Case Report
Prevalence of Oncogenic Human Papillomavirus (HPV 16 And 18) Infection and Its Implications for Screening and Vaccination Protocols in Ethiopia: A Systematic Review and Meta-Analysis
1 Amhara Public Health Institute, Ethiopia.
2 Health Biotechnology Division, Institute of Biotechnology, Bahir Dar University, Ethiopia.
*Corresponding Author: Alemayehu Abate, Amhara Public Health Institute, Ethiopia.
Citation: Abate A., Tadesse S., Munshea A., Nibret E. (2025). Prevalence of Oncogenic Human Papillomavirus (HPV 16 And 18) Infection and Its Implications for Screening and Vaccination Protocols in Ethiopia: A Systematic Review and Meta-Analysis, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 8(3):1-10. DOI: 10.59657/2837-4681.brs.25.187
Copyright: © 2025 Alemayehu Abate, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: June 17, 2025 | Accepted: July 01, 2025 | Published: July 08, 2025
Abstract
Background: Cervical cancer (CC) arises most frequently from persistent infection with oncogenic high risk (HR-) human papillomavirus (HPV) types. The objective of this systematic review and meta-analysis was to estimate the combined prevalence of HPV type 16 and type 18 and the genotypes of high-risk Human Papillomavirus (HR-HPV) distribution in Ethiopia.
Methods: We searched electronic databases (MEDLINE/PubMed, Google Scholar, EMBASE, CINAHL, and Cochrane Central library) to retrieve primary studies published in English language and published in the years between 2014 and 2023. The appraisal of retrieved articles followed the PRISMA guidelines. Meta-analysis was done using STATA 15 with metan package. A pooled estimate of the combined proportions of genotypes 16 and 18 was analyzed with a random effect model. The presence of heterogeneity across studies was assessed using inverse of variance (I2). Study characteristics such as list of authors, year of publication, study region, study area, study design, and sample size were also extracted. All included articles used Polymerase chain reaction (PCR), Onco E6 protein test, and genotyping to determine the genotype distribution of HPV. The sample size of each included article varied from 83 to 1020.
Results: The findings of the present systematic review and meta-analysis revealed that a total of 18 HR-HPV types were identified from the nine included articles. The top five identified genotypes are HR-HPV types 16, 58, 52, 31, and 35. The pooled proportion of HR-HPV indicated that HR-HPV type 16 (38.9%) was the highest prevalent of all types. HPV type 18 was not in the top five HR-HPV infection, but its pooled prevalence was 5.32%. The combined pooled prevalence of both HPV types 16 and 18 was 44.22%.
Conclusion: The results of this review demonstrated that HR-HPV type 16, HR-HPV type 58, HR-HPV type 52, HR-HPV type 31, and 1HR-HPV type 35 are relatively highly prevalent in Ethiopia. The screening laboratory methods in Ethiopia should consider these highly prevalent genotypes and others. The vaccination program in Ethiopia should consider nano-valent HPV vaccine which has non-infectious protein antigens for HPV 6, 11, 16, 18, 31, 33, 45, 52 and 58.
Keywords: human papillomavirus; genotype; women; cervical cancer; meta-analysis; ethiopia
Introduction: Background
Oncogenic high risk (HR) human papillomavirus (HPV) infections are the most common cause of cervical cancer (CC) [1]. About 8000 base pairs make up the genome of the non-enveloped, double-stranded DNA virus known as the human papilloma virus (HPV), which belongs to the Papillomaviridae family. In both men and women, the virus has been found to be the causative agent of cervical and other anogenital cancers [2]. High-risk or carcinogenic to humans include the genotypes 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68. It has been proposed that other genotypes, such as 26, 53, 69, 73, and 82, are likely high-risk. The benign hyper-proliferative epithelial lesions are caused by the low-risk HPVs, HPV-42, -43, -6/11, and -44 [3].
In sub-Saharan African nations, the combined prevalence of HR-HPV is 32.3%. Geographical location also affected the HR-HPV genotype distribution. There is a distinct trend in the distribution among African nations. The most common genotypes found in sub-Saharan African nations were HPV-16, -18, -45, -35, and -33, whereas HPV-16, -52, -18, -39, and -31 are found in large numbers in eastern Africa. The distribution of HR-HPV, namely HPV-16, -52, -18, -58, and -45, in Ethiopia is nearly identical to that of the other East African nations [4].
