Research Article
Regenerative Medicine Computational Innovation: AI, Biomaterials, Molecular Engineering in BME
1Department of Computing, Institute of Electrical and Electronics Engineers (IEEE), USA.
2Department of Computer Science, American International University-Bangladesh (AIUB), Bangladesh.
*Corresponding Author: Zarif Bin Akhtar
Citation: Zarif B. Akhtar, Ahmed T. Rawol. (2026). Regenerative Medicine Computational Innovation: AI, Biomaterials, Molecular Engineering in BME, Journal of Clinical Research and Clinical Trials, BioRes Scientia Publishers. 5(1):1-17. DOI: 10.59657/2837-7184.brs.26.063
Copyright: © 2026 Zarif Bin Akhtar, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: November 24, 2025 | Accepted: December 30, 2025 | Published: January 06, 2026
Abstract
Recent advancements in molecular engineering and computational biology have revolutionized healthcare, diagnostics, and therapeutic development. This study explores the interdisciplinary integration of artificial intelligence (AI), machine learning (ML), and bioinformatics in regenerative medicine, biomaterials, and tissue engineering. Key advancements such as organ-on-a-chip models, AI-driven bioprinting, and computational simulations for tissue regeneration are discussed in the context of their biomedical applications. Additionally, this research examines the role of predictive modeling, biomedical data analytics, and AI-assisted drug discovery in immune engineering, precision medicine, and gene editing. Through a computational and case study-driven approach, this work provides insights into how AI and machine learning are shaping the future of biologics, drug screening, and personalized medicine. The findings underscore the transformative potential of data-driven methodologies in accelerating medical research and therapeutic interventions, bridging the gap between regenerative medicine and biomedical informatics.
Keywords: artificial intelligence; machine learning; computational biology; bioinformatics; biomedical data mining; regenerative medicine; bioprinting; precision medicine
Introduction
The domain of molecular engineering is an innovative, interdisciplinary field that controls the precise design and manipulation of molecular interactions to develop focused materials, systems, and processes. By integrating artificial intelligence (AI), computational biology, and machine learning (ML), molecular engineering is transforming biomedicine, regenerative medicine, and personalized healthcare [1-3]. This "bottom-up" approach enables the engineering of macroscopic systems by controlling molecular structures, bridging multiple scientific domains, including bioinformatics, biomedical engineering, and computational modeling.
Unlike traditional trial-and-error methods, molecular engineering employs data-driven computational approaches, allowing researchers to analyze complex biological systems with high precision [4-6]. These advancements facilitate the development of novel biomedical applications, including AI-assisted biomaterials design, predictive modeling for drug discovery, and bioinformatics-driven tissue engineering [7-9]. Additionally, computational simulations are enhancing bioprinting, organ-on-a-chip models, and regenerative therapies, bridging the gap between experimental research and clinical applications.
Recent breakthroughs in AI-driven regenerative medicine are revolutionizing treatments for previously incurable conditions. Technologies such as machine learning-assisted gene editing (e.g., CRISPR-Cas9), deep learning for tissue imaging, and AI-driven drug discovery pipelines are reshaping the landscape of precision medicine [10-12]. Moreover, innovations in AI-powered CAR T-cell therapy, bioinformatics-based drug screening, and wearable health devices are improving patient outcomes, disease prevention, and medical decision-making.
This research investigates the interdisciplinary field of molecular engineering and computational biology, with a particular focus on their applications in regenerative medicine, biomedical informatics, and AI-assisted drug development. By exploring recent advancements in these areas, this study aims to provide a comprehensive, computationally-driven perspective on how AI, ML, and bioinformatics are shaping the future of healthcare, biomedical research, and personalized medicine.
Methods and Experimental Analysis
This research employed a systematic, computationally-driven approach to investigate the impact of molecular engineering and regenerative medicine within the field of biomedical informatics. The study leveraged artificial intelligence (AI), machine learning (ML), and bioinformatics tools to analyze large-scale biomedical datasets and extract meaningful insights for personalized medicine and AI-driven biomedical applications.
Background Research Explorations and Data Acquisition
The first phase involved conducting an extensive Background Research Exploration Investigations to identify research gaps at the intersection of computational biology, AI, and regenerative medicine. This review established the study's context, highlighting recent advancements in machine learning-assisted biomaterials design, AI-driven tissue engineering, and predictive modeling in regenerative medicine.
Data Collection and Preprocessing
Biomedical data was collected and analyzed using advanced computational platforms, including KNIME, Python, and MATLAB, to process large-scale datasets related to biomaterials, AI-assisted drug discovery, and regenerative medicine applications. A series of data preprocessing steps were implemented to ensure data integrity, normalization, and relevance.
AI and Machine Learning Techniques
A range of machine learning algorithms and data mining techniques were applied to extract patterns and trends from the biomedical datasets.
The study utilized:
- Supervised and unsupervised learning models to classify and cluster biomaterial properties.
- Deep learning architectures for biomedical image analysis in tissue engineering and regenerative medicine.
- Predictive modeling frameworks to optimize AI-driven biomaterials selection and regenerative therapy outcomes.
Data Visualization and Performance Benchmarking
To facilitate interpretation, graphical representations, heatmaps, and AI-driven visual analytics were utilized to present insights into the effectiveness of computational methods. The study compared the performance of AI-powered regenerative medicine models with traditional biomedical approaches, benchmarking effectiveness using standard evaluation metrics such as accuracy, precision, recall, and F1-score.
Results Interpretation and Future Prospects
The findings were analyzed in relation to the research objectives, emphasizing their implications for AI-assisted regenerative medicine. The study explored future directions for machine learning-enhanced tissue engineering, AI-driven precision medicine, and bioinformatics-based therapeutic discovery. Additionally, the research acknowledged limitations in dataset availability, model generalization, and computational scalability, proposing future investigations into integrating generative AI and federated learning models for biomedical applications.
This methodology provided a comprehensive computational framework for integrating molecular engineering, AI, and regenerative medicine, paving the way for future advancements in biomedical informatics and personalized healthcare solutions.
Background Research and Investigative Exploration for Available Knowledge
Regenerative medicine is an evolving field within biomedical engineering that aims to restore or replace damaged tissues and organs by enhancing cellular activity. Tissue regeneration is often constrained by limited intrinsic healing potential, impacting key cellular processes such as migration, proliferation, and differentiation [1-11]. Advances in biomaterial science have facilitated the development of innovative strategies to overcome these challenges, enabling more effective and patient-friendly regenerative treatments. However, conventional in vitro cell culture systems, primarily based on polystyrene dishes, fail to replicate the complex microenvironment of native tissues. This discrepancy results in altered cellular behaviors in vitro, leading to inconsistencies between preclinical laboratory findings and clinical outcomes. Addressing this gap requires the development of biomaterials that can more accurately mimic the native extracellular matrix (ECM) and enhance cellular function both in vitro and in vivo.
Role of Biomaterials in Enhancing Cell Activity
Biomaterials are essential in regenerative medicine as they support cellular functions by providing structural and biochemical cues that promote tissue regeneration. These materials are categorized into natural and synthetic biomaterials, each offering unique advantages [11-22]. Natural biomaterials such as collagen, gelatin, alginate, chitosan, and silk fibroin are widely used due to their biocompatibility and bioactivity. Synthetic biomaterials, including poly (lactic acid) (PLA) and poly (lactic-co-glycolic acid) (PLGA), provide tunable mechanical properties and controlled degradation rates for various applications in tissue engineering and drug delivery.
Natural Biomaterials
Collagen and Gelatin: Collagen is a primary ECM component that supports cellular adhesion, differentiation, and proliferation. It has been widely employed in tissue engineering for bone, cartilage, muscle, and cancer research. Gelatin, derived from collagen, exhibits high biocompatibility and can be used in hydrogels to promote cell viability and controlled drug release. Gelatin-based biomaterials have shown efficacy in enhancing cardiac function and stimulating the proliferation of stromal and endothelial cells in regenerative applications.
