Review Article
Pioneering New Frontiers: Breakthroughs and Barriers in Clinical Trials
- Kashish Kaithwas
- Akash Yadav *
- Neelam Balekar
- Dinesh Kumar Jain
IPS Academy College of Pharmacy, Indore, Madhya Pradesh, India.
*Corresponding Author: Akash Yadav, IPS Academy College of Pharmacy, Indore, Madhya Pradesh, India.
Citation: Kaithwas K., Yadav A., Balekar N., Dinesh K. Jain. (2025). Pioneering New Frontiers: Breakthroughs and Barriers in Clinical Trials, Journal of Clinical Research and Clinical Trials, BioRes Scientia Publishers. 4(5):1-13. DOI: 10.59657/2837-7184.brs.25.057
Copyright: © 2025 Akash Yadav, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: March 27, 2025 | Accepted: October 03, 2025 | Published: October 10, 2025
Abstract
Clinical trial design and interpretation are fundamental components of evidence-based medicine, serving as the basis for well-informed treatment plans and healthcare decisions. In order to reduce bias and guarantee the validity of study findings, this review article examines the fundamental components of clinical trial design, such as randomization, blinding, control groups, sample size calculation, and ethical issues. The essay also looks at how clinical trial results are interpreted, emphasizing the significance of statistical significance, clinical relevance, and the possibility of confounding variables. It also covers issues including the impact of protocol violations, generalizability, and incorporating trial results into clinical practice. By thoroughly examining both design and interpretation, this review seeks to offer insightful information to researchers, physicians, and policymakers in optimizing the quality and applicability of clinical trial results.
Keywords: clinical trial design; trials of adaptation; significance of clinical trials
Introduction
A clinical trial is an investigation of the safety, efficacy, and possible adverse effects of novel medical procedures, medications, equipment, or therapies that is carried out on human subjects. By establishing whether novel interventions are more effective than current treatments, these studies are crucial for expanding medical knowledge and enhancing healthcare.
Organizations like the International Conference on Harmonization (ICH), European Medicines Agency (EMA), and U.S. Food and Drug Administration (FDA) have established stringent ethical standards and regulations that clinical trials must adhere to.
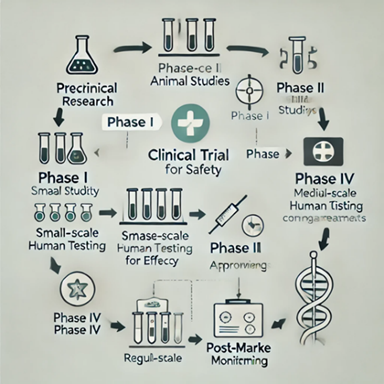
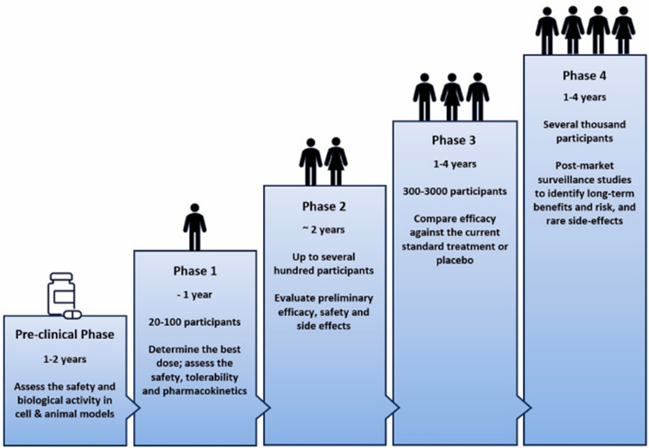
Phase I: Evaluates a new medication or treatment's dose and safety in a limited number of subjects.
Phase II: Assesses the treatment's efficacy and safety in further detail.
Phase III: Examines the new treatment in a broader population and compares it to traditional treatments. Following approval, phase IV is carried out to track the treatment's efficacy and long-term consequences in practical setting.
Interventional trials also known as treatment trials, are used to test novel medications, medical equipment, surgeries, or behavioral therapies to see how they affect patient outcomes.
Observational Trials: Track subjects without getting involved, gathering information to identify risk variables and disease trends.
Prevention Trials: Research methods for preventing recurrence or preventing diseases in those who have never experienced them.
Screening Trials: Examine novel approaches to early disease detection.
Diagnostic Trials: Evaluate novel methods or tests for illness diagnosis.
Trials on Quality of Life (Supportive Care): These studies concentrate on methods to enhance the comfort and standard of living of people suffering from long-term conditions.
Studies on bioequivalence and bioavailability compare generic and name-brand medications to make sure they function similarly.
Figure 1: Phases of Clinical Trails.
Artificial Intelligence in Clinical Trials of Lung Cancer
Globally, the incidence and mortality rates of cancer are rising, with lung cancer now having the greatest incidence. Lung cancer is the most common cause of cancer related fatalities globally because of its high mortality rate. One Lung cancer is predicted to be the leading cause of cancer related fatalities in the US in Lung cancer is both the most common and the deadliest and often diagnosed malignancy. The two main histological types of lung cancer are non-small cell lung cancer (NSCLC), which accounts for 80–85% of cases, and small cell lung cancer (SCLC), which accounts for 10%-15% of cases. Lung cancer is derived from respirator epithelial cells.
Preclinical Drug Discovery
There is a lot of promise and promise in using AI in drug development, especially in the preclinical studies of medications for lung cancer. By simulating human intelligence, artificial intelligence (AI) lowers costs and improves efficacy by reducing the human and material resources needed for medication discovery and development. Figure shows how AI is being used in the preclinical development of medications for lung cancer [5-8].
