Case Report
The Hidden Face of Lupus: Overlapping Aseptic Meningitis and Intracranial Hypertension as an Unusual Neuroinflammatory Onset
- Chaïmaâ Zeroual *
- Leïla Barakat
- Mina Moudatir
- Khadija Echchilali
- Safaâ Mourabit
- Hassan El Kabli
Department of Internal Medicine & Geriatrics, Ibn Rochd University Hospital Center, Casablanca, Morocco.
*Corresponding Author: Chaïmaâ Zeroual, Department of Internal Medicine & Geriatrics, Ibn Rochd University Hospital Center, Casablanca, Morocco.
Citation: Zeroual C, Barakat L, Moudatir M, Echchilali K, Mourabit S, et al. (2025). The Hidden Face of Lupus: Overlapping Aseptic Meningitis and Intracranial Hypertension as an Unusual Neuroinflammatory Onset, Journal of Brain Research and Neurology, BioRes Scientia Publishers. 4(1):1-5. DOI: 10.59657/2992-9768.brs.25.016
Copyright: © 2025 Chaïmaâ Zeroual, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: June 28, 2025 | Accepted: July 12, 2025 | Published: July 19, 2025
Abstract
Background: Overlapping Intracranial hypertension (IH) and aseptic meningitis (AM) is an uncommon but serious initial manifestation of systemic lupus erythematosus (SLE). While IH typically presents with headache, papilledema and visual symptoms, AM is marked clinically by the presence of fever, headache and meningismus. In the absence of accurate and prompt management, IH may unfortunately lead to permanent vision loss and AM to cognitive or behavioral complications.
Case Description: 17-year-old adolescent presented with severe headache, fever and visual disturbances. Ophthalmology examination revealed papilledema and lumbar puncture showed elevated intracranial pressure. In the cerebrospinal fluid, there was pleocytosis with a lymphocytic predominance. Imaging excluded mass lesions or thrombosis and laboratory findings confirmed SLE. The patient received corticosteroid therapy resulting in rapid and sustained resolution of the meningeal syndrome. Whereas for IH, ventriculoperitoneal shunt placement was necessary.
Conclusion: SLE can rarely present with an overlap of IH and AM, often posing diagnostic challenges especially in young patients. Early recognition is critical, as delays risk permanent ocular and neurological damage.
Keywords: intracranial hypertension; aseptic meningitis; systemic lupus erythematosus; aseptic meningitis
Introduction
Systemic lupus erythematosus (SLE) is a complex autoimmune disease characterized by multisystem involvement and broad spectrum of clinical manifestations, including neuropsychiatric complications [1]. The latter affect 12–95% of SLE patients [2], with approximately 30% directly attributed to SLE activity [3]. Common symptoms include cognitive dysfunction, headache, mood disorders, seizures, psychosis, and sensory alterations. Juvenile-onset SLE shows higher neuropsychiatric involvement (up to 70%) and is linked to increased morbidity [4]. Intracranial hypertension (IH) and aseptic meningitis (AM) are rare manifestations that are often associated with diagnostic challenges, especially in younger population (5,6,9). IH in SLE typically manifests with headache, papilledema and elevated cerebrospinal fluid (CSF) pressure in the absence of space-occupying injuries or other identifiable causes. In parallel, AM corresponds to the inflammation of the meninges and is developed by CSF testing negative for bacteria or fungi. Prompt detection of both IH and AM as an initial presentation of SLE can prevent irreversible complications mainly the visual and neurological ones. We describe 17-year-old adolescent whose SLE was revealed at diagnosis by co-occurrence of IH and AM. Timely management made of corticosteroid therapy and ventriculoperitoneal (VP) shunt led to good evolution. This case report aims to raise awareness among clinicians about the existence of atypical neurological presentations that could possibly reveal SLE. An early recognition will avoid misdiagnosis and optimization treatment outcomes.
Case Report
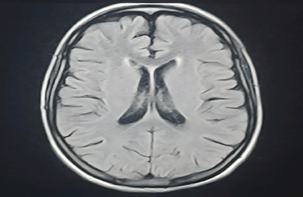
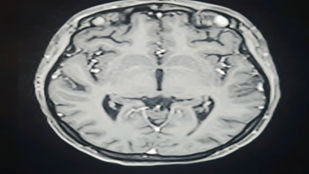
She was a 17-year-old adolescent with no significance past medical history, who was hospitalized for a complete intracranial hypertension syndrome associated with a fever meningeal syndrome. The symptoms had begun gradually two weeks prior Upon admission, the patient was feverish at 40°C, had a Glasgow Coma Scale (GCS) score of 14/15 (eye response E5, verbal response V4, motor response M5), a normal blood pressure of 110/70 mmHg, tachycardia at 103 bpm, eupneic at 20 breaths per minute, with good oxygen saturation in room air (95%). Neurological examination revealed neck stiffness with positive Kernig and Brudzinski signs. Furthermore, there was no involvement of the cranial nerve pairs, notably III, IV, VII, and VIII. Global and segmental muscle strength were preserved, deep tendon reflexes were normal, and the plantar reflex was indifferent. Ophthalmological examination showed bilateral stage III papilledema. brain magnetic resonance imaging (MRI) angiography was unremarkable (Figures 1,2) and CSF analysis revealed clear (rock water) fluid, 100% lymphocytic, with no organisms detected (Table 1).
