x 109/l | (0.18-1.7) x 109/l | |
| Basophils | 00% | - | - | |
Diagnostic Findings
Urine microscopy/culture/sensitivity (M/C/S):
- Amber, slightly turbid sample
- Moderate growth of Staphylococcus aureus after 24 hrs at 37°C
- Antibiotic susceptibility: Sensitive to gentamicin (+++), streptomycin (+++), ciprofloxacin (++), augmentin (++), ofloxacin (+), pefloxacin (+)
- Resistant to: nalidixic acid, ampicillin, ceporex, septrin
- Microscopy revealed epithelial cells (+++), pus cells (2–6/hpf), and bacterial cells (+) Tick identification: Rhipicephalus sanguineus Fecal examination: No helminth eggs or oocysts
Blood smear: No hemoparasites detected
Treatment and Outcome
The dog received combination antimicrobial therapy, anti-inflammatory medication, ectoparasite control, and deworming:
- Day 1–5: Inj. Gentamicin (40 mg) + Amoxicillin (150 mg) IM
- Day 1–3: Inj. Dexamethasone 1 mg/kg IM
- Day 2: Fipronil spray (topical, stat)
- Day 3: Pet wormer (Praziquantel 50 mg + Levamisole 50 mg) PO stat
- Day 4: Inj. Ivermectin 5 mg/kg SC stat
The patient showed clinical improvement by Day 5, with reduced straining and improved urination. The client was advised on hygiene, provision of clean water, and early reporting of illness.
Discussion
This case highlights several important aspects of veterinary and public health significance:
Zoonotic potential of Staphylococcus aureus
The zoonotic potential of Staphylococcus aureus, particularly methicillin-resistant Staphylococcus aureus (MRSA), highlights significant public health concerns due to its capacity to cause severe infections in both humans and animals. The emergence of MRSA as a pathogen has transitioned from hospital settings to community and agricultural environments, with animals such as dogs acting as potential reservoirs for these resistant strains. Given the background of how this bacterium behaves in various environments, it is crucial to explore the patterns of transmission, the role of different animal species, and the implications of antimicrobial resistance.
First, studies indicate that pets, including dogs, can carry MRSA and potentially transmit it to humans, especially those with compromised immune systems, such as the elderly and young children (Krueger et al., 2025, (Dalton et al., 2021. Close contact with MRSA-positive animals can lead to transmission through direct contact or environmental contamination Avberšek et al., 2020). For instance, research shows that MRSA strains in dogs can serve as a source of infection for their owners, thereby emphasizing the importance of monitoring such veterinary cases, particularly in healthcare settings where therapy dogs are involved (Dalton et al., 2021, Avberšek et al., 2020).
In addition to dogs, livestock such as pigs, cattle, and chickens have been identified as significant reservoirs of livestock-associated MRSA (LA-MRSA) strains, particularly clonal complex CC398. The emergence of this strain has been attributed to the intensive use of antibiotics in agricultural practices, which can facilitate the spread of resistance genes (Fetsch et al., 2021, Dube et al., 2021). Notably, persistent colonization in farm personnel who frequently interact. Staphylococcus aureus is a known pathogen in humans, causing skin infections, urinary tract infections, and systemic illnesses. Dogs can serve as reservoirs of methicillin-resistant S. aureus (MRSA), which can be transmitted to humans, particularly immunocompromised individuals and children. The antimicrobial resistance profile observed here, with resistance to several first-line antimicrobials, raises concerns about the spread of MDR bacteria at the animal–human interface.
Tick infestation and zoonoses
Rhipicephalus sanguineus (Figure 2) is an important vector of tick-borne pathogens. Although no blood parasites were detected in this case, the tick’s presence underscores the potential risk of transmission of zoonotic pathogens to humans and other animals in the household. Tick infestations, particularly those involving Rhipicephalus sanguineus, known as the brown dog tick, present significant public health concerns due to their status as vectors of various zoonotic pathogens. The presence of R. sanguineus in domestic environments highlights the potential risk for transmission of pathogens such as Ehrlichia canis, Anaplasma spp., and Rickettsia species to humans and animals within the household context (Çelik et al., 2021; Shih, 2025). Although specific studies may not reveal the immediate presence of blood parasites, the overall ecological profile of R. sanguineus as a vector remains a cause for concern, particularly after recent studies in diverse geographical areas.