Clinical trial designs for rare diseases variables, such as improved access to diagnostic and treatment services, public education, and screening for early diagnosis. services, enhanced supportive care, better training, and quality assurance in the prov Rapid advancements in cancer diagnosis and treatment have been made during the last three decades, which has improved cancer patients' survival and other results. This progress has been made possible by a number of ision of treatment. Research has been the primary driver of advancement, with both public and private sector funding for preclinical and clinical studies resulting in a quick growth of the body of evidence. For instance, the number of cancer patients taking part in research increased by more than five times after the establishment of the United Kingdom's (UK) government sponsored National Cancer Research Network in 2001.so that a national portfolio of trials includes 20% of all cancer patients. Because of this, treatment is frequently predicated on inadequate data, and researchers have limited access to cutting-edge medications and technologies.
This poses a significant public order for 20% of all cancer patients to take part in a nationwide study portfolio. Unsurprisingly, common malignancies have been the focus of research activity: The major cancers are given priority by the industry. Those with the greatest clinical need are given priority by public sector and possible market fund Furthermore, it may not be viable to conduct a credible study in a single nation for a rare malignancy in a fair amount of time, and conducting and delivering sufficiently-powered research requires a significant number of patients. As a result, innovative treatments and technology for research are not readily available, and treatment is frequently predicated on inadequate data.
This is a serious problem for public health. Paradoxically, rare malignancies are a prevalent issue, making up 22% of all cancer diagnoses, which is more than any one common malignancy. It isn't getting better IRCI, or the International Rare Cancers Initiative, was established in 2011 as a collaboration between the United States of America (USA) National Cancer Institute Cancer Therapy Evaluation Program (CTEP), Cancer Research UK, the National Institute of Health Research Cancer Research Network (NCRN) in England, and the European Organization for Research and Treatment of Cancer (EORTC).
The goal of IRCI is to support and encourage the growth of global clinical trials for individuals with uncommon malignancies. It focuses on interventional measures, typically clinical trials that are randomised and have the express purpose of enhancing patient outcomes.
IRCI Has Two Significant Obstacles to Overcome
The Initial One is Organizational: bringing together researchers from many nations, reaching an agreement, and getting past the numerous financial and regulatory obstacles that can prevent international clinical research from operating smoothly.
The Second is Methodological: traditional trial designs may necessitate unrealistically high recruitment targets for the setting, even with international collaboration, necessitating the use of creative research approaches. In September 2013, a multidisciplinary workshop was conducted in Amsterdam to examine the procedures utilized in ICRI portfolio trials. Additional unrealized techniques were also talked about. Here, we outline a few of the approaches that are available and provide examples from the IRCI portfolio to support them.
Methods, Findings: Each of the IRCI trials is offered as an example of a potential strategy to address the difficulties in creating reliable studies for uncommon tumors. A number of trials employed several
Utilise A Phase II Design (Anal cancer): When there aren't enough patients for a phase III study, one option is to create a future collaboration using a randomised phase II design, like InterAACT, an international, multicentre, open label, randomised controlled trial (RCT). It's the initial First line therapy for individuals with inoperable locally recurrent or metastatic squamous cell carcinoma of the anus (SCCA) is being studied prospectively. Eighty patients will be assigned at random to receive carboplatin + paclitaxe or cisplatin 5 FU. 2011 age- standardized incidence rates for novel. SCCA instances were 1.5 in the UK and 1.7 in the USA per100,000, accounting for 0.4% of newly diagnosed malignancies. Local 20% of individuals receiving chemotherapy experience recurrence, and 10% to 17 percentage develop metastases. In line with the Monitoring, Epidemiology, and Outcomes (SEER) program, the 5-year survival rate for metastatic illness is anticipated to be 32 percentage.
Inter AACT's primary goals are to: establish the best chemotherapy foundation for use in conjunction with novel targeted agents in upcoming trials; enable more investigation of tumor biology provide prospective randomized evidence for first-line treatment of inoperable locally advanced or metastatic SCCA; and promote the future advancement of targeted treatment approaches; and determine whether international SCCA trials may be set up and recruited.
A phase III RCT of adding more agents to the selected region from InterAACT is the longer-term goals. The primary issue throughout the planning phase was the recruitment rate; despite worldwide cooperation, expected enrollment is just 30 points each year.
The response rate, which is predicted to be 40% with cisplatin + 5-FU, is the main outcome measure. An improvement that is clinically significant by Using a typical sample size calculation with a 2-sided 5% significance level, 80% power, and an impractical 25-year accrual, 10–50% with carboplatin + paclitaxel required 388 patients/arm. Instead, in order to achieve the same target difference and power, researchers used a Simon, Wittes, and Ellenberg randomized selection trial design, which required 40 patients per arm.
Accrual should be finished in about three years. However, this architecture offers little defense against Type 1 errors. The process will choose one at random if the response rates to the trial regimens are extremely similar. The regimen that exhibits the fewest serious effects will be approved if the rate of response is the same. Better quality of life will determine whether toxicity is also equal.
Accept A Greater Type I Error (Salivary Gland Cancer)
The likelihood of incorrectly rejecting a null hypothesis (H0) and incorrectly determining that the research treatment is effective, active, or fascinating is known as the type I error.
Traditionally, this is 5 percentage, or 1/20. The likelihood of mistakenly accepting the null hypothesis and overlooking an intriguing therapy is known as the type II error. Generally speaking, a higher probability of type 2 errors typically 1/10 or 1/5, or 90 percentage or 80 percentage power, respectively is acceptable. Accepting a type I error that is more similar to a normal type II error could reduce the necessary sample size.
This method is applied in the heterogeneous EORTC 1206 HNCG of salivary gland carcinomas collection of uncommon tumors. Less than 5% of head and neck malignancies have SGC histologies. Chemotherapy (CT) patients had poor results and low response rates. Case series provide strong evidence of susceptibility to androgen deprivation therapy (ADT) in SGCs that express the androgen receptor (AR) [5-8]. Thus, limited to salivary duct cancer and adenocarcinoma, two histology where AR expression is more prevalent, this trial contrasts ADT and CT in treatment-naive recurrent and/or metastatic SGC The progression Free survival (PFS) metric is the main outcome measure. An experimental sub study assesses ADT in patients who have already received treatment, but the main study requires patients to be treatment- naïve in order to estimate PFS.