Alginate: Derived from seaweed, alginate is a polysaccharide commonly used in encapsulation systems for stem cells and tissue engineering. Alginate-based hydrogels have been employed to support osteogenesis and mineralization while also serving as injectable cell delivery systems that eventually degrade, preventing long-term interference with tissue regeneration.
Chitosan: A biopolymer derived from chitin; chitosan is recognized for its biocompatibility and versatility. It has been integrated into scaffolds for bone, cartilage, nerve, and skin tissue engineering. Chitosan-based hydrogels and composites demonstrate enhanced osteoconductivity and bioactivity, facilitating tissue-specific differentiation and regeneration.
Silk Fibroin: Extracted from silkworms, silk fibroin is utilized in scaffolds for bone and cartilage regeneration. Its unique mechanical properties, combined with the ability to support cell adhesion and differentiation, make it a promising biomaterial for diverse tissue engineering applications.
Synthetic Biomaterials
Agarose: Comprising D-galactose and 3.6-anhydro-L-galactopyranose units, agarose has been extensively used due to its water absorption capacity and ability to form hydrogels without harmful crosslinkers. It has demonstrated potential in nerve and cornea regeneration by supporting cell encapsulation and oxygen permeability.
Matrigel: A basement membrane matrix derived from mouse Engelbreth-Holm-Swarm tumor, Matrigel plays a critical role in in vitro cancer modeling. It maintains tumor-like characteristics in cancer cells, aiding in studies on cancer cell behavior, invasion, and drug response. When combined with other biomaterials, Matrigel enhances the accuracy of in vitro tissue engineering models.
Poly (Lactic Acid) (PLA): PLA, with an elastic modulus similar to bone, is widely used in bone tissue engineering. It is often combined with hydroxyapatite (HA) to enhance ECM remodeling and osteogenic potential. PLA-based scaffolds have been optimized using 3D printing technologies, improving osteoblast adhesion and differentiation.
Poly (Lactic-co-Glycolic Acid) (PLGA): PLGA is a biodegradable copolymer extensively used in medical applications such as drug delivery and tissue engineering. By adjusting its composition, PLGA properties can be tuned to match specific regenerative needs. It has been particularly effective in neural tissue engineering, supporting Schwann cell migration and axonal growth, contributing to peripheral nerve regeneration.
The integration of biomaterials with cutting-edge technologies such as 3D bioprinting, nanotechnology, and bioactive coatings continues to expand the possibilities of regenerative medicine [22-33]. Future research should focus on optimizing biomaterial formulations to enhance biocompatibility, degradation rates, and cellular responses.
Additionally, combining biomaterials with advanced biofabrication techniques can improve the structural and functional mimicry of native tissues, bridging the gap between in vitro models and clinical applications. Through these advancements, biomaterials will play an increasingly critical role in achieving successful tissue regeneration and improving patient outcomes in regenerative medicine.
Regenerative Medicine: Computational Biomaterial Applications
Biomaterials play a critical role in regenerative medicine by providing structural support and facilitating tissue regeneration, organ replacement, and chronic disease treatment. With the integration of computational biology, artificial intelligence (AI), and machine learning (ML), biomaterials research has evolved significantly, enhancing their design, optimization, and clinical application. AI-driven predictive modeling, bioinformatics tools, and high-throughput simulations now allow for precise material selection and improved performance of biomaterials in biomedical applications.
AI and Computational Modeling in Biomaterial Design
Various Recent advances within the domain towards biochemistry, molecular biology, and biomedical engineering have enabled the accelerated development of AI-assisted biomaterial scaffolds that mimic the extracellular matrix (ECM).
By leveraging machine learning algorithms and computational simulations, researchers can analyze biomaterial properties such as biodegradability, biocompatibility, mechanical strength, and cellular interactions. This enables the rational design of biomaterials that optimize cell adhesion, differentiation, and tissue regeneration.
Smart Biomaterials for Tissue Engineering
Modern biomaterials, including natural hydrogels (e.g., chitosan, collagen, decellularized tissues) and synthetic polymers (e.g., polyethylene glycol (PEG), polylactic acid (PLA), and polyglycolic acid (PGA)), exhibit tunable properties that can be optimized using computational biology tools. Deep learning models and AI-driven simulations assist in predicting how biomaterials interact with stem cells, immune cells, and tissue microenvironments, allowing for personalized biomaterial designs tailored to specific medical applications.
Computational Challenges and AI-driven Solutions
Despite the tremendous potential of biomaterials, several computational challenges remain in their real-time modeling, biocompatibility assessment, and large-scale fabrication. AI-based generative models can simulate biomaterial behavior, predict potential cytotoxicity risks, and optimize material synthesis. Additionally, multi-scale computational modeling helps in replicating the complex and dynamic interactions of the native cellular microenvironment, enhancing biomaterial performance in tissue engineering.
Digital Twin and Predictive Analytics for Biomaterials
Emerging digital twin technologies-which create AI-driven virtual models of biomaterials and tissue scaffolds-enable real-time biomedical simulations and predictive analytics. These systems integrate biomechanical data, cellular responses, and patient-specific parameters, paving the way for precision medicine-driven biomaterials that align with individual patient needs.
The Future of AI-Assisted Biomaterials in Regenerative Medicine
With the increasing adoption of AI and computational informatics in biomaterials research, the field is witnessing rapid advancements in bioprinting, nanotechnology, and smart biomaterials. AI-driven biomaterial platforms can now:
- Automate biomaterial screening for regenerative applications.
- Optimize material properties for specific organ and tissue regeneration.
- Predict long-term biomaterial interactions with biological systems.
As a consequence, biomaterials are now residing at the apex forefront of AI-driven regenerative medicine, at the same time, bridging the gaps amongst the domains for scientific research and clinical applications. Their attitude towards mimicking the native cellular microenvironment, supported with AI-assisted tissue engineering, and enhance drug delivery systems is primarily driving major transformations within healthcare. As computational biology, AI, and bioinformatics continue to revolutionize biomaterials research, personalized and precision-driven regenerative therapies will become the future standard of medical treatment.
Table 1: Selection of the commercially available biomaterials for regenerative medicine.
| Product | Tissues/Organs | Description | Company |
| AlloDerm® | Skin | Acellular dermal matrix for soft-tissue augmentation and replacement | LifeCell Corp. |
| Apligraf® | Skin | Allogeneic fibroblasts on a bovine collagen I matrix with upper keratinocyte cell layer | Organogenesis |
| Dermagraf® | Skin | Allogeneic fibroblasts on a vicryl mesh scaffold | Shire Regenerative Medicine, Inc |
| GraftJacket® | Skin | Acellular dermal matrix for soft-tissue augmentation and chronic wound treatment | Wright Medical Technology Inc. |
| TransCyte® | Skin | Allogeneic fibroblasts on a nylon mesh with upper silicone layer | Shire Regenerative Medicine, Inc |
| Oasis® Wound Matrix | Skin | Decellularized porcine small intestinal submucosa | Cook Biotech |
| Integra® Bilayer Wound Matrix | Skin | Type I bovine collagen with chondroitin-6-sulfate and silicone | Integra Life Sciences |
| Epicel® | Skin | Autologous keratinocyte cell sheets | Genzyme |
| Carticel® | Cartilage | Autologous chondrocytes | Genzyme |
| NeoCart® | Cartilage | Autologous chondrocytes on type I bovine collagen | Histogenics |
| VeriCartTM | Cartilage | Type I bovine collagen | Histogenics |
| AlloMatrix® | Bone | Demineralized bone matrix combined with calcium sulfate | Weight Medical Technology Inc. |
| Osteocel® Plus | Bone | Allogeneic bone with mesenchymal stem cells | NuVasive |
| Pura-MatrixTM | Bone | Hydrogel composed of a self-assembling peptide | 3DMatrix |
| OsteoscafTM | Bone | Poly (lactic-co-glycolic acid) and calcium phosphate scaffold | Tissue Regeneration Therapeutics |
| INFUSE® bone graft | Bone | Recombinant human bone morphogenetic proteins-2 in combination with bovine type I collagen | Medtronics |
| LifelineTM | Blood vessels | Autologous fibroblast tubular cell sheet integrated with endothelial cells | Cytograft Tissue Engineering |
| Omniflow® | Blood vessels | Polyester mesh with cross-linked ovine collagen | Binova |
| AngineraTM | Heart | Allogeneic fibroblasts on vicryl mesh | Theregen |
| CardioValve® SynerGraft Pulmonary Heart Valve | Heart | Decellularized allogeneic pulmonary valve | Cryolife |
Regenerative Medicine: AI-Driven Advancements
The domain of regenerative medicine has emerged as an evolving, computationally enhanced field focused towards repairing and revitalizing damaged or diseased cells, tissues, organs. By integrating artificial intelligence (AI), machine learning (ML), computational biology, modern regenerative medicine and device integrations is rapidly advancing beyond traditional approaches, offering a much better precision-driven, data-informed therapeutic solutions.