Emerging evidence underscores that R. sanguineus has adapted well to human-associated environments and largely relies on canine hosts, resulting in increased tick densities in homes where dogs live. This anthropogenic association raises the likelihood of pathogen transmission to humans, notably in rural or suburban settings where interaction between domestic animals and humans is frequent (Ackermann et al., 2024; Pascoe et al., 2022). Current research suggests that climate change may further exacerbate this risk, as rising temperatures can lead to increased tick activity and host interactions. Studies indicate that R. sanguineus may attach more frequently to humans as environmental temperatures rise, thus enhancing the risk of transmission of zoonotic pathogens, including the Severe Fever with Thrombocytopenia Syndrome virus (SFTSV) (Villarroel et al., 2024).
Additional studies have documented the ability of R. sanguineus to harbor a variety of pathogens, including those responsible for notable human and animal diseases. Rhipicephalus sanguineus, commonly known as the brown dog tick, is a significant vector for various zoonotic pathogens, and its infestation poses serious public health risks. Recent studies have highlighted the prevalence of R. sanguineus in urban and peri-urban areas where it infests domestic animals, particularly dogs, acting as reservoirs for the transmission of pathogens to humans and other animals.
The role of R. sanguineus in the transmission of tick-borne pathogens has been increasingly documented. For instance, a recent study reported the detection of bacterial pathogens, including Rickettsia massiliae, in ticks infesting stray dogs in Taiwan, indicating a potential risk for zoonotic transmission to humans (Chao et al., 2022). This reflects broader concerns since various pathogens transmitted by R. sanguineus can lead to diseases such as ehrlichiosis and rickettsiosis, which are particularly dangerous for immunocompromised individuals or those with underlying health conditions (Ali et al., 2023).
Furthermore, the epidemiological significance of R. sanguineus is underscored by its association with stray dog populations, often found in close proximity to human dwellings, thereby facilitating zoonotic pathogen spillover into human populations (Ali et al., 2023). Infestations in these dogs not only pose risks to their health but also elevate the likelihood of disease transmission within household and community settings.
The interconnectedness of wildlife, domestic animals, and human health a concept encapsulated in the One Health approach further emphasizes the importance of monitoring and controlling tick populations like R. sanguineus. In regions where these ticks proliferate, such as those with high stray.
One Health implications
This case exemplifies the interconnectedness of animal, human, and environmental health. Inadequate tick control, poor hygiene, and inappropriate antimicrobial use in companion animals can contribute to zoonotic disease transmission and antimicrobial resistance. Integrating veterinary surveillance with public health systems is vital to reducing these risks. The One Health framework emphasizes the interconnectedness among animal, human, and environmental health, underscoring that the health of one component affects the others. Recent attention has focused on the implications of inadequate tick control, poor hygiene practices, and inappropriate antimicrobial use in companion animals, as these factors significantly contribute to zoonotic disease transmission and antimicrobial resistance (AMR).
In the context of tick-borne diseases (TBDs), research has shown the crucial role that wildlife populations, especially white-tailed deer, play in influencing human disease risks. A study utilizing a One Health perspective integrated human health data with ecological and wildlife statistics to identify risk factors associated with TBD transmission, highlighting the necessity of interdisciplinary collaboration in disease surveillance (Maxwell et al., 2023. Furthermore, comprehensive tick control measures are vital in urban and peri-urban areas, where frequent human-animal interactions can significantly mitigate the risk of disease transmission (Maxwell et al., 2023, Maxwell et al., 2021).
The impact of antimicrobial use in companion animals is profound, as these animals share close environments with humans and can act as reservoirs for resistant pathogens that contribute to AMR spread. Evidence indicates that patterns of antimicrobial prescriptions in companion animals closely mirror those in human healthcare systems. This is especially concerning, as there is a trend of inappropriate prescriptions for conditions that may not require such treatments, including cases of respiratory infections where both species are frequently prescribed antimicrobials despite many instances being viral.
Conclusion
The clinical management of this case demonstrates that urinary tract infections in dogs can be complicated by zoonotic pathogens such as Staphylococcus aureus and concurrent ectoparasite infestations. The emergence of antimicrobial resistance in such pathogens underscores the need for prudent antibiotic use in veterinary medicine. A One Health approach is essential to prevent the spillover of zoonotic pathogens and multidrug-resistant bacteria from animals to humans.