The difficulty lay in creating a trial that struck a reasonable balance between the degree of scientific evidence and trial size and length feasibility. It was challenging to obtain reliable PFS estimates for patients treated with CT because published studies were few and varied in terms of histology, AR expression, and chemotherapy. The argument for randomization is strengthened by the lack of reliable reference data. Prior to choosing a comparative randomized design, single arm and non-comparative randomized designs were eliminated. The effects of easing type I and type II errors were the main topic of discussion.
To prevent arbitrary time, point selection, a time to event (PFS curve comparison) approach was chosen as the PFS evaluation method instead of a binary (progression free rate at one time point relative to accrual). This decision resulted in a smaller sample size 80 percentage power and 10 percentage one sided significance were chosen as the pragmatic values for type I and type II errors.
Based on 16 patients receiving ADT, the goal effect size of hazard ratio (HR) 0.56 was later selected; this is equal to raising 6-month PFS from 60% to 75%. After surveying interested institutions, it was determined that the design's requirement of 76 randomly assigned patients over two years could be met. One year following the completion of accrual, fifty-five PFS events are anticipated. Frequentist analysis is the main one. The conclusions will be evaluated for robustness for different prior distributions of the treatment effect hazard ratio using a sensitivity analysis based on Bayesian methods [1-4].
International Bladder Cancer Group Consensus Statement on Clinical Trial Design for Patients with Bacillus Calmette Guérin Exposed High Risk Non-Muscle Invasive Bladder Cancer
For newly diagnosed treatment-naïve non muscle invasive bladder (NMIBC), the current standard intravesical therapy is founded on level 1 evidence from prospective randomized trials and meta-analyses. Using postoperative single-dose chemotherapy is one example of this and induction and BCG maintenance for 1-3 years However, second line therapeutic advancements have been sluggish until lately. It is possible to hypothesize that the lack of promising new treatments had a role in the field's stagnation, but it is undeniable that inconsistent trial designs and the lack of clearly defined NMIBC disease states have also been major roadblocks.
Evidence Acquisition: To find published clinical trials, reviews, clinical practice guidelines, systematic reviews, and meta-analyses that reported clinical trial outcomes or addressed clinical trial design in NMIBC, a search was done in the Cochrane Library, Medline, and Embase between January 1995 and March 2021."Non-muscle invasive bladder cancer," "clinical trials," "study designs," "high-risk," "BCG failure," "BCG refractory," "BCG relapsing," and "intravesical treatment" were among the keywords. On ClinicalTrials.gov, active or recruiting clinical trials were found by searching for "BCG and Bladder" and "NMIBC and BCG."
Abstracts of certain trial results were used in presentations at the annual meetings of the Society of Urologic Sciences, the European Association of Urology (EAU), and the American Urological Association (AUA). American Society of Clinical Oncology, European Society of Medical Oncology, and Oncology.
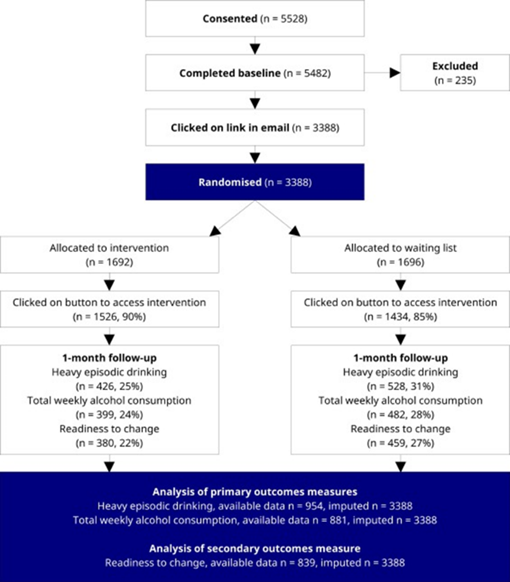
Effects of a Waiting List Control Design on Alcohol Consumption Among Online Help-Seekers: A Randomized Controlled Trial
The gold standard for estimating the effects of interventions is the randomised controlled trial (RCT), where effects are defined as the differences between participants randomly assigned to various circumstances. Although time and energy are often invested in creating and when discussing the treatment condition in RCTs, the control condition receives less attention in the research literature than the treatment condition. This neglect is troubling since choices about the control conditions could skew effect estimates, making it more difficult to interpret the results. Any such bias, if it exists and is not taken into consideration, may also affect how research findings are applied when making decisions.
Figure 2: Effects of a waiting list control design on alcohol consumption among online help-seekers: A randomized controlled trial.
Participants
Adults looking for online support to cut back on drinking were the target demographic. People had to be at least eighteen years old and be categorized as having dangerous or toxic alcohol consumption. Having six or more drinks at least once in the previous month or more than nine drinks in the previous week were considered hazardous or dangerous alcohol use. Twelve grams of alcohol was considered a drink. Although the study center was located in Sweden, we were able to recruit participants online without regard to national boundaries. Understanding English was a requirement for participation because all study materials were in that language.
We used internet marketing to find study volunteers. After clicking on the advertisement, participants were directed to the study's webpage, which included trial details and materials for informed consent. Those who agreed were requested to complete a baseline survey that evaluated their eligibility as well. Lastly, in order to get updates regarding the study's future steps, participants were asked to supply an email address. Participants had to click on a hyperlink in the email that was delivered right away to confirm their involvement in the study. Those who clicked on the URL were assigned at random to either the waiting list group or the intervention group.
Interventions
We gave each group tailored comments and guidance based on the baseline survey results. Our earlier studies based on theories of health behavior that are socially cognitive in nature. The resources included advice on how to cut back on drinking and stay away from triggers in the surroundings, an evaluation of current consumption in relation to recommendations, and content aimed at boosting motivation and self-efficacy. The varied information we gave participants after they clicked the email's link and before they got feedback and guidance is what causes the experimental contrast in this study.