Figure 1: Axial view of brain MRI before Gadolinium injection.
Figure 2: Axial view of brain MRI after Gadolinium injection.
Table 1: The results of the cytobacteriological, chemical, and immunological analysis of the cerebrospinal fluid.
| Cerebrospinal Fluid (CSF) | |
| Cytological Study | |
| Leukocytes | 4068/mm³ |
| Neutrophils | 0% |
| Lymphocytes | 100% |
| Bacteriological Study | |
| Direct Examination | No organisms |
| Culture | Sterile |
| Chemical Study | |
| Proteins | 0.44 g/L |
| Glucose | 0.55 g/L |
| Immunological Study | |
| Antinuclear Antibodies (ANA) | Absent |
The measurement of CSF pressure was elevated, reaching 31 cm H₂O. On laboratory evaluation, the patient presented with a normochromic, normocytic, and regenerative anemia, with hemoglobin at 10 g/ dL and a positive direct Coombs test. Leukopenia was observed at 2,640/mm³, accompanied by both neutropenia and lymphopenia, as well as thrombocytopenia at 113,000/mm³. Both C- reactive protein and procalcitonin levels were negative. Fibrinogen concentration was within normal limits at 2.76 g/L. blood urea was measured at 0.3 g/L, and serum creatinine at 6.1 mg/L. Furthermore, transaminase levels were appropriate. Repeated aerobic, anaerobic, and fungal blood cultures yielded no growth. Cytobacteriological examination of the urine did not identify any pathogens, and the 24-hour proteinuria was negative at 0.1 g/ day. Transthoracic echocardiography revealed no abnormalities, and Thoracoabdominopelvic CT scan was unremarkable. The diagnosis of SLE was established according to the 2019 American College of Rheumatology (ACR)/ European League Against Rheumatism (EULAR) criteria, with a total score of 25 points (Table 2).
Table 2: The 2019 ACR/EULAR classification criteria for systemic lupus erythematosus enabled the diagnosis to be established in our patient.
| Parameter | Points |
| ANA positive by indirect immunofluorescence (IIF) at 1: 320, homogeneous pattern | Entry criterion |
| Fever at 40°C | 2 |
| Leukopenia | 3 |
| Thrombocytopenia | 4 |
| Autoimmune hemolysis | 4 |
| Low C3 at 0.5 g/L | 4 |
| Low C4 at 0.06 g/L | |
| Positive lupus anticoagulant | 2 |
| Anti- cardiolipin IgM (+) at 56, IgG (-) | |
| Anti- β 2GPI IgM (+) at 210, IgG (-) | |
| Anti-native DNA antibodies (+) | 6 |
On a therapeutic level, she received a bolus of methylprednisolone at a dosage of 500 mg per day for three days, which was followed by full-dose oral corticosteroid therapy in combination with acetazolamide and antiplatelet therapy. Synthetic antimalarials were contraindicated in the patient due to the alteration of the bilateral visual field. Neither corticosteroid therapy nor acetazolamide was sufficient to resolve the IH, and the patient ultimately required VP shunt to achieve clinical improvement. The course was favorable, characterized by the disappearance of the meningeal syndrome and the resolution of IH. From a laboratory perspective, the hematological manifestations of SLE were corrected.
Discussion
SLE is multisystem autoimmune disease with diverse clinical presentations, among which neuropsychiatric manifestations, called neuropsychiatric systemic lupus erythematosus (NPSLE), are particularly challenging due to their heterogeneity and potential severity. While central nervous system (CNS) involvement es well-recognized in SLE, the co-existence of IH and AM as initial presentation of SLE is rare phenomenon that has been rarely described previously. Understanding the interplay between these two entities in the context of SLE is crucial for timely diagnosis and management. Uncommon, AM in SLE has been reported in approximately 1.4–2.0% of patients [7]. The clinical presentation typically includes fever, headache and sometimes nuchal rigidity. classical meningeal signs may be absent or subtle. CSF analysis generally reveals lymphocytic pleocytosis, elevated protein levels, and normal glucose concentrations, distinguishing Item from bacterial meningitis. The pathogenesis es thought to involve immune -mediated inflammation of the meninges, possibly triggered by autoantibody deposition or cytokine release [8,9].