The primary objective remains to restore normal biological function disrupted by injury, disease, or aging, but AI-driven innovations are significantly enhancing the field’s capabilities.
AI and Computational Biology in Regenerative Medicine
Traditional regenerative medicine strategies-such as tissue engineering, therapeutic stem cell applications, and artificial organ development-are now benefiting from AI-assisted biomaterial design, predictive modeling, and bioinformatics-driven simulations. Deep learning algorithms and computational modeling are accelerating the discovery of optimal biomaterial compositions, scaffold structures, and immune-modulating factors for personalized treatments.
AI-Driven Immunomodulation and Graft Engineering
The scope of regenerative medicine has expanded to include AI-powered immunomodulation strategies, which optimize the body's immune response to grafts, biomaterials, and engineered tissues.
Computational models can now simulate immune system interactions, helping to predict and enhance graft acceptance rates while minimizing autoimmune complications.
- AI-driven molecular simulations analyze immune pathways and predict inflammatory responses to biomaterial implants.
- Machine learning-based biomaterial screening identifies the best scaffold materials to reduce rejection risks.
- Personalized AI-driven treatment planning tailors’ regenerative therapies to individual patient immune profiles.
Computationally Enhanced Biomaterials and Smart Scaffolds
Biomaterials which are particularly inclusive towards metals, ceramics, polymers, composites-are intrinsically foreign to the human body, requiring advanced strategies to ensure biocompatibility.
AI-enhanced biomaterials integrate real-time computational feedback loops, adjusting their properties dynamically for adaptive, patient-specific applications.
- AI-assisted scaffold optimization uses predictive analytics to enhance biomaterial properties such as hydrophobicity, crosslinking density, and surface charge for improved immune tolerance.
- Deep learning-driven decellularized ECM scaffolds are now being computationally optimized to mimic native tissue environments, reducing immune rejection risks.
- Smart scaffolds embedded with biosensors enable real-time tracking of tissue regeneration and immune responses, allowing early intervention in case of graft failure.
AI and Nanotechnology for Precision Regenerative Therapies
Innovations in nanotechnology and AI-driven regenerative medicine are revolutionizing drug delivery, cellular engineering, and organ regeneration. AI-powered nanomedicine platforms enable:
- Targeted, immune-modulating drug delivery with real-time adaptation.
- AI-based bioinformatics analysis to personalize stem cell differentiation pathways.
- Predictive modeling of regenerative processes using AI-assisted multi-omics analysis.
Future Outlook: AI-Driven Personalized Regenerative Medicine
Advances in AI, computational biology, and regenerative medicine are paving the way for precision-driven, patient-specific treatments.
The integration of AI-driven predictive modeling, bioinformatics, and immune optimization is creating next-generation regenerative therapies that reduce rejection risks, improve treatment efficacy, and accelerate clinical translation. As AI continues to enhance biomaterial design, scaffold engineering, and immune modulation, the future of regenerative medicine is shifting toward fully personalized, computationally guided therapeutic solutions.
Regenerative Medicine and Tissue Engineering: A Computational Deep Dive
Regenerative medicine and tissue engineering are revolutionizing modern healthcare by integrating artificial intelligence (AI), machine learning (ML), and computational biology to repair or replace damaged or diseased tissues and organs. While regenerative medicine broadly leverages the body’s self-healing mechanisms, tissue engineering constructs functional tissues using biomaterials, stem cells, and bioinformatics-driven predictive models. Together, they represent a shift from disease management to curative solutions, offering AI-assisted, precision-driven therapeutic innovations across multiple medical disciplines.
AI-Driven Tissue Engineering and Computational Modeling
Tissue engineering has evolved beyond traditional biomaterials, incorporating AI-driven scaffold optimization, digital simulations, and bioinformatics-based material selection. The process now integrates:
- AI-assisted scaffold design, leveraging deep learning to optimize structural and biochemical properties for personalized tissue regeneration.
- Computational biology techniques to analyze cell-matrix interactions, mechanical properties, and biodegradability in engineered tissues.
- Digital twin simulations, where virtual models of engineered tissues predict regenerative outcomes before clinical application.
An example is bioengineered liver tissues on decellularized scaffolds, where AI-driven multi-omics analysis helps predict drug metabolism and regenerative potential, reducing reliance on animal models for drug testing. The FDA-approved applications of AI-assisted artificial skin and cartilage regeneration highlight how computational methods are accelerating clinical translation.
AI-Enhanced Regenerative Medicine and Personalized Healing
Regenerative medicine extends beyond biomaterials and scaffolds by integrating AI-powered precision medicine tools. This includes:
- Predictive modeling in stem cell differentiation, using AI to forecast cell fate and improve therapeutic outcomes.
- ML-based immune response simulations, optimizing biomaterial acceptance and reducing graft rejection risks.
- AI-assisted biosensors for real-time tissue monitoring, allowing early detection of biocompatibility issues in regenerative therapies.
AI-driven biosensors and organ-on-a-chip models now enable real-time assessment of cellular responses, facilitating drug toxicity evaluation and advancing personalized regenerative medicine.
Overcoming Challenges with AI and Computational Strategies
Despite rapid progress, tissue engineering and regenerative medicine face significant technical barriers, including:
- Complex Organ Vascularization: AI-guided bioprinting and angiogenesis modeling are optimizing blood vessel integration in engineered tissues.
- Immune System Adaptation: AI-driven immunomodulation simulations help predict and mitigate inflammatory responses.
- Long-Term Tissue Viability: ML-based biodegradability assessments ensure scaffold materials sustain cellular function over time.
AI-Powered Breakthroughs and Future Innovations
The field is now witnessing AI-driven innovations that are pushing regenerative medicine toward clinical mainstream adoption:
- AI-assisted Liver Tissue Engineering: Predicts hepatic cell behavior and enhances drug metabolism simulations.
- ML-optimized Cartilage Regeneration: Uses deep learning models to determine optimal biomaterial properties for joint repair.
- Computational Kidney Regeneration: AI-driven patient-specific models forecast regenerative potential in chronic kidney disease treatments.
The integration of AI-driven vascularized constructs, bioinformatics-based biomaterial selection, and computational regenerative simulations is paving the way for fully personalized, AI-assisted tissue engineering solutions.
The Future: AI-Enabled Precision Regenerative Medicine
Regenerative medicine and tissue engineering are on the verge of full-scale transformation with AI-driven innovations in bioprinting, digital twin technology, and real-time biosensors. As computational modeling, machine learning, and bioinformatics continue to refine biomedical applications, the future promises:
- Fully AI-optimized organ regeneration for clinical use.
- AI-driven tissue engineering platforms for automated biomaterial screening and scaffold design.
- Real-time AI monitoring of regenerative therapies, ensuring high precision and improved patient outcomes.