Acknowledgments
We acknowledge the clinical team at Niger State Veterinary Hospital, Minna, and the Ministry of Livestock and Fisheries for their support in the management of this case.
References
- Ackermann, R., Gall, C., Brayton, K., Collins, N., Wyk, I., Wentzel, J., … Oosthuizen, M. (2024). The bacterial microbiome and pathogen reservoir potential of Rhipicephalus sanguineus ticks in South Africa.
Publisher | Google Scholor - Akmal, Z., Shahbaz, S., Rehman, W., Aman, N., & Ahsan, F. (2023). Antimicrobial susceptibility pattern of uropathogens in urinary tract infections (UTI) patients of Punjab, Pakistan. International Journal of Health Sciences, 6811–6830.
Publisher | Google Scholor - Algammal, A., Hetta, H., Elkelish, A., Alkhalifah, D., Hozzein, W., Batiha, G., & Mabrok, M. (2020). Methicillin-resistant Staphylococcus aureus (MRSA): One health perspective approach to the bacterium epidemiology, virulence factors, antibiotic resistance, and zoonotic impact. Infection and Drug Resistance, 13:3255–3265.
Publisher | Google Scholor - Ali, A., Ullah, S., Numan, M., Almutairi, M., Alouffi, A., & Tanaka, T. (2023). First report on tick-borne pathogens detected in ticks infesting stray dogs near butcher shops. Frontiers in Veterinary Science, 10.
Publisher | Google Scholor - Alkuraythi, D., & Alkhulaifi, M. (2024). Methicillin-resistant Staphylococcus aureus prevalence in food-producing animals and food products in Saudi Arabia: A review. Veterinary World, 1753–1764.
Publisher | Google Scholor - Alshomrani, M., Alharbi, A., Alshehri, A., Arshad, M., & Dolgum, S. (2023). Isolation of Staphylococcus aureus urinary tract infections at a community-based healthcare center in Riyadh. Cureus.
Publisher | Google Scholor - Ataya, H., Soliman, S., Kayaf, A., Marouf, S., & Al-Amry, K. (2023). Incidence, bacterial causes, and antibiotic resistance patterns of urinary tract infection in pet animals. Journal of Applied Veterinary Sciences.
Publisher | Google Scholor - Avberšek, J., Golob, M., Papić, B., Dermota, U., Košnik, I., Kušar, D., … Zdovc, I. (2020). Livestock-associated methicillin-resistant Staphylococcus aureus: Establishing links between animals and humans on livestock holdings. Transboundary and Emerging Diseases, 68(2):789–801.
Publisher | Google Scholor - Bartges, J. W. (2004). Diagnosis of urinary tract infections. Veterinary Clinics of North America: Small Animal Practice, 34(4):923–933.
Publisher | Google Scholor - Bowman, D. D. (2014). Georgis’ Parasitology for Veterinarians (10th ed.). Elsevier.
Publisher | Google Scholor - Çelik, B., Ayan, A., Yılmaz, A., Çelik, Ö., Kılınç, Ö., & Ayan, Ö. (2021). Molecular identification of Ehrlichia canis in Rhipicephalus sanguineus ticks from Siirt Province. Acta Veterinaria, 71(3):247–255.
Publisher | Google Scholor - Chao, L., Robinson, M., Liang, Y., & Shih, C. (2022). First detection and molecular identification of Rickettsia massiliae, a human pathogen, in Rhipicephalus sanguineus ticks collected from southern Taiwan. PLoS Neglected Tropical Diseases, 16(11):e0010917.
Publisher | Google Scholor - Charles, R., Pow-Brown, P., Gordon-Dillon, A., Blake, L., Nicholls, S., Jordan, A., … Georges, K. (2024). Completing the puzzle: A cluster of hunting dogs with tick-borne illness from a fishing community in Tobago, West Indies. Pathogens, 13(2):161.
Publisher | Google Scholor - Cornell, K. K. (2000). Canine prostate cancer: Epidemiology and clinical management. Journal of Veterinary Internal Medicine, 14(6):556–562.