On Nov 17, 2020, WHO launched a global initiative to accelerate the elimination of cervical cancer, and set up a historical milestone—through the adoption of a resolution by 194 countries at the World Health Assembly—that pledged, for the first time, to eliminate a malignant disease by pursuing three important steps: vaccination, screening, and treatment. Expanding the coverage of HPV vaccines is an essential part of the 90–70-90 target proposed by the World Health Organization (WHO) for eliminating cervical cancer [5].
Currently, three preventative HPV vaccines that target high-risk HPV varieties are marketed and available in numerous nations across the world to help prevent diseases linked to HPV. Bivalent, quadrivalent, and nanovalent HPV vaccines are among them. HPV 16 and 18 non-infectious protein antigens are present in the bivalent vaccine, HPV 6, 11, 16, and 18 non-infectious protein antigens are present in the quadrivalent vaccine, and HPV 6, 11, 16, 18, 31, 33, 45, 52, and 58 non-infectious protein antigens are present in the nanovalent vaccine [6].
Screening and vaccination program in Ethiopia focuses on only the HPV 16 and HPV 18 genotypes even though there are other highly prevalent HPV types. Considering of the distribution of high-risk HPV genotypes present in Ethiopia, HPV distribution should be central for vaccination, screening and managements of cervical cancer. The objective of this systematic review and meta-analysis study was to review the proportion of HPV type 16 and type 18, the distribution of high-risk Human Papillomavirus (HR-HPV) in Ethiopia. The results will help to fill the current information gaps regarding the proportion of HR-HPV type 16 and 18, and the prevalence and genotype distribution of HR-HPV infection in Ethiopia.
Methods
Study design and setting
The review protocol was prepared according to a statement recommendation made by the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) in 2015 [7]. Institution and population-based studies done on the distribution of human papilloma virus among women in Ethiopian from 2014 to 2023 were used for this systematic review and meta-analysis. Ethiopia is one of the east African countries situated in the horn of Africa having a total population of 123,408,643, with 62,465,261 persons between 15 and 64 years old (30,576,750 males / 31,889,734 females) [8].
Sources of Studies and Searching Strategies
We conducted a systematic review and meta-analysis of published articles to estimate the proportion of HPV types 16 and 18 in Ethiopia. Electronic databases (MEDLINE/PubMed, Google Scholar, EMBASE, CINAHL and Cochrane Central library) were systematically searched. Research articles written in English language and published in peer-reviewed journals were accessed from the above databases by using the following key words or terms/mesh terms either singly or in combination: distribution/prevalence, high risk Human Pappiloma Virus/HR-HPV, genotypes, cervical cancer, cervical neoplasia, Ethiopia. ((((Distribution [tw] OR Prevalence [tw]) OR ((High [All Fields] AND ("risk"[MeSH Terms] OR "risk"[All Fields]) AND Human Pappilomavirus[tw]) OR HR-HPV [tw])) OR (Genotypes[tw] OR ("Genotype"[Mesh] OR "Genetic Profile"[Mesh]))) was the query used to search records.
Eligibility Criteria
Eligible research articles for this systematic review and meta-analysis study were selected in three stages: title alone, abstracts, and then full-text articles. Inclusion and exclusion criteria were defined before the articles were identified. Publications eligible for inclusion were those studies that reported the prevalence and distribution of HR-HPV in Ethiopia. The criteria used to include the articles in this analysis were if the full article and its related data are accessible and if the HR-HPV prevalence is calculated and at least two genotypes identified. Articles with non-English language publications and published without original data were excluded. Based on the PRISMA procedure, duplicated articles were excluded and the titles and abstracts of the remaining papers were screened sequentially for inclusion in full-text evaluation.