By combining biomedical engineering, AI, and computational biology, regenerative medicine is transitioning from experimental science to clinical reality, fulfilling its transformative potential in modern healthcare.
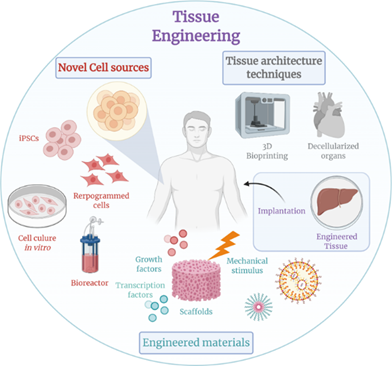
Figure 1: Recent Advancements in Tissue Engineering.
Insights: AI-Driven Organ-on-a-Chip Technology
The perspectives concerning Organ-on-a-chip (OOC) technology has significantly and drastically transformed biomedical research, computational drug discovery, precision medicine by integrating artificial intelligence (AI), microfluidics, 3D cell cultures. This micro engineered integrated accelerated platforms replicate human organ physiology at a microscale, bridging the gaps between traditional cell culture methods and in vivo studies.
With AI-assisted data analysis, OOCs are accelerating advancements in drug development, disease modeling, and personalized medicine by providing highly accurate in vitro approximations of human biological responses.
AI and Machine Learning in Organ-on-a-Chip Technologies
Recent advancements in AI and machine learning (ML) have enhanced OOCs by enabling:
- AI-driven predictive modeling to simulate cellular behavior, disease progression, and drug interactions.
- Automated real-time biosensors that track cellular responses, organ functionality, and toxicity levels.
- Computational fluid dynamics (CFD) simulations to optimize microfluidic flow conditions, ensuring physiological accuracy.
By incorporating bioinformatics-driven algorithms, AI is making OOC platforms more reliable and scalable for high-throughput drug screening.
AI-Powered Applications of Organ-on-a-Chip Systems
AI-enhanced OOCs are being developed for various biomedical applications, including:
Brain-on-a-Chip
- AI-powered neural network simulations for studying neurological diseases, synaptic activity, and blood-brain barrier interactions.
- Machine learning-based drug screening for conditions such as Alzheimer’s, Parkinson’s, and epilepsy.
Gut-on-a-Chip
- Simulates gut microbiome interactions and drug absorption using AI-driven predictive metabolic models.
- Studies inflammatory bowel diseases (IBD) and gut-liver axis metabolism.
Lung-on-a-Chip
- AI-driven modeling of alveolar-capillary dynamics for studying respiratory diseases, infections, and COVID-19 treatments.
- Predictive toxicity analysis of airborne pollutants and drug aerosols.
Heart-on-a-Chip
- AI-powered cardiac electrophysiology simulations for assessing arrhythmias and cardiotoxicity.
- Deep learning algorithms for predicting drug-induced cardiac effects in personalized medicine.
Liver-on-a-Chip
- AI-driven bioinformatics for hepatic metabolism simulations and personalized drug responses.
- Predicts drug-induced liver injury (DILI) to reduce clinical trial failures.
Kidney-on-a-Chip
- ML-powered renal function modeling for studying nephrotoxicity and kidney diseases.
- AI-assisted drug screening for chronic kidney disease (CKD) and personalized renal therapy.
Skin-on-a-Chip
- AI-assisted biophysical modeling for testing pharmaceuticals, cosmetics, and skin diseases.
- Predicts dermal drug absorption and inflammatory responses using computational deep learning models.
AI-Driven Human-on-a-Chip: Next-Generation Biomedical Simulation
An evolution of OOC technology, human-on-a-chip (HOC) integrates multiple organ systems into a single AI-enhanced microfluidic platform.
This AI-powered system is designed to:
- Simulate inter-organ metabolic pathways and systemic drug effects using predictive ML models.
- Mimic patient-specific physiological responses through real-time computational bioinformatics.
- Reduce reliance on animal testing by offering highly accurate, scalable human biology simulations.
HOC technology is revolutionizing drug safety testing, improving clinical translation rates, and accelerating therapeutic discoveries.
Challenges and AI-Powered Solutions for Future OOC Systems
Despite its promise, OOC technology faces several challenges, which AI is helping to overcome:
- Physiological Complexity: AI-driven multi-organ interaction simulations enhance predictive accuracy.
- Long-term Cell Viability: AI-powered cellular monitoring optimizes nutrient supply and waste removal.
- Scalability for Drug Testing: ML-based high-throughput screening models improve drug efficacy predictions.
- Data Integration & Standardization: AI-assisted data harmonization platforms improve OOC research reproducibility.
The Future of AI-Powered Organ-on-a-Chip Technology
As AI, deep learning, and computational biology continue to enhance OOC and HOC technologies, the future holds transformative advancements, including:
- Fully automated AI-driven OOC platforms for real-time drug testing and disease modeling.
- Digital twin technology integration, where AI creates virtual organ replicas for personalized medicine.
- Advanced AI-powered microfluidic sensors for real-time monitoring of physiological responses.
Organ-on-a-chip and human-on-a-chip systems are at the forefront of biomedical research, offering unparalleled insights into human biology.
By integrating AI, computational modeling, and bioinformatics, these technologies are poised to redefine drug discovery, precision medicine, and translational research in the coming years.
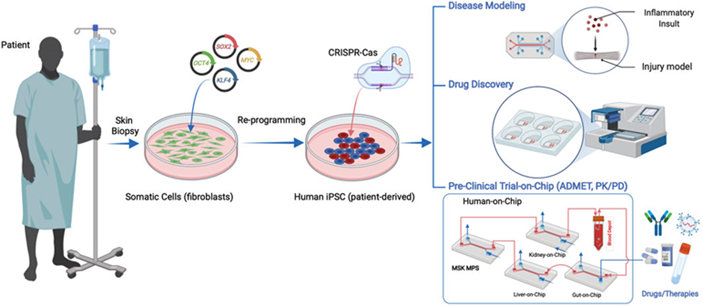
Figure 2: A Visual Representation of Organ-on-a-Chip Retrospective.
Bioprinting: AI-Driven Functional Tissues and Organs
Bioprinting, an innovative form of AI-enhanced additive manufacturing, which leverages artificial intelligence (AI), machine learning (ML), bioinformatics towards constructing complex, functional tissue structures layer by layer.
Unlike the traditional formation of 3D printing, the AI-powered bioprinting is rapidly transforming regenerative medicine, drug discovery, precision medicine, offering a more data-driven precise solutions for personalized healthcare. By mixing computational modeling, deep learning, advanced bioinformatics, bioprinting is currently redefining the future of tissue engineering and organ fabrication.
AI-Powered Core Stages of Bioprinting
Pre-Bioprinting: AI-Driven Digital Blueprinting
The bioprinting process begins with AI-assisted digital design using imaging technologies such as CT, MRI, and computational modeling tools. AI-based segmentation enhances:
- Precision in tissue architecture reconstruction.
- Predictive analysis of biomaterial-cell interactions.
- Optimization of bioink formulations using machine learning algorithms.
Bioinks, consisting of living cells and biomaterials, are computationally modeled for structural integrity and biological function, ensuring optimal cell viability before printing.
Bioprinting: AI-Assisted Layer-by-Layer Construction
During AI-driven bioprinting, machine learning models:
- Analyze extrusion parameters, adjusting nozzle temperature, pressure, and flow rates in real time.
- Optimize cell-laden bioink deposition, ensuring high-resolution tissue formation.
- Predict structural stability using AI-enhanced finite element analysis (FEA) simulations.
AI-powered bioprinting software allows researchers to dynamically adjust printing conditions, improving tissue viability and functional biomaterial integration.
Post-Bioprinting: AI-Guided Tissue Maturation
Once the structure is printed, AI-enhanced crosslinking algorithms select optimal stabilization techniques (e.g., UV light, ionic solutions, or enzymatic treatments). AI-powered cell incubation monitoring systems:
- Track cell differentiation and proliferation.