Publisher | Google Scholor - Couper, L., MacDonald, A., & Mordecai, E. (2020). Impact of prior and projected climate change on US Lyme disease incidence. Global Change Biology, 27(4):738–754.
Publisher | Google Scholor - Dalton, K., Ruble, K., Redding, L., Morris, D., Mueller, N., Thorpe, R., … Davis, M. (2021). Microbial sharing between pediatric patients and therapy dogs during hospital animal-assisted intervention programs. Microorganisms, 9(5):1054.
Publisher | Google Scholor - Dube, F., Söderlund, R., Salomonsson, M., Troell, K., & Börjesson, S. (2021). Benzylpenicillin-producing Trichophyton erinacei and methicillin-resistant Staphylococcus aureus carrying the mecC gene on European hedgehogs – A pilot study. BMC Microbiology, 21(1).
Publisher | Google Scholor - Fetsch, A., Etter, D., & Johler, S. (2021). Livestock-associated meticillin-resistant Staphylococcus aureus—Current situation and impact from a one health perspective. Current Clinical Microbiology Reports, 8(3):103–113.
Publisher | Google Scholor - Gaddafi, M., Yakubu, Y., Garba, B., Bellò, M., Musawa, A., & Lawal, H. (2020). Occurrence and antimicrobial resistance patterns of methicillin-resistant Staphylococcus aureus (MRSA) among practicing veterinarians in Kebbi State, Nigeria. Folia Veterinaria, 64(4):55–62.
Publisher | Google Scholor - Godani, S., N., C., & M., W. (2022). Zoonotic tick-borne pathogens in ticks at human–wildlife–livestock interfaces in Kwale County, Kenya.
Publisher | Google Scholor - Haynes, E., Garrett, K., Grunert, R., Bryan, J., Sidouin, M., Oaukou, P., … Cleveland, C. (2024). Surveillance of tick-borne pathogens in domestic dogs from Chad, Africa, in a one health context.
Publisher | Google Scholor - Heaton, C., Gerbig, G., Sensius, L., Patel, V., & Smith, T. (2020). Staphylococcus aureus epidemiology in wildlife: A systematic review. Antibiotics, 9(2):89.
Publisher | Google Scholor - Irwin, P. J. (2002). Companion animal parasitology: Clinical and diagnostic significance. International Journal for Parasitology, 32(5):583–593.
Publisher | Google Scholor - Johnson, T., Armstrong, J., Johnston, B., Merino-Velasco, I., Jamborová, I., Singer, R., … Bender, J. (2022). Occurrence and potential transmission of extended-spectrum beta-lactamase-producing extraintestinal pathogenic and enteropathogenic Escherichia coli in domestic dog faeces from Minnesota. Zoonoses and Public Health, 69(7):888–895.
Publisher | Google Scholor - Joosten, P., Cleven, A., Sarrazin, S., Paepe, D., Sutter, A., & Dewulf, J. (2020). Dogs and their owners have frequent and intensive contact. International Journal of Environmental Research and Public Health, 17(12):4300.
Publisher | Google Scholor - Krueger, M., Bajric, S., Godden, S., Bender, J., Mani, R., & Sreevatsan, S. (2025). Phylogenomic associations among methicillin-resistant Staphylococcus aureus isolates derived from pets, dairies, and humans. Microbiology Spectrum, 13(6).
Publisher | Google Scholor - Ling, G. V., et al. (1998). Urolithiasis in dogs: Risk factors, management, and prevention. Journal of the American Veterinary Medical Association, 213(9):1256–1263.
Publisher | Google Scholor - Lobetti, R. G. (2014). Bacterial prostatitis in dogs. Compendium on Continuing Education for the Practicing Veterinarian, 36(7):E1–E5.
Publisher | Google Scholor - Mahlobo-Shwabede, S., Zishiri, O., Thekisoe, O., & Makalo, M. (2021). Molecular detection of Coxiella burnetii, Rickettsia africae and Anaplasma species in ticks from domestic animals in Lesotho. Pathogens, 10(9):1186.
Publisher | Google Scholor - Moreno-Madriñán, M., & Kontowicz, E. (2023). Stocking density and homogeneity, considerations on pandemic potential. Zoonotic Diseases, 3(2):85–92.
Publisher | Google Scholor - Mummah, R., Hoff, N., Rimoin, A., & Lloyd-Smith, J. (2020). Controlling emerging zoonoses at the animal-human interface. One Health Outlook, 2(1).