Selection Process of Studies
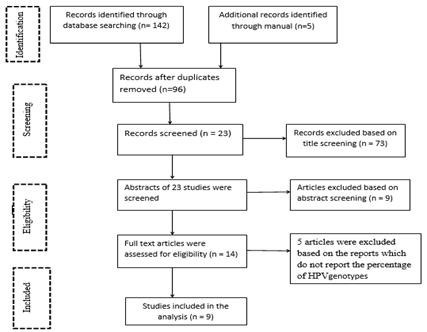
A total of 142 articles were identified through searching the electronic international databases and other sources. Furthermore, five relevant articles were selected by a manual search and included as identified articles. A total of 51 duplicates were excluded, and then 96 articles were reviewed by title, which led to the elimination of 73 articles. Twenty-three articles were screened by title and abstract and nine articles were excluded based on abstract screening. The remaining 14 articles were checked for eligibility by the full-text review. After the full-text screening, five articles were excluded based on the inclusion/exclusion criteria. Finally, nine articles were included in this systematic review and meta-analysis. Figure 1 shows the process of literature retrieval and screening using a flow diagram.
Figure 1: The PRISMA flow diagram of literature retrieval and screening.
Data Extraction and Recording
After collecting findings from all databases, the data from the included articles were exported to a Microsoft Excel spreadsheet. All included articles were extracted, reviewed, and screened by reviewers. Data from eligible abstract and/or full text of the articles were extracted by considering the outcome variables (i.e., prevalence or proportion of HPV genotypes, magnitude of cancer-causing HPVs or high risk (HR) HPV genotypes and low-risk HPV genotypes). Study characteristics such as list of authors, year of publication, study duration, study region, study area, study design, sample size was also extracted
Critical Appraisal of Studies
The validity and methodological quality of all included studies were assessed using Joanna Briggs Institute Critical Appraisal Checklist for systematic review [9]. The tool consists of eight criteria that were checked as ‘yes’, ‘no’, ‘unclear’ or ‘not applicable’. After carefully evaluating the included articles against each criterion, the studies were finally classified in to three groups; a studythat fulfilled > 80% of the criteria was considered as ‘good quality’. Similarly, a study that scored 50–80 and < 50> 80 and judged methodologically good.
Risk of Bias (Quality) Assessment
The risk of bias was assessed using the checklist of Meta-analysis Of Observational Studies in Epidemiology (MOOSE). Moreover, the completeness of outcome data and other sources of bias were effectively assessed by the review authors using Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) detailed guidance to make domain-level judgments about the risk of bias [10].
Strategy for Data Synthesis
The data were entered using a Microsoft Excel spreadsheet. The extracted data from the articles include name of the first author, publication year, region, study area, study setting and study design, sample size, prevalence of HR-HPV, genotype identified, methodology used for laboratory diagnosis and study participants. The proportion of HR-HPV type 16 and type 18, the top five genotypes from each study and pooled combined prevalence of HR-HPV type 16 and type 18 were analyzed using statistical software for data science (STATA V15). The data were extracted from the included articles using the excel data extraction sheet developed by the first author.
Results
Search results
From the electronic international databases and other sources, a total of 147 articles were retrieved and sequentially screened for inclusion in the analysis as depicted in PRISMA flow chart (Fig. 1). Nine articles met our inclusion criteria and were included in this systematic review and meta-analysis study.
Study Description of the included studies
The Description of the included studies is summarized in Table 1. All included studies were published in the period of 2014 and 2023. All articles used institution and population-based study design. Studies were undertaken from five different regions of the Ethiopia. All included articles used Polymerase chain reaction (PCR), Onco E6 protein test, and genotyping to determine the genotype distribution of HPV. The sample size in each included article varied from 83 [11] to 1020 [12]. All included articles used similar study participants, have similar interventions, and similar outcomes.
Table 1: Study Description of the articles that are included in this systematic review and meta-analysis study.