Text To Intervention Group: This study is divided into two groups. You belong to the group that gets prompt assistance and feedback based on your answers to the questionnaire you previously filled out. This information has been created for those who are worried about their alcohol consumption.
Text to waiting list group: This study is divided into two groups. You belong to the group that will have to wait for the supporting documentation and comments. In four weeks, we will get in touch with you once again, and you will be provided with the information. We have produced a report in the interim based on your answers to the earlier questionnaire.
Run-In Periods and Treatment Outcomes in Asthma Trials: Randomized controlled trials (RCTs) are critically important to assess the effects of a particular treatment on a defined population of patients, as part of the regulatory approval process of new medicines, and to form the evidentiary basis for recommendations made in respiratory clinical practice guidelines. The design and conduct of an RCT is of the utmost importance, underpinning the validity of study outcomes. One element of RCTs often used in respiratory disease trials is the run-in period the period between the screening visit and randomization to study treatments the design of which requires close consideration [9-11].
OARSI Clinical Trials Recommendations
Key Analytic Considerations in Design, Analysis, And Reporting of Randomized Controlled Trials in Osteoarthritis
Several design elements mentioned in this paper should be given particular consideration by researchers planning and carrying out randomized controlled trials to determine the effectiveness of therapies for OA: modifications to the trial's design, blinding, placebo definition, and selection of main result, the best time to evaluate it, and how to avoid informational censorship. We offer suggestions for resolving these problems during the design and analysis phases. We also stress the importance of information analysis for setting research priorities, standardized reporting, and economic evaluation in addition to clinical trials.
Osteoarthritis Trials
Drugs that alter structure as well as symptoms make up the pharmacological treatment for OA. The main goals of non-pharmacologic therapies are often symptom alleviation and functional restoration, but it can also be assessed for structural change utilizing imaging or molecular indicators. The IDEA trial, for instance, examined the effectiveness of exercise and weight control in reducing pain, but it also shown effects on MRI and inflammatory biomarkers.
Clinical outcomes are also crucial for therapies that alter structure. For instance, the time needed for joint replacement could be an endpoint of the structure modifying intervention. A biomarker may be used in place of the genuine clinical endpoint (such as a decrease in joint space) provided the treatment's impact on the biomarker is consistent with its impact on the genuine endpoint [12-16].
Sex As a Biological Variable in Early-Phase Oncology Clinical Trials: Enhancing the Path to Personalised Medicine
By customizing care for certain patient groups, personalized medicine seeks to increase medication effectiveness and reduce adverse effects. Oncology medication research has seen significant changes in the past ten years, moving from broad, organ centric methods to precisely targeting genetic abnormalities unique to individual tumors. Although patient outcomes have significantly improved as a result of this concentration on genetics, neglecting to take into account more comprehensive patient characteristics runs the danger of ignoring the influence of other intricate disease and treatment response modulators. When it comes to sex, a basic biological characteristic and driver of health and disease throughout a patient's life, this neglect is particularly noticeable.
The outcomes evaluated in phase 1 and phase 2 clinical trials, which assess the safety, tolerability, pharmacokinetics, pharmacodynamics, and initial efficacy of novel cancer treatments, are significantly influenced by sex. While these in early phase trials, a small group of patients is given increasing dosages of the medicine to determine the maximum tolerated dose (MTD). Dosage selection for testing in bigger, phase 3 registrational studies is guided by this information. This paradigm, however, is predicated on the idea that "one dose fits all"; the majority of antibodies and cytotoxic chemicals are dosed based on body weight and body Surface area (BSA) respectively.
Figure 3: Sex as a biological variable in early phase oncology clinical trials: enhancing the path to personalized medicine.
Sex-Specific Variations in Cancer Development and Therapeutic Targets
The sex chromosomes and the amounts of sex hormones that male and female cells are exposed to are the two main distinctions between them. Sex hormones and chromosomes interact to affect systemic factors like immune system function and cell metabolism as well as local factors like cancer initiating cells and the tumor microenvironment A number of processes, including as the existence or lack of single or double copies of the gene, genetic imprinting, meiotic effects, and X chromosome inactivation, can result in phenotypic changes based on sexual genotype (XX in females, XY in men).
In fact, at least eight of the 23 tumor suppressor genes on the X chromosome are immune to X chromosome inactivation, which gives female patients some protection against the development of cancer. Extreme Y chromosome deletion or downregulation in males is strongly linked to 12 main non-reproductive malignancies. Loss of both X and Y homologues is necessary for the development of squamous-like tumors in the context of pancreatic cancer, where LOY occurs in around 40% of male cases on the other hand, albeit uncommon, biallelic deletion of the X linked KDM6A indicates poor prognoses for females with squamous-like pancreatic cancer.
It increases susceptibility to BET inhibitors, underscoring the necessity of sex-specific biomarkers and customized therapeutic approaches. Due to the inadequacy of current analytical techniques to handle their particular technical hurdles, sex chromosomes are frequently left out of genome-wide association studies. As a result, little is known about the genetic basis of sex differences in cancer outcomes and the preferential study of autosomes. Nevertheless, considerable sex-biased patterns in autosomal gene expression, mutation patterns, and loads have been found by thorough molecular investigations conducted across a variety of tumor types. Significantly, sex-biased signals were found in 53% of clinically actionable genes [17-19].