Publisher | Google Scholor - Noden, B., Bradt, D., & Sanders, J. (2021). Mosquito-borne parasites in the Great Plains: Searching for vectors of nematodes and avian malaria parasites. Acta Tropica, 213, 105735.
Publisher | Google Scholor - Pascoe, E., Nava, S., Labruna, M., Paddock, C., Levin, M., Marcantonio, M., … Foley, J. (2022). Predicting the northward expansion of tropical lineage Rhipicephalus sanguineus sensu lato ticks in the United States and its implications for medical and veterinary health. PLoS One, 17(8):e0271683.
Publisher | Google Scholor - Paudel, S., Guedry, S., Obernuefemann, C., Hultgren, S., Walker, J., & Kulkarni, R. (2023). Defining the roles of pyruvate oxidation, TCA cycle, and mannitol metabolism in methicillin-resistant Staphylococcus aureus catheter-associated urinary tract infection. Microbiology Spectrum, 11(4).
Publisher | Google Scholor - Ramzan, M., Murtaza, G., Saghira, A., Munawar, N., Ullah, A., Ejaz, A., … Fasiha, K. (2021). Techniques for managing ticks and tick-borne diseases under changing climate: A review. Egyptian Academic Journal of Biological Sciences B Zoology, 13(1):117–128.
Publisher | Google Scholor - Samutela, M., Kwenda, G., Simulundu, E., Nkhoma, P., Higashi, H., Frey, A., … Hang’ombe, B. (2021). Pigs as a potential source of emerging livestock-associated Staphylococcus aureus in Africa: A systematic review. International Journal of Infectious Diseases, 109, 38–49.
Publisher | Google Scholor - Sánchez-Montes, S., Salceda-Sánchez, B., Bermúdez, S., Aguilar-Tipacamú, G., Ballados-González, G., Huerta, H., … Colunga-Salas, P. (2021). Rhipicephalus sanguineus complex in the Americas: Systematic, genetic diversity, and geographic insights. Pathogens, 10(9):1118.
Publisher | Google Scholor - Shih, C. (2025). Molecular survey and genetic analysis of Ehrlichia canis in Rhipicephalus sanguineus ticks infesting dogs in northern Taiwan. Microorganisms, 13(6):1372.
Publisher | Google Scholor - Singla, R., Mishra, A., Joshi, R., Jha, S., Sharma, A., Upadhyay, S., … Medhi, B. (2020). Human–animal interface of SARS-CoV-2 (COVID-19) transmission: A critical appraisal of scientific evidence. Veterinary Research Communications, 44(3–4):119–130.
Publisher | Google Scholor - Smith, J. (2008). Canine prostatic disease: A review. New Zealand Veterinary Journal, 56(1):1–10.
Publisher | Google Scholor - Tahulending, J., Tulung, M., Pelealu, J., & Doda, D. (2022). Molecular detection of pathogenic bacteria in Rhipicephalus sanguineus (sensu lato) ticks from Bitung, North Sulawesi, Indonesia. Biodiversitas Journal of Biological Diversity, 23(12).
Publisher | Google Scholor - Tang, Y., Qiao, Z., Wang, Z., Li, Y., Ren, J., Wen, L., … Jiao, X. (2021). The prevalence of Staphylococcus aureus and the occurrence of MRSA CC398 in monkey feces in a zoo park in eastern China. Animals, 11(3):732.
Publisher | Google Scholor - Villarroel, P., Chaiphongpachara, T., Nurtop, E., Laojun, S., Pangpoo-nga, T., Songhong, T., … Wichit, S. (2024). Severe fever with thrombocytopenia syndrome virus (SFTSV) in Thailand: Seroprevalence study in humans and molecular detection in ticks.
Publisher | Google Scholor - Wall, R., & Shearer, D. (2001). Veterinary ectoparasites: Biology, pathology and control. Blackwell Science.
Publisher | Google Scholor - Yudhanto, S., Hung, C., Maddox, C., & Varga, C. (2022). Antimicrobial resistance in bacteria isolated from canine urine samples submitted to a veterinary diagnostic laboratory, Illinois, United States. Frontiers in Veterinary Science, 9.
Publisher | Google Scholor