| Authors and Year of publication | Region | Study area | Study setting and Study design | Sample size | Tested | Prevalence of HPV | Genotype Identified | Methodology | Study participants |
| Bogale et al., 2022 | Addis Ababa | Addis Ababa | Hospital, cross sectional | 578 | 558 | 23.30% | 14 | Abbott Multiplex real-time PCR | HIV posetive women |
| Seyoum et al., 2023 | Dire Daw | Dire Daw | Hospital, cross sectional | 901 | 832 | 13.10% | 14 | Real-time multiplex assay, Anyplex™II HPV HR kit | sexually active women |
| TEKA et al., 2020 | South | Butajira | population-based, cross sectional | 1020 | 764 | 23.20% | 27 | multiplexed genotyping (MPG) by BSGP5+/6+ PCR with Luminex read out. | rural women |
| Temesgen et al., 2021 [19] | Amhara | Amhara | Hospital, cross-sectional | 337 | 337 | 7.10% | 2 | OncoE6TM Cervical Test | women aged 21 to 49 years of age |
| Haile et al., 2019 | Oromia | Adama | Hospital, cross-sectional | 83 | 66 | 22.70% | 12 | RIOTOL quantitative polymerase chain reaction (qPCR) HPV genotyping test | women aged 21 to 49 years of age |
| Ali et al., 2019 | Addis Ababa | Addis Ababa | Hospital, cross-sectional | 366 | 366 | 13.70% | 14 | Abbott Real-Time PCR system | Women age 18-68 |
| Derbie et al., 2023 | Amhara | Bahir dar | cross-sectional | 355 | 355 | 53% | 13 | Abbott Alinity m system | Women age >30 |
| Leyh-Bannurah et al. | South | Attat, Gurage | cross-sectional | 537 | 537 | 17.50% | 14 | Genotyping Kit HPV GP and genotyped with a GP5+/6 + - and SPF10-primer based system | women 15 to 64 years |
Statistical analysis
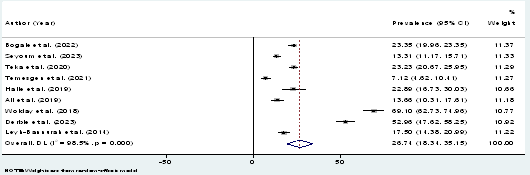
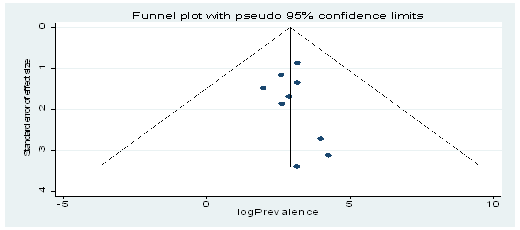
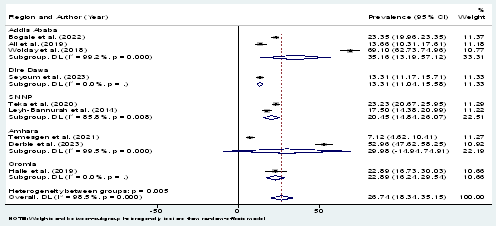
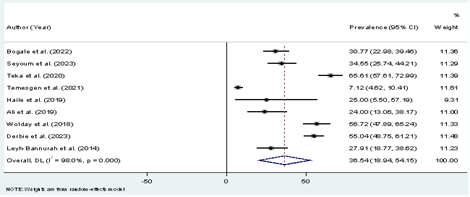
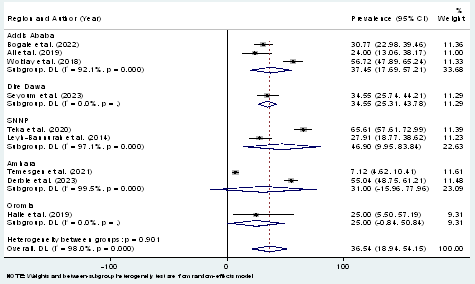
The statistical pooled prevalence of HPV was performed according to the random-effects model using a statistical software for data science (STATA V15) with its 95% Confidence Interval (CI) assuming the true effect size varies between studies using the random-effects inverse-variance model with DerSimonian-Laird estimate of tau². The heterogeneity of the studies was evaluated based on the Cochrane Q and I2 tests. Egger's test was conducted for small-study effects. Test of H0: no small-study effects with P = 0.559 , since p>0.05, we accept null hypothesis. That means there is no publication bias (similar to funnel plot (symmetrically). The forest plots were employed to present the pooled prevalence of HR-HPV infection and genotypes. The pooled prevalence of HPV was 27%. Subgroup analysis was conducted based on the study region. Accordingly, Adiss Abeba and Amhara regions have the highest prevalence of HPV with 35.16% and 29.98% respectively (figure.4).
Figure 2: Forest plot showing the pooled proportion of HR-HPV in Ethiopia 2023.