Promoting Diversity in Clinical Trials
Individuals and the healthcare system may suffer as a result of racial and ethnic discrepancies in clinical research; variations in drug exposure and reaction among racial and ethnic groups may have an effect on clinical data, confusing expectations and treatment results in practice. Despite regulatory permission, underrepresentation in clinical research may limit impacted groups' access to potentially effective medicines. Furthermore, there can be significant societal costs associated with health disparities; according to a US study, reducing 1% of health inequalities by improved representation in clinical research would result in an additional $60 billion in savings for the treatment of heart disease. Systemic racism,
Which includes discrimination in clinical practice and healthcare inequities, is a significant contributing cause to underrepresentation investigation. Non-White communities' mistrust of healthcare is still fueled by discriminatory practices, such as those seen during the Tuskegee Syphilis research, which may deter them from participating in clinical studies. In order to make it easier to attract racially and ethnically underrepresented populations (URPs) into clinical trials, we will focus on modifiable criteria here.
includes discrimination in clinical practice and healthcare inequities, is a significant contributing cause to underrepresentation investigation. Non-White communities' mistrust of healthcare is still fueled by discriminatory practices, such as those seen during the Tuskegee Syphilis research, which may deter them from participating in clinical studies. In order to make it easier to attract racially and ethnically underrepresented populations (URPs) into clinical trials, we will focus on modifiable criteria here [20].
Research Design Characteristics of Published Pharmacologic Randomized Clinical Trials for Irritable Bowel Syndrome and Chronic Pelvic Pain Conditions
Lower abdominal and pelvic chronic pain disorders are common, frequently difficult to treat, and can have a detrimental impact on psychological and sexual well-being as well as other aspects of health-related quality of life. There are currently very few approved medicines for these illnesses from the European Medicines Agency (EMA) or the Food and Drug Administration (FDA) in the United States. The methodological difficulties in creating randomized clinical trials (RCTs) to assess the effectiveness of new treatments in certain illnesses contribute to the lack of available medicines. First, lower abdominal discomfort frequently co-occurs with other symptoms (such as diarrhea, constipation, or frequent urination), which might impact the intensity of the pain. Furthermore, the illness may cause flare-ups of pain and pain that only happens during particular activities
Designing a single main endpoint that is both clinically meaningful to patients and responsive to treatment is difficult because of these features. While it is feasible to evaluate multiple outcomes in RCTs, defining a single major end point or statistically correcting for multiple primary analyses is required to reduce false positive results in clinical trials and appropriately assess the overall efficacy of a treatment in conditions with many symptoms [13-16].
Leveraging Mathematical Models to Improve the Statistical Robustness of Cancer Immunotherapy Trials
Clinical results for cancer have improved as a result of well-planned cancer immunotherapy trials and clinical investigations that try to use a patient's immune system to fight cancer patients. Advanced trial designs, such as master protocols or biomarker-driven trials, seek to find efficient therapies by utilizing the ever-increasing amount of clinical data. Although patient needs and technical advancements are directly responsible for the rising complexity of clinical trials, this presents difficult problems for trial design and interpretation
How many patients are required to get enough statistical power is one of the key questions in trial design. How long do we keep an eye on patients to assess the effectiveness of treatment Which impact size risk difference or hazard ratio, for example will be applied If the medication proves to be more effective than expected, will interim analyses be conducted to end the trial early, and if so, when? Can we lower the number of people who must get a placebo if the experiment is randomized It is necessary to make some assumptions about the treatment effect in order to provide answers to such inquiries beforehand. These presumptions are primarily statistical at the moment. To decide how many patients must be included, for instance, the "proportional hazards assumption" might be used in conjunction with an estimate of the treatment effect size [4-8].
Use of Pragmatic and Explanatory Trial Designs in Acute Care Research
Almost every facet of society and medicine has been impacted by the COVID-19 epidemic. It has resulted in significant emotional and financial hardship on a global scale, but recent advancements in vaccinations to stop infections and serious illnesses Hope for a post-pandemic life is provided by and efficient treatments for early and late stage [8-10]. COVID-19. Furthermore, there is hope that the pandemic's unprecedented investment in clinical research has opened up new avenues for medical research in other fields. For instance, recently validated technologies like mRNA vaccines are already being considered as treatments for a number of cancers, HIV, and chikungunya.
During the epidemic, there has been a comparable but less acknowledged development in the planning, carrying out, and evaluating of clinical trials comparing treatments that are already similar clinical use, resulting in glaring differences between the methods employed by various nations to assess and employ repurposed therapies (corticosteroids, hydroxychloroquine, convalescent plasma, anticoagulation techniques, andlopinavir ritonavir) as COVID-19 hospitalization treatments. Small explanatory studies that concentrated on intermediate patient- centered outcomes were carried out in the context of the widespread clinical use of these medicines in several nations, including the USA, before significant results were apparent.
Off-label use of COVID-19 medicines was prohibited in the UK in favor of extensive pragmatic trials that concentrated on mortality. The United Kingdom Studies allowed for changes to conventional consent processes to enable quick enrollment and integrated trial protocols into clinical care. While a lot of focus has been placed on how experiments, like those carried out in the UK, employed a single infrastructure to answer several questions (adaptive platform design) and used contemporary statistical analysis techniques to increase the possibility of offering conclusive proof for every intervention (Bayesian sequential analysis),14 but little focus has been placed on how these pragmatic trials' approaches to enrollment, consent, and intervention delivery varied from those of conventional, explanatory trials.
To identify issues with and possible solutions for the planning, execution, and interpretation of clinical trials in critically ill adults, the annual 3CT Workshop brings together a broad range of international clinical trialists, epidemiologists, bioethicists, regulators, staff from federal funding agencies, industry representatives, clinicians, and patient advocates [18-19].
Next-Gen Spinal Cord Injury Clinical Trials
Deficits in the sensory and autonomic nerve systems, as well as varied degrees of motor paralysis, are hallmarks of spinal cord injury, a devastating neurological disorder.
Although spinal cord injuries are rare, they have a significant detrimental effect on a person's quality of life and place a heavy financial and social burden on society. An effective neurorestorative strategy is still elusive despite a growing understanding of the molecular and cellular targets involved in protecting and repairing the damaged spinal cord. It was largely due to a lack of effort until recently that there was no successful intervention that restored neurological function following spinal cord injury.