- Predict tissue growth outcomes using ML-driven bioinformatics.
- Automate bioreactor conditions for enhanced tissue maturation.
Deep learning-assisted imaging provides real-time feedback, allowing for adaptive modifications to tissue environments.
AI-Driven Applications of Bioprinting
AI in Drug Development
- AI-printed tissue models replace animal testing, enabling faster and ethically responsible drug screening.
- ML algorithms analyze pharmacokinetics and predict drug toxicity using bioprinted tissue models.
Artificial Organ Fabrication
- AI-guided bioprinting of vascularized tissues accelerates functional organ production.
- Predictive analytics optimize organ growth conditions, reducing transplant rejection rates.
AI-Enhanced Wound Healing & Regenerative Medicine
- AI-driven bioink formulation customizes treatments for burn victims, bone fractures, and reconstructive surgery.
- Deep learning-assisted skin regeneration adapts tissue structures for individual patient needs.
Future Prospects: AI and 4D Bioprinting
The next frontier in bioprinting is 4D bioprinting, where AI-driven dynamic biomaterials respond to environmental changes. Future advancements include:
AI-powered vascularization modeling, enabling full blood vessel integration into tissues. Deep learning-driven bioink development, optimizing cell viability and scaffold degradation rates. Personalized AI-guided regenerative medicine, where machine learning algorithms tailor treatments to specific genetic profiles.
With AI-driven bioprinting innovations, functional human tissues and organs are rapidly transitioning from experimental research to clinical applications, transforming precision medicine and personalized healthcare.
Case Studies Investigation Analysis: AI-Driven Drug Delivery and Immune Engineering
The oral distribution of immunotherapeutics represents a pioneering advancement within biomedical informatics, drug delivery systems (DDS), immune engineering. While the oral supervision is convenient and non-invasive, it also faces many types of critical challenges like enzymatic degradation, gastrointestinal (GI) pH variations, first-pass metabolism, mucosal-epithelial barriers. These blockades are particularly problematic for fragile biologics, including monoclonal antibodies, mRNA, DNA-based immunotherapies. However, with the integration of artificial intelligence (AI), bioinformatics, computational modeling, precision-driven biomaterial design is concurrently revolutionizing the domain of oral immunotherapy delivery, enabling towards targeted immune modulation and enhanced bioavailability.
AI-Optimized Drug Delivery Systems for Oral Immunotherapy
Advanced biomaterials and AI-driven DDS are transforming oral-to-systemic drug absorption by optimizing:
AI-enhanced drug targeting algorithms, which predict the optimal release profile and absorption site within the GI tract. Machine learning (ML)-powered nanocarrier design, improving drug encapsulation efficiency for targeted immune modulation. Computational fluid dynamics (CFD)-driven mucoadhesive modeling, optimizing biomaterial-mucosal interactions for improved retention. These AI-driven models enhance the stability and absorption of immunotherapeutics, ensuring high therapeutic efficiency with minimal systemic loss.
AI-Enhanced Biomaterials and Mucoadhesive Nanoparticles
One of the most promising AI-powered strategies for oral drug delivery involves mucoadhesive biomaterials, which:
Increase drug retention time in the GI tract by forming molecular bonds with the mucosa. Improve absorption efficiency using AI-assisted bioinformatics-driven polymer engineering. Reduce enzymatic degradation through ML-optimized drug coatings and polymeric encapsulation. Thiolated polymers are predominantly effective due to their ability to form strong disulfide bonds within the mucosal layer, enhancing drug absorption. AI-driven bioinformatics also aids in predicting and optimizing mucosal adhesion properties, ensuring a more comprehensive high-precision immunotherapy delivery for critical conditions such as ulcerative colitis and inflammatory bowel diseases (IBD).
AI-Powered Nanoparticle Drug Delivery and Bioavailability Optimization
AI-driven nanoparticle engineering has enabled the development of:
- Mucolytic agent-equipped nanoparticles, which disrupt mucus barriers, allowing deeper penetration of immunotherapeutic agents.
- Self-nanoemulsifying drug delivery systems (SNEDDS) integrated with AI-powered mucoadhesive properties, enhancing drug solubility, adhesion, and bioavailability.
- Deep learning-assisted predictive models for drug dispersion and controlled release.
These AI-optimized biomaterials are particularly valuable for targeted delivery of cytokines, growth factors, and immunomodulatory drugs within specific gut regions, enhancing immune regulation with minimal side effects.
AI-Driven Strategies for Overcoming the Epithelial Barrier
To bypass the epithelial barrier, AI-driven computational bioinformatics models optimize:
Passive and carrier-mediated diffusion simulations to predict optimal permeabilizing agents. Machine learning-enhanced prodrug design, improving drug transport efficiency. Deep learning-based antigen delivery prediction, refining molecular motor-based vaccine administration. One key breakthrough is the use of cell-penetrating peptides (CPPs), optimized through AI-driven structural modeling, allowing enhanced drug penetration into target cells.
AI-Assisted Innovations in Oral-to-Systemic Immunotherapy
The latest AI-powered biomaterial innovations include:
Chitosan-based nanoparticles engineered via bioinformatics-driven molecular docking for enhanced gut absorption. AI-driven MucoJet devices, optimizing mucosal vaccine delivery with precise, pressure-controlled drug administration. AI-powered oral-to-systemic immunotherapy simulations, predicting drug interactions with host immune responses.
Future Outlook: AI-Powered Oral Immunotherapeutics
As AI and computational biology continue to evolve, future advancements will focus on a wide range of perspectives. Deep learning-driven predictive pharmacokinetics for personalized oral immunotherapy regimens. AI-enhanced polymer biomaterials, dynamically adapting to patient-specific GI environments. Automated AI-driven drug formulation platforms, reducing R&D costs and accelerating clinical translation. By integrating AI, bioinformatics, and advanced biomaterials, oral immunotherapeutics are rapidly transitioning from concept to clinical reality. These precision-driven innovations hold transformative potential in immune engineering, offering targeted, efficient, and patient-specific treatment options for a wide range of immune-related disorders.
Gene Editing & Precision Medicine: An AI-Driven Deep Dive
Precision medicine, empowered by artificial intelligence (AI), machine learning (ML), and genome editing technologies, is revolutionizing personalized healthcare by tailoring treatments to individual patients. AI-driven multi-omics analysis, predictive modeling, and CRISPR-based gene editing are accelerating precision-driven disease management, bringing us closer to data-driven, patient-specific medical interventions. However, widespread adoption faces critical challenges related to data privacy, regulatory frameworks, cost, and ethical considerations. This analysis explores key AI-powered advancements, challenges, and solutions shaping the future of precision medicine and genome editing.
AI-Driven Genome Editing: Advancing Precision Medicine
One of the most transformative innovations in precision medicine is CRISPR-Cas9, which enables precise genetic modifications to correct disease-causing mutations. However, the success of genome editing relies on AI-driven predictive analytics, which:
Enhance target specificity, reducing off-target effects through AI-optimized guide RNA (gRNA) design. Improve CRISPR efficiency, using deep learning models to predict gene-editing outcomes. Personalize treatments, integrating multi-omics data (genomics, proteomics, transcriptomics) for patient-specific therapy optimization.AI-powered tools like DeepCRISPR, CRISPRpred, and GUIDE-Seq have significantly improved the accuracy and safety of gene editing, advancing clinical applications for rare genetic diseases, cancer immunotherapy, and regenerative medicine.
From Reactive to AI-Powered Predictive Healthcare
AI and population-scale genomics are very much shifting healthcare from reactive to proactive care. Initiatives like the Estonia’s 100,000 Genomes Project leverages ML-powered risk prediction models to precisely and preemptively diagnose diseases and adapt preventive therapies. AI-driven healthcare solutions include:
Predictive genomics, using AI algorithms to identify high-risk individuals for early intervention. AI-assisted drug repurposing, accelerating precision medicine trials for patient-specific treatments. Machine learning-enhanced polygenic risk scores (PRS), optimizing personalized preventive care. These AI-driven advances are transforming disease prediction, early intervention, and precision-targeted therapies.