Figure 3: Funnel plot of standard error by effect size
Figure 4: Forest plot of Sub-group analysis by region
HR-HPV Genotype Distribution
A total of 18 HR-HPV genotype distributions were identified from all included studies. These HR-HPV genotypes include HPV 16, HPV 18, HPV 26, HPV 31, HPV 33, HPV 35, HPV 39, HPV 45, HPV 51, HPV 52, HPV 53, HPV 56, HPV 58, HPV 59 and HPV 66, HPV 68, HPV 73 and HPV 82. The details of percentage distribution of HR-HPV are stated in Table 2.
Table 2: Percentage distribution of HR-HPV infection.
| Authors | List of Genotypes | Percentage distribution of HR-HPV | Study Area |
| Bogale et al. [13] | HPV-16 | 26.20% | AddissAbeba |
| HPV-18 | 4.60% | ||
| 0thers (31,33,35,39,45,51,52,56,58,59,66,68) | 69.20% | ||
| Seyoum et al. [14] | HPV-16 | 31.80% | Dire Dawa |
| HPV-18 | 2.70% | ||
| HPV-31 | 19.10% | ||
| HPV-33 | 2.70% | ||
| HPV-35 | 10% | ||
| HPV-39 | 7.27% | ||
| HPV-45 | 6.36% | ||
| HPV-51 | 6.36% | ||
| HPV-52 | 11.80% | ||
| HPV-56 | 7.27% | ||
| HPV-58 | 10.90% | ||
| HPV-59 | 6.36% | ||
| HPV-66 | 7.27% | ||
| HPV-68 | 8.18% | ||
| TEKA et al. [12] | HPV-16 | 57.10% | Butajera |
| HPV-18 | 9% | ||
| HPV-26 | 3.60% | ||
| HPV-31 | 14.10% | ||
| HPV-33 | 1.04% | ||
| HPV-35 | 20.40% | ||
| HPV-39 | 2.10% | ||
| HPV-45 | 9.60% | ||
| HPV-51 | 4.20% | ||
| HPV-52 | 15.80% | ||
| HPV-53 | 3.10% | ||
| HPV-56 | 3.10% | ||
| HPV-59 | 1.04% | ||
| HPV-66 | 2.60% | ||
| HPV-68 | 1.04% | ||
| HPV-73 | 0.50% | ||
| HPV-82 | 2.60% | ||
| HPV-58 | 1.50% | ||
| Haile et al. [11] | HPV-16 | 16.70% | Adama |
| HPV-18 | 8.30% | ||
| HPV-31 | 16.70% | ||
| HPV-45 | 16.70% | ||
| HPV-51 | 41.70% | ||
| HPV-58 | 16.70% | ||
| HPV-68 | 25% | ||
| HPV-6 | 16.70% | ||
| HPV-67 | 16.70% | ||
| Ali et al. [15] | HPV-16 | 20% | AddissAbeba |
| HPV-18 | 4% | ||
| Others (31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68 | 76% | ||
| Wolday et al. [16] | HPV-16 | 53% | AddissAbeba |
| HPV-18 | 3.70% | ||
| HPV-31 | 5.20% | ||
| HPV-35 | 7.50% | ||
| HPV-39 | 2.20% | ||
| HPV-45 | 7.50% | ||
| HPV-52 | 1.50% | ||
| HPV-56 | 4.50% | ||
| HPV-58 | 3% | ||
| HPV-59 | 3.70% | ||
| HPV-68 | 0.70% | ||
| HPV-53 | 1.50% | ||
| HPV-66 | 0.70% | ||
| HPV-70 | 3% | ||
| HPV-73 | 2.20% | ||
| Derbie et al. [17] | HPV-16 | 50.40% | Bahir Dar |
| HPV-18 | 4.70% | ||
| HPV-45 | 3.50% | ||
| HPV-35 | 3.10% | ||
| HPV-56 | 1.60% | ||
| HPV-33 | 8.50% | ||
| HPV-59 | 3.90% | ||
| HPV-68 | 5.80% | ||
| HPV-66 | 0.80% | ||
| HPV-52 | 1.90% | ||
| HPV-39 | 5.80% | ||
| HPV-58 | 0.40% | ||
| HPV-31 | 9.70% | ||
| Leyh-Bannurah et al. [18] | HPV-16 | 24.40% | Gurage |
| HPV-18 | 3.50% | ||
| HPV-45 | 4.70% | ||
| HPV-35 | 5.80% | ||
| HPV-56 | 10.50% | ||
| HPV-33 | 2.50% | ||
| HPV-59 | 1.20% | ||
| HPV-68 | 4.70% | ||
| HPV-66 | 2.30% | ||
| HPV-52 | 11.60% | ||
| HPV-39 | 5.80% | ||
| HPV-31 | 10.50% | ||
| HPV-51 | 7% |
Proportion of HPV types 16 and 18
HPV genotypes 16 and 18 were reported by all studies conducted in Ethiopia. The proportion varies from 24% to 65.7%. The highest proportion of HPV 16 and 18 was observed in a study done in Butajera, SNNP [12], whereas the lowest proportion of HPV 16 and 18 was observed in a study done in Addiss Abeba region [14] (Table 3). Sub-group analysis result shows the combined proportion of types 16 and 18 was 46.9%, 37.45%, and 31% in SNNP, Adiss Abeba and Amhara region respectively. One study from Amhara region did not differentiate the proportion of type 16 and 18.