During the almost 100 experiments conducted between 1980 and 2000 to restore neurological function in stroke patients,14 only a few neurorestorative clinical trials including patients with spinal cord injuries were started. Following the National Acute Spinal Cord Injury Studies (NASCIS) and Sygen, research focused on managing chronic spinal cord injury, specifically examining the effects of aminopryidine. Humans made their first attempts at using cell-based therapies to restore the damaged spinal cord ten years after Sygen. Trials that were eventually stopped before they reached their pre-established endpoints Neurorestorative spinal cord injury trials have exploded, with several reporting results in the last four to five years, despite worries that early terminations would discourage future funding. On the one hand, recently developed or "next-gen" studies and their predecessors are "failures" ineffective attempts to regain neurological function following spinal cord injury.
However, we contend that they are a step forward and priceless contributions that have given important information and a foundation for upcoming clinical trials. This is because new clinical studies for spinal cord injuries are showing important lessons learned after "failing." Instead of summarizing trial findings, the purpose of this personal opinion is to draw attention to aspects of trial design that indicate progress and a promising future for spinal cord injury.
Adaptative Designs: The New Normal: Spinal cord injury has historically taken a conventional, randomised controlled trial (RCT) approach. While tried and tested, other fields have turned to innovative study designs. Among them, adaptive designs are an intuitive option, highlighted in recent spinal cord injury trials.
In the Riluzole in Acute Spinal Cord Injury (RISCIS) Study, investigators adopted a sequential adaptive design that incorporated a pre-specified interim analysis to adjust sample size. The importance of this design lies in accounting for the challenges in estimating the degree of spontaneous recovery in the placebo treated individuals, with small imbalances in injury severity between treated and placebo groups, difficult to predict at the trial onset, masking otherwise meaningful therapeutic effects. Another example of an adaptive design is evidenced in the AXER-204 trial a seminal study examining the safety and efficacy of Nogo decoy therapy after spinal cord injury.
The AXER 204 experiment, a landmark investigation on the safety and effectiveness of Nogo decoy therapy following spinal cord damage, provides yet another illustration of an adaptive design. 27 Researchers used an adaptive seamless design in this study, combining a randomized, placebo-controlled, double-blinded trial (part 2) with an open-label, single ascending dosage (part 1). This approach's primary objective was to expedite clinical trial testing, which was partially accomplished by enrolling part 1 participants in part 2. A recent cell-based trial called Stem Cells in Spinal Cord Injury (SCI2) similarly used this flexible, seamless method, with a planned interim analysis as the "phase II study expands into a phase III study."
A strong body of evidence supports dietary recommendations, including excellent clinical trials, some of which have been created to demonstrate a causal link between dietary changes and lowering the risk of ASCVD.
However, the difficulty of carrying out these studies has led to criticism of food and nutrition since the quality and strength of the evidence for certain medicinal therapies is not up to par.
The purpose of this paper is to raise knowledge of the benefits of ASCVD demonstrated by nutrition-related clinical trials and the ways in which this data has influenced dietary recommendations.
Clinical studies pertaining to nutrition entail a number of special issues when compared to those involving pharmacological treatments. Numerous lines of data, such as those from epidemiological studies, randomized controlled trials, animal and in vitro research, and other studies, are used to support evidence-based dietary recommendations. The basic evidence for the significance of nutrition in ASCVD prevention, management, and treatment has been supplied by this research. A significant consensus has developed regarding the essential components based on the clinical trials that have been carried out. Because dietary exposures are multi-factorial, interdependent, and interconnected, finding a tested, repeatable intervention is a prerequisite for carrying out nutrition-focused clinical studies.
Thus, nutrition-focused Pharmaceutical clinical studies, in which the intervention group and the control group are only exposed to a known amount of a single medicine, are fundamentally different from clinical trials. Questions concerning the necessity and worth of nutrition interventions arise in light of the strong evidence that pharmaceutical agents, such as lipid/lipoproteins with statins4, blood pressure-lowering drugs, and GLP 1 receptor agonists, can significantly reduce ASCVD events and major CVD risk factors.
However, this ignores the advantages of maintaining a balanced diet for primary and fundamental prevention at the individual, social, and health system levels. eliminating other significant chronic illnesses, and raising life expectancy and quality of life Approximately one in five deaths in economically developed nations and a growing percentage in emerging cultures are attributed to atherosclerotic cardiovascular disease (ASCVD), which remains the world's leading cause of death.
The mainstay of ASCVD prevention is adhering to a healthy dietary pattern. However, due to the numerous complexities, creating dietary recommendations based on excellent clinical trials that demonstrate a causal link between food and ASCVD risk is difficult. Related to planning and carrying out these investigations.
This paper aims to give a summary of the difficulties in carrying out clinical studies with a nutrition emphasis that support high evidence ratings during the guideline generation process and the consequences for dietary recommendations [4].
Chronic Pain Disorders That Affect the Lower Abdomen and Pelvis
This are common and frequently difficult to control, and may have a detrimental impact on sexual and psychological well-being as well as health-related quality of life. There are now approved therapies for certain illnesses from the European Medicines Agency (EMA) or the Food and Drug Administration (FDA). The methodological difficulties in creating randomized clinical trials (RCTs) to assess the consti effectiveness of new treatments in certain illnesses contribute to the lack of available medicines. First, lower abdominal discomfort frequently co-occurs with other symptoms (such as diarrhea, ation, or frequent urination), which might impact the intensity of the pain.
Furthermore, pain may only occur during particular activities (such as sexual activity) and the illness may feature flare-ups of pain, which are periods of heightened symptom severity. It is difficult to create a single primary endpoint that has clinical significance for patients because of these features and receptive to therapy. While various outcomes can be evaluated in RCTs, in order to properly examine.
To reduce false positive results in clinical trials, it is vital to determine a single primary end point or statistically correct for numerous primary analyses when evaluating the overall effect of a treatment in conditions with various symptoms.