IH, also known as pseudotumor cerebri, is another rare but recognized NPSLE. Its pathophysiology in lupus patients mainly involves impaired CSF absorption and blood-brain barrier dysfunction. Clinically, IH presents with severe persistent headaches, visual disturbances due to papilledema and sometimes cranial nerve palsies. Diagnosis es established by demonstrating elevated CSF opening pressure on lumbar puncture with neuroimaging excluding mass lesions or venous thrombosis [10,11]. The presence of IH in SLE patients can be particularly challenging to manage, as it may mimic or coexist with other CNS manifestations requiring careful evaluation.
Several clinical and therapeutic findings in our case support a true overlap of AM and IH, rather than IH as a secondary consequence of AM. Both syndromes appeared simultaneously, with typical features of AM (fever, meningeal signs, lymphocytic CSF) and confirmed IH (papilledema, elevated opening pressure at 31 cm H₂O), while brain imaging was entirely normal. The CSF showed no marked protein elevation or hypoglycorrhachia, arguing against a reactive IH. Importantly, corticosteroid therapy led to rapid resolution of the meningeal syndrome but failed to improve IH, which required a ventriculoperitoneal shunt. This differential treatment response strongly suggests distinct pathophysiological mechanisms, supporting the notion of an overlap AM/IH.
Management of NPSLE manifestations centers on controlling underlying lupus activity and alleviating intracranial pressure. The treatment requires a tailored approach based on the specific neurological manifestations and their severity. High-dose corticosteroids are the cornerstone for acute and severe neuroinflammatory presentations including IH and AM [7, 12, 13].
The choice of immunosuppressant agents depends on disease severity, organ involvement and patient- specific factors including tolerability and comorbidities. Cyclophosphamide (CYC) mycophenolate mofetil (MMF), azathioprine (AZA) and methotrexate are key immunosuppressants used in managing NPSLE. CYC is a potent alkylating agent often reserved for severe or life- threatening cases, such as lupus cerebritis or transverse myelitis. CYC is typically administered as monthly intravenous pulses during induction therapy [14,15]. MMF, an inhibitor of lymphocyte proliferation, has shown comparable efficacy to CYC in moderate NPSLE and is increasingly favored due to its more favorable safety profile [16]. AZA, a purine synthesis inhibitor, is primarily used for maintenance therapy or in milder NPSLE. Intrathecal methotrexate combined with dexamethasone has shown benefit in severe or refractory NPSLE, particularly in cases with CNS involvement. This regimen may reduce disease activity and relapse rates by directly targeting CNS inflammation, though Item remains an off-label and specialized treatment option [17,18].
Biological therapies, particularly rituximab and belimumab, have shown promise in treating NPSLE, especially when standard immunosuppressive treatments fail. Rituximab has demonstrated rapid improvement in CNS- related neuropsychiatric symptoms, while belimumab has shown favorable effects in some patients, although robust clinical trial data for these specific manifestations are limited and more research es needed [19,20].
Adjunctive therapies, including acetazolamide for IH and symptomatic treatments such as antiepileptics or antipsychotics, may be necessary to control specific symptoms. When neuropsychiatric Symptoms are thought to reflect a thrombotic process, particularly in the presence of antiphospholipid antibodies, anticoagulation or antiplatelet therapy es indicated. VP shunt is considered in patients with SLE who develop IH or hydrocephalus that does not respond to medical management with corticosteroids and immunosuppressants. The VP shunt works by diverting excess CSF from the brain's ventricles to the peritoneal cavity, thereby relief intracranial pressure and improving symptoms such as headache, cognitive dysfunction, gait disturbance and incontinence [21]. While the procedure es generally effective in providing symptomatic relief, it carries risks such as infection, shunt malfunction and the potential need for future adjustments or replacements [22].
Conclusion
The dual presentation of IH and AM as initial manifestation of SLE remains rare underscoring the importance of a multidisciplinary approach. Early recognition and aggressive management are essential to prevent permanent neurological, ocular sequelae and optimize patient outcomes. Further research and accumulation of case data are needed to improve characterize atypical neurological manifestations of SLE and refine their therapeutic strategies.
Declarations
Disclosure of Conflict of Interest
No conflict of interest to be disclosed.
Sources of Funding
This study was not supported by any institution or company.
Statement of Ethics Approval
All procedures performed in this study are in accordance with the ethical standards of the institutional and/or national research committee.
Statement of Informed Consent
Written Informed consent was obtained from the patient for publication of this case report.
References
- Schwartz N, Stock AD, Putterman C. (2019). Neuropsychiatric lupus: new mechanistic insights and future treatment directions. Nat Rev Rheumatol. 15(3):137-152.
Publisher | Google Scholor - Unterman A, Nolte JE, Boaz M, Abady M, Shoenfeld Y, et al. (2011). Neuropsychiatric syndromes in systemic lupus erythematosus: to meta-analysis. Semin Arthritis Rheum. 41:1-11.