Table 3: Proportion of HPV types 16 and 18 among studies done in Ethiopia.
| Authors | Study Area | HPV type | Percentage | Combined HPV 16 and 18 |
| Bogale et al. | AddissAbeba | HPV-16 | 26.20% | 30.87% |
| HPV-18 | 4.60% | |||
| Seyoum et al. | Dire Dawa | HPV-16 | 31.80% | 34.5% |
| HPV-18 | 2.70% | |||
| Leyh-Bannurah et al. | Gurage | HPV-16 | 24.40% | 27.9% |
| HPV-18 | 3.50% | |||
| TEKA et al. | Butajera | HPV16 | 57.1% | 65.7% |
| HPV-18 | 9% | |||
| Haile et al. | Adama | HPV-16 | 16.7% | 25% |
| HPV-18 | 8.30% | |||
| Ali et al. | AddissAbeba | HPV-16 | 20% | 24% |
| HPV-18 | 4% | |||
| Wolday et al. | AddissAbeba | HPV-16 | 530% | 56.7% |
| HPV-18 | 3.7% | |||
| Derbie et al. | Bahir Dar | HPV-16 | 50.40% | 55.1% |
| HPV-18 | 4.70% |
Figure 5: Forest plot showing a combined prevalence of HPV-16 and-18 genotypes.
Figure 6: Forest plot showing the combined prevalence of HPV-16 and 18 genotypes across regional states of Ethiopia
Top five HR-HPV infections and pooled prevalence
The top five identified genotypes are HR-HPV types 16, 58, 52, 31 and 35. The pooled prevalence of HR-HPV shows HPV-16 covers the highest percentage (38.9%), and HPV-58 (13.8%), HPV-52 (13.1%), HPV-31 (12.55%) and HPV-35 (9.2%) covers the second, third, fourth and fifth highest percentage respectively (Table 4).
Table 4: Top five identified HR-HPV infections.
| Seyoum et al. | Leyh-Bannurah et al. | TEKA et al. | Haile et al. | Wolday et al. | Derbie et al. | Pooled Prevalence | |||||||
| HPV type | % | HPV type | % | HPV type | % | HPV type | % | HPV type | % | HPV type | % | HPV type | % |
| 16 | 31.8% | 16 | 24.4% | 16 | 57.1% | 51 | 41.7% | 16 | 53% | 16 | 50.4% | 16 | 38.9% |
| 31 | 19.1% | 52 | 11.6% | 35 | 20% | 68 | 25% | 35 | 7.5% | 31 | 9.7% | 31 | 12.55% |
| 52 | 11.8% | 31 | 10.5% | 52 | 15.8% | 16 | 16.7% | 45 | 7.5% | 33 | 8.5% | 52 | 13.1% |
| 58 | 10.9% | 56 | 10.5% | 31 | 14.1% | 31 | 16.7% | 31 | 5.2% | 39 | 5.8% | 58 | 13.8% |
| 35 | 10% | 51 | 7% | 45 | 9.6% | 58 | 16.7% | 56 | 4.5% | 68 | 5.8% | 35 | 9.2% |
Discussion
Human papillomavirus (HPV) is the most common sexually transmitted virus. Cervical persistent infection with any of the high-risk HPV genotypes, such as HPV 16,18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68, is associated with the development of cervical intraepithelial lesions and invasive cervical cancer [20]. The current systematic review and Meta-analysis was conducted to combine the findings of primary studies to generate better estimated evidences on the prevalence of HR-HPV and to identify the proportion of HR-HPV types 16 and 18 in Ethiopia. To conduct this study, a total of nine eligible studies were included. It was performed based on the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). The highest prevalence of HR-HPV was HR-HPV type 16 (57.1%) in a study done in Butajira, SNNP region, whereas the lowest highest prevalence of HR-HPV was HR-HPV type 73 (0.5%) was also observed in a study conducted in Butajera. HPV types 16 and 18 were found in all included studies.