AI-Powered Solutions for Data, Ethics, and Regulation Challenges
Despite its promise, precision medicine faces key challenges:
Data Privacy and AI-Driven Security
- AI-powered federated learning models enable secure, decentralized analysis of genomic data, protecting patient privacy.
- Blockchain technology enhances data integrity and secure sharing of medical records.
Ethical & Regulatory Considerations in AI-Guided Genome Editing
- AI-driven bioethics frameworks help navigate heritable genetic modifications and equity concerns.
- Harmonized global AI-driven regulatory standards streamline clinical translation of precision medicine therapies.
Cost and Accessibility of AI-Powered Personalized Medicine
- AI-based automated gene therapy manufacturing reduces costs of CRISPR and gene-editing therapies.
- ML-driven drug pricing models optimize affordability and reimbursement strategies.
AI and Computational Biology for Genomic Data Management
- AI-enhanced cloud computing optimizes storage, retrieval, and analysis of multi-omics data.
- AI-driven data harmonization frameworks facilitate cross-border genomic research collaborations.
These AI-powered innovations are addressing scalability, affordability, and ethical concerns in precision medicine adoption.
AI-Driven Patient Engagement and Public Trust in Precision Medicine
AI-powered genetic counseling tools enhance patient understanding and informed decision-making. Deep learning models improve risk communication for genome-editing therapies. Machine learning-assisted equity models ensure diverse genomic representation in research studies. Educational initiatives leveraging AI-powered health literacy programs are essential for demystifying precision medicine and increasing public trust.
Future Prospects: AI-Driven Innovations in Precision Medicine
As AI-driven precision medicine evolves, key future directions include:
- AI-optimized CRISPR-Cas gene-editing therapies for personalized, mutation-specific treatments.
- Digital twin technology, enabling AI-powered simulations of patient responses to precision therapies.
- AI-driven drug discovery pipelines, integrating predictive modeling and real-world clinical data.
By combining AI, computational biology, and multi-omics analysis, precision medicine is transitioning from experimental science to clinical reality, bringing personalized, AI-powered treatments to the forefront of healthcare.
Results and Findings
Regenerative medicine encompasses four major domains: cell transplantation, tissue engineering, drug discovery, and gene therapy. Each of these fields relies on the activity and functionality of highly active cells, with AI-powered bioinformatics and computational modeling playing a pivotal role in optimizing biomaterial-cell interactions, immune modulation, and regenerative outcomes.
AI-Driven Insights into Cell Activity and Immune Modulation
Recent research underscores the critical role of immune cells-including neutrophils and macrophages (M1/M2 phenotypes)-in tissue regeneration. AI-powered immune response modeling and deep learning-driven biomaterials screening have revealed key insights:
- Predictive modeling of macrophage polarization, optimizing pro-regenerative immune responses. Machine learning-based bio-corona analysis, improving immune evasion strategies for nanomaterials.
- Computational biomaterial-immune cell interaction studies, enhancing biocompatibility and degradation control. AI-enhanced nanomaterial design and immune system simulations are revolutionizing biomaterials-based regenerative therapies, ensuring enhanced cell activity while precisely modulating immune responses.
AI-Powered Tissue Engineering and Ethical Considerations
Tissue engineering—a cornerstone of regenerative medicine-is integrating AI-driven bioinformatics and predictive modeling to accelerate functional tissue fabrication. Key advancements include:
- Deep learning-assisted scaffold optimization, predicting cell adhesion, proliferation, and differentiation. AI-driven bioprinting and tissue fabrication, enhancing precision, scalability, and structural integrity. Computational tissue growth simulations, improving vascularization and long-term viability.
- Ethical considerations remain paramount, particularly concerning human cell sourcing and embryonic stem cell applications. AI-assisted bioethics frameworks and automated compliance monitoring systems are being developed to ensure adherence to global ethical guidelines while maintaining scientific integrity and donor rights.
AI-Enhanced Organ-on-a-Chip (OoC) Technology for Biomedical Research
OoC technology is at the forefront of AI-integrated biomedical research, enabling real-time, precision-driven disease modeling and drug testing. AI-driven computational biophysics and bioinformatics simulations are advancing:
- Deep learning-powered OoC data analysis, enabling real-time physiological monitoring and biomarker detection. Multi-organ-on-a-chip (MoC) integration, simulating systemic interactions between organs. AI-driven fluid dynamics modeling, optimizing microfluidic flow rates and nutrient exchange in OoC systems.
- State-of-the-art fabrication techniques, such as 3D printing and bioprinting, are significantly enhancing OoC efficiency. Integrated AI-powered biosensors provide continuous real-time physiological data, improving predictive analytics and translational research outcomes.
Future Prospects: AI-Driven Human-Body-on-a-Chip and Automated Precision Medicine
The long-term objective is to develop a comprehensive AI-powered human-body-on-a-chip model, which could:
- Replace traditional animal testing, accelerating drug discovery pipelines. Enhance precision medicine, using AI-driven digital twin technology to simulate patient-specific treatment responses. Enable automated AI-assisted medical procedures, optimizing regenerative therapies and clinical decision-making.
- As AI, bioinformatics, and computational modeling continue to evolve, these technologies are reshaping regenerative medicine and organ-on-a-chip research, offering groundbreaking personalized healthcare solutions and accelerating biomedical advancements.
To provide further information and retrospect understanding concerning the matters of perspectives in line with the research explorations with associative investigations the research results with their available findings which are summarized within Tables 2, 3, 4 to fully provide an overview visualization of the research findings, contextualizing advancements and their challenges towards regenerative medicine and organ-on-a-chip technologies along with their respective selections for biomaterials within research and development for regenerative medicine applications. With AI integrations and accelerated computing evolving at such a high pace only time will tell and in the future years which is still under progress and a work in progress within innovations.
Table 2: Advancements and Challenges in Regenerative Medicine and Organ-on-a-Chip Technologies 1.
| Regenerative Medicine Domain | AI-Driven Innovations | Key Applications |
| Cell Transplantation | AI-powered bioinformatics and computational modeling | Optimizing biomaterial-cell interactions and immune modulation |
| Tissue Engineering | AI-driven bioinformatics and predictive modeling | Accelerating functional tissue fabrication, optimizing scaffolds, and enhancing bioprinting precision |
| Drug Discovery | AI-integrated biomedical research, deep learning-powered data analysis | Real-time disease modeling, drug testing, and multi-organ-on-a-chip (MoC) integration |
| Gene Therapy | AI-powered bioinformatics and computational modeling | Optimizing biomaterial-cell interactions and immune modulation |
| Nanomaterials | AI-enhanced design and immune system simulations | Enhanced cell activity and precise immune response modulation |
| Organ-on-a-Chip (OoC) Technology | AI-driven computational biophysics and bioinformatics simulations | Real-time physiological monitoring, biomarker detection, and fluid dynamics modeling |
| Future Prospects | AI-powered human-body-on-a-chip, digital twin technology | Replacing animal testing, enhancing precision medicine, and automating medical procedures |
Table 3: Advancements and Challenges in Regenerative Medicine and Organ-on-a-Chip Technologies 2.