Investigate is required to determine the elements of research design and techniques that can optimize the clinical significance and assay sensitivity of end points in these trials (e.g., qualitative patient interviews regarding the impact of various symptom patterns; secondary analyses that compare the effect sizes associated with various end points in existing trial data).
Additionally, to enhance the dissemination and integration of results into clinical practice, more consistent reporting of design characteristics that are crucial for the interpretation of trial results and for meta-analyses of results across studies is necessary [14-15].
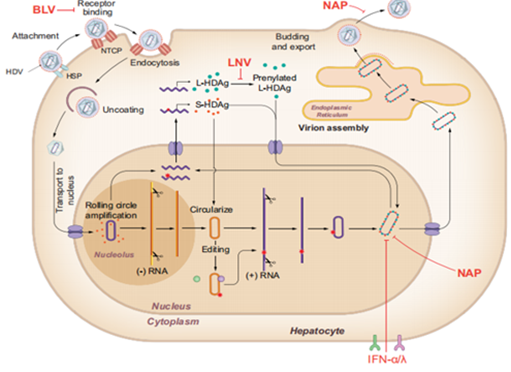
Guidance on Treatment Endpoints and Study Design for Clinical Trials Aiming to Achieve Cure in Chronic Hepatitis B And D
As a follow-up to similar conferences held in 2016 and 2019, the American Association for the Study of Liver Diseases (AASLD) and the European Association for the Study of the Liver (EASL) jointly organized an HBV Treatment Endpoints Conference in Washington, DC, on June 3 and 4, 2022, with the aim of promoting and facilitating the planning and execution of new clinical trials with the goal of developing finite treatments resulting in a "functional cure" for chronic hepatitis B (CHB). This meeting brought together participants from academics, industry, regulatory authorities (the European Medicines Agency [EMA] and the US Food and Drug Administration [FDA]), international organizations, and patient advocacy groups. With a focus on endpoints, trial design, safety, and monitoring for novel therapies aiming to achieve an HBV or HDV "cure," this report highlights nine "key questions" that were addressed at the conference and provides an overview of the discussions and viewpoints of experts and patient advocacy groups that attended the two-day gathering. The meeting organizers were chosen by AASLD and EASL, and the writers' report reflects the opinions of the attendees, which the conference's speakers and moderators have examined and accepted.
The primary objective of the conference was to establish a consensus on HBV and HDV treatment endpoints trials that seek to "cure" chronic HBV and HDV infections in light of the development of innovative medicines
Hepatitis B
The main objective of CHB treatment is to increase survival by averting liver-related mortality, cirrhosis, hepatic failure, and HCC. It is not practical to use these clinical endpoints as the primary treatment endpoints for clinical trials of novel investigative medicines because they usually take decades to develop. Therefore, in order to replace these prolonged, delayed results, clinical research have turned to surrogate markers and shorter-term, intermediate endpoints. Biochemical normalization of serum alanine aminotransferase virological HBV DNA Since HBsAg seroclearance is linked to long-lasting off-treatment improvements in clinical outcomes, HBsAg loss is seen to be the most significant of these surrogate endpoints.
Furthermore, it has been demonstrated that HBsAg reduction, especially in cirrhosis patients, provides an extra clinical advantage over HBV DNA suppression alone (lower incidence of HCC and hepatic decompensation). Additionally, HBsAg seroclearance can be easily measured using widely accessible and standardized tests. Because of the persistence of both HBV DNA that is directly integrated into the host genome and covalently closed circular DNA (cccDNA), a stable, nonintegrated form of viral DNA found in the hepatocyte nucleus, current therapies (pegylated interferon alfa [pegIFNa] and nucleoside analogs [NAs] are ineffective. While immune systems can quiet and/or degrade cccDNA during acute infection, persistent infection results in defective immune responses that are difficult for existing treatments to repair. With over 800,000 deaths from CHB each year, new limited-duration medications are required in addition as an expansion of current antiviral treatments to lessen the disease's devastating effects on world health.
Management of On-Treatment and Off-Treatment ALT Flares and Virological Rebound
Periodically over the course of an infection and while receiving pegIFNa and NAs there may be an abrupt rise in blood ALT levels or an ALT flare. ALT flares are a concern with novel therapies because they have the potential to exacerbate the underlying liver injury or cause hepatic decompensation, despite the fact that the majority of ALT flares are believed to be a host immune response against HBV-infected hepatocytes.
Additionally, there are unclear management approaches for these events. ALT flares may be immune-mediated, such as an agent that either directly or indirectly enhances host immune recognition, subsequent to immune-mediated hepatitis caused by immune checkpoint inhibitors and the destruction of HBV infected hepatocytes, including a host cytolytic response following a rapid decrease in viral replication and antigen, depending on the mechanism of action of novel therapies. Destruction of HBV-infected hepatocytes, including a host cytolytic response following a sharp decline in viral replication and HBV antigen expression; virally mediated, resulting in a resurgence of intrahepatic viral spreading and an increase in viral replication that may be caused by viral escape, reactivation, or resistance to an antiviral agent, such as a CAM, or by stopping NA before HBsAg loss and DILI as a result of direct or indirect hepatotoxicity of the study drug, such as CAM and inarigivir [16-19].
Figure 4: Guidance on treatment endpoints and study design for clinical trials aiming to achieve cure in chronic hepatitis B and D.
Conclusion
The design and interpretation of clinical trials are fundamental to advancing medical knowledge, ensuring patient safety, and developing effective treatments. A well-structured trial design-incorporating randomization, blinding, appropriate sample sizes, and rigorous statistical analysis-minimizes bias and enhances the reliability of results. Interpreting clinical trial outcomes requires careful consideration of statistical significance, clinical relevance, and potential biases. Regulatory approval and real-world implementation depend on not only positive findings but also long-term safety and cost-effectiveness. Ultimately, clinical trials serve as the backbone of evidence-based medicine, guiding healthcare decisions and improving patient outcomes. However, ethical considerations, patient diversity, and real-world applicability should always be factored into trial design and interpretation to ensure meaningful and equitable advancements in medical science.