In our review, the pooled proportion of HR-HPV indicates that HR-HPV type 16 (38.96%) was the highest prevalence of all types. HPV type 18 was not in the top five HR-HPV infections but its pooled prevalence was 5.32%. The combined pooled prevalence of both HPV types 16 and 18 was 44.22%. Findings of this systematic review and Meta-analysis have similar findings with the systematic review done in east Africa which revealed that HPV 16 (58.4%), HPV 18 (9.9%), HPV 45 (8.7%), HPV 52 (8%) and HPV 56 (7.3%) are the top five HRHPV causing cervical cancer in Ethiopia [21]. The findings of this study will have important implications for the selection of HPV screening, diagnostic methods, good managements of cervical cancer, and for an effective vaccination program. The current systematic review and Meta-analysis identify the proportion of vaccine targeted genotypes which are type 16 and type 18 is less than 50%, which indicates the screening methods and vaccination program is effective only to less than 50% of the infection caused by HR-HPV. This result is in agreement with other study done in Ethiopia which states that the current HPV vaccines confer only type-specific (HPV-16 and -18) immunity, leaving a small proportion of Ethiopian women unprotected against other HR-HPV genotypes such as 52, 56, 31 and 58. Thus, future HPV vaccines such as the nanovalent vaccine may be more useful to Ethiopia as they will protect women against more genotypes [22].
Conclusion
The results of this review demonstrated a proportion of HR-HPV type 16 and type 18 in Ethiopia. The combined pooled proportion of these two genotypes was 44.22%. The top five genotypes which circulate in Ethiopia are HPV-16, HPV-58, HPV-52, HPV-31, and HPV-35. HPV- 18 was not indicated in the top five prevalent genotypes. HR-HPV type 16, 31, 52, 58, and 35 together covers 87.55% of the HPV infection; this indicates diagnostic modalities which can detect the top five genotypes and providing nano-valent vaccine can prevent 87.55% of HR-HPV infection and cervical cancer. The current screening methods and vaccine given are targeted only for HPV-16 and HPV-18 genotypes in Ethiopia. The screening, diagnostic, and vaccine program in Ethiopia should consider these high prevalent genotypes.
Future Directions
The screening and diagnostic laboratory methods in Ethiopia should consider highly prevalent genotypes including Genotypes 16, 58, 52, 31, and 35. The vaccination program in Ethiopia should consider nano-valent HPV vaccine which has non-infectious protein antigens for HPV 6, 11, 16, 18, 31, 33, 45, 52 and 58.
References
- Di Salvo, I., Mnzava, D., Nicoletti, G. J., Senkoro, E., Ndege, R. C., et al. (2023). Upscaling cervical cancer screening and treatment for women living with HIV at a rural referral hospital in Tanzania: Protocol of a before-and-after study exploring HPV testing and novel diagnostics. BMC Health Services Research, 23(1):234.
Publisher | Google Scholor - Olana, M. D., & Kumburi, G. T. (2019). Distribution of high risk human papilloma virus genotypes among women with cervical cancer in East Africa: Systematic review. Journal of Family Medicine and Health Care, 5(1):8–14.
Publisher | Google Scholor - Nartey, Y., Amo-Antwi, K., Hill, P. C., Dassah, E. T., Asmah, R. H., Nyarko, K. M., et al. (2023). Human papillomavirus genotype distribution among women with and without cervical cancer: Implication for vaccination and screening in Ghana. PLOS ONE, 18(1):e0280437.
Publisher | Google Scholor - Seyoum, A., Seyoum, B., Gure, T., Alemu, A., Belachew, A., et al. (2023). Genotype heterogeneity of high-risk human papillomavirus infection in Ethiopia. Frontiers in Microbiology, 14:1116685.