| Domain of AI Integration | Key AI-Driven Innovations | Practical Applications/Benefits |
| Oral Immunotherapy Delivery | AI-enhanced drug targeting algorithms, machine learning-powered nanocarrier design, computational fluid dynamics (CFD) modeling | Optimizes drug release and absorption, improves encapsulation efficiency, enhances biomaterial-mucosal interactions |
| Biomaterials and Nanoparticles | AI-assisted bioinformatics-driven polymer engineering, ML-optimized drug coatings, AI-driven bioinformatics | Increases drug retention time, improves absorption, reduces enzymatic degradation, optimizes mucosal adhesion properties |
| Nanoparticle Drug Delivery | AI-driven nanoparticle engineering, deep learning-assisted predictive models | Disrupts mucus barriers for deeper penetration, enhances drug solubility and bioavailability, enables controlled release of drugs |
| Epithelial Barrier Bypass | AI-driven computational bioinformatics models, ML-enhanced prodrug design, deep learning-based antigen delivery prediction | Predicts optimal permeabilizing agents, improves drug transport efficiency, refines molecular motor-based vaccine administration |
| Oral-to-Systemic Immunotherapy | Bioinformatics-driven molecular docking, AI-driven MucoJet devices, AI-powered immunotherapy simulations | Enhances gut absorption, optimizes mucosal vaccine delivery, predicts drug interactions with immune responses |
| Future Outlook | Deep learning-driven predictive pharmacokinetics, AI-enhanced polymer biomaterials, automated AI-driven drug formulation platforms | Personalizes oral immunotherapy regimens, adapts to patient-specific GI environments, reduces R&D costs |
Table 4: Selection of biomaterials within research and development for regenerative medicine applications.
| Tissues/Organs | Cell types | Types of hydrogels | Applications |
| Bone | Osteoblasts | Poly (ethylene glycol), (PEG), poly (lactic acid) (PEG-PLA) | Drug delivery, cell encapsulation, scaffold for bone regeneration |
| Heart | Bone marrow cells, embryonic stem cells, cardiomyocytes | Fibrin, PEG, alginate, hyaluronic acid (HA), superabsorbent polymer (SAP) | Scaffold for heart tissue engineering |
| Cartilage | Chondrocytes | Fibrin, PEG, SAP | Drug delivery, cell encapsulation, scaffold for cartilage regeneration |
| Eye | - | HA | Corneal transplantation |
| Skin | Fibroblast | Collagen, fibrin, HA | Abdominal wall, ear, nose and throat reconstruction, grafting |
| Blood Vessels | Stem cells, endothelial cells | PEG, alginate, HA | Vascular grafting |
Discussions and Future Directions
Looking towards the near future of tissue engineering and regenerative medicine which is rapidly evolving, driven by innovations within artificial intelligence (AI), additive manufacturing, medical imaging, biomaterials, cellular engineering. These innovations are accelerating the development towards AI-powered, patient-specific vascularized tissue constructs, offering more precise transformative solutions for complex medical conditions. However, several scientific and technological challenges remain, mainly inclusive of cell-biomaterial interactions, tissue maturation, vascularization, innervation.
AI-Driven Innovations in Tissue Engineering and Regenerative Medicine
AI-powered computational biology and bioinformatics are playing a pivotal role in optimizing biomaterials selection, scaffold design, and cellular interactions. Advanced AI-driven models are enhancing:
Deep learning-assisted biomaterial selection, predicting mechanical and biochemical properties. Machine learning-based vascularization modeling, optimizing tissue perfusion and oxygenation. AI-driven stem cell differentiation simulations, improving tissue maturation and functional integration. By integrating multi-omics data and AI-driven regenerative modeling, researchers are overcoming key challenges in tissue engineering, paving the way for clinical translation and real-world applications.
AI-Powered Oral Drug Delivery Systems for Immunotherapy
In the field of drug delivery, oral immunotherapeutics present a non-invasive alternative to injections and intravenous treatments, significantly improving patient compliance and convenience. However, biological barriers, including enzymatic degradation, mucosal layers, and epithelial permeability, continue to limit bioavailability and therapeutic efficacy. AI-driven solutions are addressing these challenges by:
Machine learning-assisted nanoparticle engineering, optimizing mucoadhesive and permeabilizing drug carriers. AI-driven drug stability modeling, enhancing oral bioavailability and controlled release. Deep learning-based predictive absorption simulations, improving targeted immune modulation. Recent advancements in AI-powered oral drug delivery systems, such as mucoadhesive and permeabilizing nanoparticles, are enhancing drug retention, absorption, and systemic availability, accelerating clinical translation for next-generation immunotherapeutics.
Future Prospects: AI-Driven Personalized Regenerative Medicine and Drug Optimization
As AI and computational biology continue to drive innovation, the future of tissue engineering and drug delivery is shifting towards precision medicine and AI-guided therapeutic interventions. Key advancements include:
AI-powered digital twin models, simulating patient-specific regenerative medicine strategies. Automated AI-driven tissue fabrication, optimizing bioprinting and scaffold design. Deep learning-assisted personalized drug formulations, customizing doses and treatment regimens for individualized immunotherapy.
With continued multidisciplinary research and AI integration, these cutting-edge technologies will enable fully personalized, highly effective regenerative and drug delivery solutions, revolutionizing biomedicine and patient care.
Conclusions
For immunotherapeutic approaches to be effectively integrated into immune engineering, it is critical that they preserve natural immune functions while remaining transient and highly efficient. Alternative delivery methods, such as sublingual and buccal administration, bypass first-pass metabolism and offer rapid systemic absorption, presenting a promising avenue for immunotherapy. Although still in the early stages of development, these non-invasive delivery routes show great potential for enhancing patient compliance and therapeutic effectiveness. Moving forward, AI-driven biomaterial design will play a pivotal role in constructing immune-targeting formulations, ensuring precise drug delivery while maintaining gastrointestinal integrity.
The investigative exploration of biologics still remains a central towards the future of pharmaceutical innovation, particularly within addressing complex diseases such as cancer, autoimmune disorders, genetic conditions. Advances towards gene-editing technologies like RNA interference (RNAi) and CRISPR-Cas9 are revolutionizing biologics-based therapies, unlocking new opportunities for precision medicine and regenerative treatments. However, small molecules are very much far from obsolete. Recent computational drug discovery breakthroughs have revived the retrospect interest in small molecules capable of modulating protein-protein interactions, expanding their therapeutic versatility. Their cell-permeability, cost-effectiveness, scalability make them indispensable for chronic disease management, where long-term affordability still remains to be a key consideration.
Ultimately, the near future of medicine will be precise by the synergistic coexistence of small molecules and biologics, offering a more diverse and complementary range of therapeutic options. This balanced approach, reinforced by AI-driven drug design and precision therapeutics, will enable the development of more effective, accessible, personalized treatments. Inclusion of these integrative accelerations for computational biology, bioinformatics, machine learning, next-generation therapies will enhance global patient care, addressing both rare and widespread diseases with greater precision and efficiency.
Supplementary Information
The various original data sources some of which are not all publicly available, because they contain various types of private information. The available platform provided data sources that support the exploration findings and information of the research investigations are referenced where appropriate.
Acknowledgments
The authors would like to acknowledge and thank the GOOGLE Deep Mind Research with its associated pre-prints access platforms. This research exploration was investigated under the platform provided by GOOGLE Deep Mind which is under the support of the GOOGLE Research and the GOOGLE Research Publications within the GOOGLE Gemini platform. Using their provided platform of datasets and database associated files with digital software layouts consisting of free web access to a large collection of recorded models that are found within research access and its related open-source software distributions which is the implementation for the proposed research exploration that was undergone and set in motion. There are many data sources some of which are resourced and retrieved from a wide variety of GOOGLE service domains as well. All the data sources which have been included and retrieved for this research are identified, mentioned and referenced where appropriate. ChatGPT was used in order to check for grammatical errors and spellings.
References
- Akhtar, Z. B. (2025). Artificial intelligence within medical diagnostics: A multi-disease perspective. Artificial Intelligence in Health, 5173.
Publisher | Google Scholor - Zhong, Z., Yang, S., Chen, F., Du, Z. H., Liu, N., et al. (2025). Molecular engineering strategies of cationic oligo (p-phenyleneethynylene) s for enhancing sensitive discrimination of multiple hazardous explosives. Chemical Engineering Journal, 510:161558.