Publisher | Google Scholor - Govoni M, Hanly JG. (2020). The management of neuropsychiatric lupus in the 21st century: still SW many unmet needs? Rheumatology. 59(5):52-62.
Publisher | Google Scholor - Natoli V, Charras A, Hahn G, Hedrich CM. (2023). Neuropsychiatric involvement in juvenile onset systemic lupus erythematosus (JSLE). Molecular and Cellular Pediatrics. 10(1):5.
Publisher | Google Scholor - Pugliese I, Posada M, Shinchi M, Aguirre-Valencia D. (2024). Intracranial hypertension as the first manifestation of systemic lupus erythematosus: A case report. Heliyon. 10(20):e38880.
Publisher | Google Scholor - Rajasekharan C, Renjith SW, Marzook A, Parvathy R. (2013). Idiopathic intracranial hypertension as the initial presentation of systemic lupus erythematosus. BMJ Case Rep. bcr2012007886.
Publisher | Google Scholor - Tsukamoto M, Shimamoto M, Terashima T, Seta N. (2018). Aseptic Meningitis With Systemic Lupus Erythematosus: Case Report and Review of the Literature. Arch Rheumatol. 34(1):108-111.
Publisher | Google Scholor - Kim MH, Kim D, Koo Y, Oh SH, Kim HS, et al. (2008). A case of systemic lupus erythematosus presenting as aseptic meningitis. J Neurocrit Care. 1:62-64.
Publisher | Google Scholor - Cheok LH, Lee WWH, Cheng WM, Cheong YK, Teh CL. (2022). Aseptic meningitis as initial manifestation of systemic lupus erythematosus: Case Series. Lupus. 31(3):373-377.
Publisher | Google Scholor - Maloney K. (2013). Idiopathic intracranial hypertension as an initial presentation of systemic lupus erythematosus. BMJ Case Rep. bcr2013010223.
Publisher | Google Scholor - Shah B, Amprayil MI, Taparia R, Pokhrel SM. (2018). Idiopathic intracranial hypertension as a cause of severe intractable headache in a patient with Systemic Lupus Erythematosus: A case report from Eastern Nepal. Journal of Advances in Internal Medicine. 7(2):40-42.
Publisher | Google Scholor - Zou M, Jiang X, Chen H, Yuan F. (2024). Systemic lupus erythematosus with chronic persistent intracranial hypertension: A case report. Lupus. 33(3):293-297.
Publisher | Google Scholor - Kalanie H, Abbasi M, Harandi AA. (2019). Intracranial hypertension as an initial clinical manifestation of systemic lupus erythematosus. Interdisciplinary Neurosurgery.17:129-130.
Publisher | Google Scholor - Bertsias GK, Ioannidis JP, Aringer M, Bollen E, et al. (2010). EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis. 69(12):2074-2082.
Publisher | Google Scholor - Hanly JG, Su L, Urowitz MB, Romero-Diaz J, Gordon C, et al. (2020). Prospective analysis of neuropsychiatric events in an international disease inception cohort of patients with systemic lupus erythematosus. Ann Rheum Dis. 79(1):56-64.
Publisher | Google Scholor - Appenzeller S, Cendes F, Costallat LT. (2012). Mycophenolate mofetil in the treatment of neuropsychiatric lupus erythematosus. J Rheumatol. 39(4):843-848.
Publisher | Google Scholor - Nie Y, Sun B, He X, Zheng M, Wu D, et al. (2023). The influence of intrathecal injection of methotrexate and dexamethasone on neuropsychiatric systemic lupus erythematosus (NPSLE): to retrospective cohort study of 386 patients. Arthritis Beef Ther. 25(1):50.
Publisher | Google Scholor - Zhou HQ, Leng XM and Zhang FC. Neuropsychiatric manifestations in systemic lupus erythematosus and the treatment of intrathecal methotrexate plus dexamethasone. Zhonghua Yi Xue Za Zhi. 86(11):771-774.
Publisher | Google Scholor - Liu Y, Tu Z, Zhang X, Du K, et al. (2022). Pathogenesis and treatment of neuropsychiatric Systemic lupus erythematosus: A review. Frontiers in Cell and Developmental Biology. 10:998328.
Publisher | Google Scholor - Legge AC, Hanly JG. (2024). Recent Advances in the diagnosis and management of neuropsychiatric lupus. Nat Rev Rheumatol. 20:712-728.
Publisher | Google Scholor - Fowler JB, De Jesus O, Mesfin FB. (2023). Ventriculoperitoneal shunt. In: StatPearls.
Publisher | Google Scholor - Merkler AE, Ch'ang J, Parker WE, Murthy SB, al. (2017). The rate of complications after ventriculoperitoneal shunt surgery. World Neurosurgery. 98:654-658.
Publisher | Google Scholor