Publisher | Google Scholor - Das, M. (2021). WHO launches strategy to accelerate elimination of cervical cancer. The Lancet Oncology, 22(1):20–21.
Publisher | Google Scholor - Moher, D., Shamseer, L., Clarke, M., Ghersi, D., Liberati, A., Petticrew, M., et al. (2015). Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic Reviews, 4:1–9.
Publisher | Google Scholor - Countrymeters. (2023). Population figures are estimates based on the latest United Nations data.
Publisher | Google Scholor - Joanna Briggs Institute. (2020). Checklist for analytical cross sectional studies.
Publisher | Google Scholor - Schünemann, H. J., Tugwell, P., Reeves, B. C., et al. (2013). Non-randomized studies as a source of complementary, sequential or replacement evidence for randomized controlled trials in systematic reviews on the effects of interventions. Research Synthesis Methods, 4:49–62.
Publisher | Google Scholor - Haile, E. L., Cindy, S., Ina, B., Belay, G., Jean-Pierre, V. G., et al. (2019). HPV testing on vaginal/cervical nurse-assisted self-samples versus clinician-taken specimens and the HPV prevalence in Adama Town, Ethiopia. Medicine, 98(35):e16970.
Publisher | Google Scholor - Teka, B., Gizaw, M., Ruddies, F., Addissie, A., Chanyalew, Z., et al. (2021). Population-based human papillomavirus infection and genotype distribution among women in rural areas of South-Central Ethiopia. International Journal of Cancer, 148(3):723–730.
Publisher | Google Scholor - Bogale, A. L., Teklehaymanot, T., Kassie, G. M., Medhin, G., Ali, J. H., & Belay, N. B. (2022). Performance of visual inspection with acetic acid for cervical cancer screening as compared to human papillomavirus DNA testing among women with HIV in Ethiopia: A comparative cross-sectional study. Cancer Control, 29:10732748221114980.
Publisher | Google Scholor - Ali, K. E., Mohammed, I. A., Difabachew, M. N., Demeke, D. S., Haile, T., et al. (2019). Burden and genotype distribution of high-risk human papillomavirus infection and cervical cytology abnormalities at selected obstetrics and gynecology clinics of Addis Ababa, Ethiopia. BMC Cancer, 19(1):768.
Publisher | Google Scholor - Wolday, D., Derese, M., Gebressellassie, S., Tsegaye, B., Ergete, W., et al. (2018). HPV genotype distribution among women with normal and abnormal cervical cytology presenting in a tertiary gynecology referral clinic in Ethiopia. Infectious Agents and Cancer, 13:28.
Publisher | Google Scholor - Derbie, A., Maier, M., Amare, B., Misgan, E., Nibret, E., et al. (2023). High-risk human papillomavirus genotype distribution among women with gynecology complaints in northwest Ethiopia. Infectious Agents and Cancer, 18(1):4.
Publisher | Google Scholor - Leyh-Bannurah, S. R., Prugger, C., de Koning, M. N., Goette, H., & Lellé, R. J. (2014). Cervical human papillomavirus prevalence and genotype distribution among hybrid capture 2 positive women 15 to 64 years of age in the Gurage zone, rural Ethiopia. Infectious Agents and Cancer, 9(1):33.
Publisher | Google Scholor - Temesgen, M. M., Alemu, T., Shiferaw, B., Legesse, S., Zeru, T., Haile, M., & Gelanew, T. (2021). Prevalence of oncogenic human papillomavirus (HPV 16/18) infection, cervical lesions and its associated factors among women aged 21–49 years in Amhara region, Northern Ethiopia. PLOS ONE, 16(3):e0248949.
Publisher | Google Scholor - Taku, O., Businge, C. B., Mdaka, M. L., Phohlo, K., Basera, W., et al. (2020). Human papillomavirus prevalence and risk factors among HIV-negative and HIV-positive women residing in rural Eastern Cape, South Africa. International Journal of Infectious Diseases, 95:176–182.
Publisher | Google Scholor - Gebremariam, T. (2016). Human papillomavirus related cervical cancer and anticipated vaccination challenges in Ethiopia. International Journal of Health Sciences, 10(1):137–143.
Publisher | Google Scholor