Publisher | Google Scholor - Akhtar, Z. B. (2024). The design approach of an artificial intelligent (AI) medical system based on electronical health records (EHR) and priority segmentations. The Journal of Engineering, 4:e12381.
Publisher | Google Scholor - Zhan, J., Wang, S., Li, X., Zhang, J. (2025). Molecular engineering of functional DNA molecules toward point-of-care diagnostic devices. Chemical Communications.
Publisher | Google Scholor - Rani, A., Zafar, F., Hussain, R., Zafar, W. U. I., Khanum, A., et al. (2024). Molecular engineering of benzothiadiazole core based non-fullerene acceptors to tune the optoelectronic properties of perovskite solar cells. Computational and Theoretical Chemistry, 1237:114637.
Publisher | Google Scholor - Lin, N., Tsuji, M., Bruzzese, I., Chen, A., Vrionides, M., et al. (2025). Molecular engineering of emissive molecular qubits based on spin-correlated radical pairs. Journal of the American Chemical Society, 147(13):11062-11071.
Publisher | Google Scholor - Cheng, Y., Wang, T., Zhu, H., Hu, X., Mi, J., et al. (2025). Molecular Engineering of Amino Acid Crystals with Enhanced Piezoelectric Performance for Biodegradable Sensors. Angewandte Chemie, 137(15):e202500334.
Publisher | Google Scholor - Liu, Y., Song, Y., Zhu, Z. H., Ji, C., Li, J., et al. (2025). Twisted‐Planar Molecular Engineering with Sonication‐Induced J-Aggregation to Design Near‐Infrared J-Aggregates for Enhanced Phototherapy. Angewandte Chemie, 137(7):e202419428.
Publisher | Google Scholor - Sun, W., Xue, B., Fan, Q., Tao, R., Wang, C., et al. (2020). Molecular engineering of metal coordination interactions for strong, tough, and fast-recovery hydrogels. Science Advances, 6(16):eaaz9531.
Publisher | Google Scholor - Lv, G., Zhu, J. (2025). Intrinsic Thermal Conductivity of Molecular Engineered Polymer. Advanced Functional Materials, 2420708.
Publisher | Google Scholor - Wan, Q., Zhang, R., Zhuang, Z., Li, Y., Huang, Y., et al. (2020). Molecular engineering to boost AIE‐active free radical photogenerators and enable high-performance photodynamic therapy under hypoxia. Advanced Functional Materials, 30(39):2002057.
Publisher | Google Scholor - Yang, R., Ai, D., Fan, S., Zhang, W., Yang, X., et al. (2025). Aramid dielectric co-polymer: from molecular engineering to roll-to-roll scalability for high-temperature capacitive energy storage. Energy & Environmental Science, 18(8):3718-3729.
Publisher | Google Scholor - Huang, Y., Liu, T., Huang, Q., Wang, Y. (2024). From organ-on-a-chip to human-on-a-chip: A review of research progress and latest applications. ACS Sensors, 9(7):3466-3488.
Publisher | Google Scholor - Sun, S. (2024). Research of Organ-on-a-chip and its application. In E3S Web of Conferences (Vol. 553, p. 05012). EDP Sciences.
Publisher | Google Scholor - Sadeghzade, S., Hooshiar, M. H., Akbari, H., Tajer, M. H. M., Sahneh, K. K., et al. (2024). Recent advances in Organ-on-a-Chip models: How precision engineering integrates cutting edge technologies in fabrication and characterization. Applied Materials Today, 38:102231.
Publisher | Google Scholor - Zhao, Y., Landau, S., Okhovatian, S., Liu, C., Lu, R. X. Z., et al. (2024). Integrating organoids and organ-on-a-chip devices. Nature Reviews Bioengineering, 2(7):588-608.
Publisher | Google Scholor - He, C., Lu, F., Liu, Y., Lei, Y., Wang, X., et al. (2024). Emergent trends in organ-on-a-chip applications for investigating metastasis within tumor microenvironment: A comprehensive bibliometric analysis. Heliyon, 10(1).
Publisher | Google Scholor - Song, S. H., Jeong, S. (2024). State-of-the-art in high throughput organ-on-chip for biotechnology and pharmaceuticals. Clinical and Experimental Reproductive Medicine, 52(2):114.
Publisher | Google Scholor - Sean, G., Banes, A. J., Gangaraju, R. (2024). Organoids and tissue/organ chips. Stem Cell Research & Therapy, 15(1):241.
Publisher | Google Scholor - Li, X., Zhu, H., Gu, B., Yao, C., Gu, Y., et al. (2024). Advancing intelligent organ‐on‐a‐chip systems with comprehensive in situ bioanalysis. Advanced Materials, 36(18):2305268.
Publisher | Google Scholor - Nasiri, R., Zhu, Y., de Barros, N. R. (2024). Microfluidics and organ-on-a-chip for disease modeling and drug screening. Biosensors, 14(2):86.
Publisher | Google Scholor - Gunasekaran, B. M., Srinivasan, S., Ezhilan, M., Rajagopal, V., Nesakumar, N. (2024). Advancements in Organ‐on‐a‐Chip Systems: Materials, Characterization, and Applications. ChemistrySelect, 9(40):e202403611.
Publisher | Google Scholor - Huang, X., Lou, Y., Duan, Y., Liu, H., Tian, J., et al. (2024). Biomaterial scaffolds in maxillofacial bone tissue engineering: a review of recent advances. Bioactive Materials, 33:129-156.
Publisher | Google Scholor - Akhtar, Z. B., Rozario, V. S. (2020). The design approach of an artificial human brain in digitized formulation based on machine learning and neural mapping. In 2020 International Conference for Emerging Technology (INCET) (pp. 1-7). IEEE.
Publisher | Google Scholor - Bagherpour, R., Bagherpour, G., Mohammadi, P. (2025). Application of artificial intelligence in tissue engineering. Tissue Engineering Part B: Reviews, 31(1):31-43.
Publisher | Google Scholor - Bauso, L. V., La Fauci, V., Longo, C., Calabrese, G. (2024). Bone tissue engineering and nanotechnology: a promising combination for bone regeneration. Biology, 13(4):237.
Publisher | Google Scholor - Angolkar, M., Paramshetti, S., Gahtani, R. M., Al Shahrani, M., Hani, U., et al. (2024). Pioneering a paradigm shift in tissue engineering and regeneration with polysaccharides and proteins-based scaffolds: a comprehensive review. International Journal of Biological Macromolecules, 265:130643.
Publisher | Google Scholor - Akhtar, Z. B., Rozario, V. S. (2025). AI perspectives within computational neuroscience: EEG integrations and the human brain. In Artificial Intelligence and Applications. 3(2):145-160.
Publisher | Google Scholor - Chen, A., Wang, W., Mao, Z., He, Y., Chen, S., et al. (2024). Multimaterial 3D and 4D bioprinting of heterogenous constructs for tissue engineering. Advanced Materials, 36(34):2307686.
Publisher | Google Scholor - Gou, Y., Huang, Y., Luo, W., Li, Y., Zhao, P., et al. (2024). Adipose-derived mesenchymal stem cells (MSCs) are a superior cell source for bone tissue engineering. Bioactive Materials, 34:51-63.
Publisher | Google Scholor - Xu, C., Liu, Z., Chen, X., Gao, Y., Wang, W., et al. (2024). Bone tissue engineering scaffold materials: Fundamentals, advances, and challenges. Chinese Chemical Letters, 35(2):109197.
Publisher | Google Scholor - Jafari, A., Vahid Niknezhad, S., Kaviani, M., Saleh, W., Wong, N., et al. (2024). Formulation and evaluation of PVA/gelatin/Carrageenan inks for 3D printing and development of tissue-engineered heart valves. Advanced Functional Materials, 34(7):2305188.
Publisher | Google Scholor - Shan, B. H., Wu, F. G. (2024). Hydrogel-based growth factor delivery platforms: strategies and recent advances. Advanced Materials, 36(5):2210707.
Publisher | Google Scholor