Declarations
Acknowledgement
I would like to show my sincere gratitude towards IPS Academy College of Pharmacy for providing the necessary requirements and facilities throughout the study. Lastly, I would like to express my appreciation to all my colleagues and peers for their helpful discussions and moral support, which contributed to the success of this work.
Conflict of Interest
The authors declare no conflict of interest.
References
- Vogel, J. M., Pollack, B., Spier, E., McCorkell, L., Jaudon, T. W., et al. (2024). Designing and Optimizing Clinical Trials for Long COVID. Life Sciences, 122970.
Publisher | Google Scholor - Zaki, M., O'Sullivan, L., Devane, D., Segurado, R., McAuliffe, E. (2022). Factors Influencing the Statistical Planning, Design, Conduct, Analysis and Reporting of Trials in Health Care: A Systematic Review. Contemporary Clinical Trials Communications, 26:100897.
Publisher | Google Scholor - Gewandter, J. S., Chaudari, J., Iwan, K. B., Kitt, R., As-Sanie, S., et al. (2018). Research Design Characteristics of Published Pharmacologic Randomized Clinical Trials for Irritable Bowel Syndrome and Chronic Pelvic Pain Conditions: An ACTTION Systematic Review. The Journal of Pain, 19(7):717-726.
Publisher | Google Scholor - Kris-Etherton, P. M., Petersen, K. S., Lamarche, B., Karmally, W., Guyton, J. R., et al. (2024). The Role of Nutrition-Related Clinical Trials in Informing Dietary Recommendations for Health and Treatment of Diseases. Journal of Clinical Lipidology.
Publisher | Google Scholor - Bloom, P. P., Chung, R. T. (2024). The Future of Clinical Trials of Gut Microbiome Therapeutics in Cirrhosis. JHEP Reports, 101234.
Publisher | Google Scholor - Zhang, Y., You, P., Liu, R., Lu, Y., Li, J., et al. (2024). Artificial Intelligence in Clinical Trials of Lung Cancer: Current and Future Prospects. Intelligent Oncology.
Publisher | Google Scholor - Schultz, A., Saville, B. R., Marsh, J. A., Snelling, T. L. (2019). An Introduction to Clinical Trial Design. Paediatric Respiratory Reviews, 32:30-35.
Publisher | Google Scholor - Pocock, S. J. (1985). Current Issues in The Design and Interpretation of Clinical Trials. Br Med J (Clin Res Ed), 290(6461):39-42.
Publisher | Google Scholor - Pocock, S. J. (1997). Clinical Trials with Multiple Outcomes: A Statistical Perspective on Their Design, Analysis, And Interpretation. Controlled Clinical Trials, 18(6):530-545.
Publisher | Google Scholor - Ceelen, W., Soreide, K. (2023). Randomized Controlled Trials and Alternative Study Designs in Surgical Oncology. European Journal of Surgical Oncology, 49(8):1331-1340.
Publisher | Google Scholor - Maltais, F., Vogelmeier, C. F., Kerwin, E. M., Bjermer, L. H., Jones, P. W., et al. (2022). Applying Key Learnings from The EMAX Trial to Clinical Practice and Future Trial Design in COPD. Respiratory Medicine, 200:106918.
Publisher | Google Scholor - McDermott, M., Meah, M. N., Khaing, P., Wang, K. L., Ramsay, J., et al. (2024). Rationale and Design of SCOT-HEART 2 Trial: CT Angiography for The Prevention of Myocardial Infarction. Cardiovascular Imaging, 17(9):1101-1112.
Publisher | Google Scholor - Bogaerts, J., Sydes, M. R., Keat, N., McConnell, A., Benson, A., et al. (2015). Clinical Trial Designs for Rare Diseases: Studies Developed and Discussed by The International Rare Cancers Initiative. European Journal of Cancer, 51(3):271-281.
Publisher | Google Scholor - Best, A. M., Lang, T. A., Greenberg, B. L., Gunsolley, J. C., Ioannidou, E., et al. (2024). The OHStat Guidelines for Reporting Observational Studies and Clinical Trials in Oral Health Research: Explanation and Elaboration. Journal of Oral and Maxillofacial Surgery.
Publisher | Google Scholor - Soler, M., Lu, E., Masch, R., Alfaro, K., Anderson, J. R., et al. (2023). Affordable Cancer Technologies: Lessons Learned from The Design and Implementation of Two Randomized Clinical Trials to Develop Innovative Treatments for Cervical Precancer. Contemporary Clinical Trials Communications, 36:101224.
Publisher | Google Scholor - Sutherland, L., Carter, L. (2024). Sex As a Biological Variable in Early-Phase Oncology Clinical Trials: Enhancing the Path to Personalised Medicine. Heliyon, 10(12).
Publisher | Google Scholor - Mauri, L., D’Agostino Sr, R. B. (2017). Challenges in the Design and Interpretation of Noninferiority Trials. New England Journal of Medicine, 377(14):1357-1367.
Publisher | Google Scholor - Bhide, A., Shah, P. S., Acharya, G. (2018). A Simplified Guide to Randomized Controlled Trials. Acta Obstetricia et Gynecologica Scandinavica, 97(4):380-387.
Publisher | Google Scholor - Finkelstein, A., Zhou, A., Taubman, S., Doyle, J. (2020). Health Care Hotspotting-A Randomized, Controlled Trial. New England Journal of Medicine, 382(2):152-162.
Publisher | Google Scholor - Harrer, S., Shah, P., Antony, B., Hu, J. (2019). Artificial Intelligence for Clinical Trial Design. Trends in Pharmacological Sciences, 40(8):577-591.
Publisher | Google Scholor