Research Article
Isolation, Identification and Antimicrobial Susceptibility Patterns of Salmonella and Co-Infection with Cryptosporidium from Diarrheic and Non-Diarrheic Calves in and Around Haramaya District and Harar Town Dairy Farm, Eastern Ethiopia
1Department of Veterinary Public Health, College of Veterinary Medicine, Haramaya University, Dire Dawa, Ethiopia.
2Department of Veterinary Microbiology, College of Veterinary Medicine, Haramaya University, Dire Dawa, Ethiopia.
*Corresponding Author: Daniel Tamirat Weldemariam, Department of Veterinary Public Health, College of Veterinary Medicine, Haramaya University, Dire Dawa, Ethiopia.
Citation: Weldemariam DT, Ali MA. (2026). Isolation, Identification and Antimicrobial Susceptibility Patterns of Salmonella and Co-Infection with Cryptosporidium from Diarrheic and Non-Diarrheic Calves in and Around Haramaya District and Harar Town Dairy Farm, Eastern Ethiopia, International Journal of Biomedical and Clinical Research, BioRes Scientia Publishers. 6(5):1-19. DOI: 10.59657/2997-6103.brs.26.122
Copyright: © 2026 Daniel Tamirat Weldemariam, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: January 22, 2026 | Accepted: March 16, 2026 | Published: March 24, 2026
Abstract
Background: Calf diarrhea is a major health problem in young cattle and results in considerable economic losses to the dairy industry worldwide. Salmonella is an important bacterial cause of calf diarrhea and is often associated with antimicrobial resistance and co-infection with other enteric pathogens such as Cryptosporidium. This study aimed to determine the prevalence of Salmonella, its antimicrobial susceptibility patterns, and its co-infection with Cryptosporidium in diarrheic and non-diarrheic calves in Eastern Hararghe, Ethiopia.
Methods: A cross-sectional study was conducted from November 2023 to May 2024 in intensive and extensive dairy farms in and around Haramaya and Harar towns, Eastern Hararghe. Fecal samples were collected from 384 calves and examined for Salmonella using standard bacteriological techniques and for Cryptosporidium using parasitological methods. Antimicrobial susceptibility testing was performed using the disk diffusion method. Data were analyzed using SPSS version 20, and logistic regression and chi-square tests were applied to assess associations between risk factors and infection.
Results: Salmonella was isolated from 63 of 384 calves, giving an overall prevalence of 16.4%. The prevalence was significantly higher in diarrheic calves (38.8%) than in non-diarrheic calves (6.7%). Multivariable logistic regression analysis showed that management system (OR = 2.9, P = 0.002), age category (OR = 4.5, P < 0.001), and fecal consistency (OR = 8.7, P < 0.001) were significantly associated with Salmonella infection. Co-infection with Cryptosporidium was detected in 63.5% (40/63) of Salmonella-positive calves and was significantly associated with age, fecal consistency, body condition score, and management system (P < 0.05). Multiple antimicrobial resistance was observed in 95.2% of the isolates. High resistance was recorded against nalidixic acid (95.2%), amoxicillin (80.9%), and cefoxitin (73.0%), while all isolates were susceptible to ciprofloxacin (100%) and most to chloramphenicol (87.3%).
Conclusions: The study demonstrated a high prevalence of Salmonella infection and a substantial level of multidrug resistance among isolates from calves in the study area. Management system, age, and fecal consistency were important risk factors for infection, and co-infection with Cryptosporidium was common among Salmonella-positive calves. Improved farm management practices, rational use of antimicrobials, and integrated control strategies targeting multiple enteric pathogens are essential to reduce calf diarrhea and its impact on dairy production.
Keywords: salmonella; antimicrobial resistance; cryptosporidium; calf diarrhea; risk factors
Introduction
Background of The Study
Ethiopia, located in Eastern Africa, is predominantly an agricultural country, where animal production is practiced across all ecological zones (Tegegne and Crawford, 2000). The total cattle population is estimated at about 53.99 million, of which 9.77% are under six months of age and 7.64% are between six months and one year (CSA, 2013). Indigenous breeds account for 98.95% of the cattle population, while hybrid and exotic breeds represent approximately 0.94% and 0.11%, respectively (CSA, 2013).
The health of replacement calves is a critical component of dairy farm profitability, as heifer calves represent the foundation of future milking herds (Fox, 2007; Razzaque et al., 2009). High calf morbidity and mortality result in substantial economic losses due to treatment costs, reduced lifetime productivity, impaired survivorship, and limitations on herd expansion and genetic improvement (Mellado et al., 2014). Calf diseases arise from complex interactions among management practices, environmental conditions, infectious agents, and host factors (Klein-Jöbstl et al., 2014).
Neonatal calf diarrhea (NCD) is among the most common and economically devastating diseases of young calves worldwide (Cho and Yoon, 2014). It is a multifactorial syndrome involving infectious agents as well as non-infectious factors related to calf immunity, nutrition, housing, hygiene, and management (Izzo et al., 2011). Due to this multifactorial nature, effective control of NCD remains challenging (Cho and Yoon, 2014). Infectious diarrhea is the leading cause of morbidity and mortality in neonatal dairy calves and is commonly associated with viruses (rotavirus and coronavirus), protozoa (Cryptosporidium parvum), and bacteria, particularly enterotoxigenic Escherichia coli (ETEC) and Salmonella spp. (Izzo et al., 2011).
Although co-infections are frequently observed in diarrheic calves, single-agent infections may also occur. The prevalence of these pathogens varies with farm location, management practices, and herd size (Cho and Yoon, 2014). Identification of causative agents during outbreaks is essential for implementing targeted preventive strategies, including vaccination and biosecurity measures, and for identifying risk factors and sources of infection (Izzo et al., 2011).
Salmonella enterica colonizes the intestinal tract of both adult cattle and calves; however, infection is most commonly observed during the first three months of life and often results in severe clinical disease (Fossler et al., 2005). The most prevalent serovars in cattle are S. enterica serovar Typhimurium and serovar Dublin, which typically cause acute enteritis and systemic disease, respectively (Cho and Yoon, 2014). Diarrhea associated with salmonellosis is often watery or mucoid and may contain blood and fibrin (Fossler et al., 2005). Infected calves may shed the organism intermittently for prolonged periods, and up to 5% of recovered animals may become asymptomatic carriers (Jay, 2003; Cho and Yoon, 2014). These carriers serve as reservoirs of infection for other animals and humans, highlighting the importance of detecting Salmonella in fecal samples for both clinical diagnosis and epidemiological control (Warnick et al., 2003).
Cryptosporidiosis is also a major cause of morbidity in neonatal calves (Szonyi et al., 2012). Although the disease is often mild and self-limiting, it may lead to severe diarrhea, dehydration, and mortality, particularly in cases of co-infection with other enteric pathogens (Santin et al., 2008; Wang et al., 2010). Infected calves can shed large numbers of infective oocysts for 3-14 days, including during asymptomatic periods (Santin et al., 2008). Clinical signs include watery or bloody diarrhea, malabsorption, dehydration, and poor growth (Wang et al., 2010).
Cryptosporidium oocysts are highly resistant to environmental conditions and many commonly used disinfectants, enabling prolonged survival under a wide range of temperatures (-22°C to 60°C). The infectious dose for neonatal calves may be as low as 17 oocysts, making control on farms particularly difficult (Thomson et al., 2017). The disease is usually self-limiting when adequate colostral immunity is present, calves are maintained under good hygienic conditions, and supportive therapy is provided, especially in the absence of concurrent infections (Thomson et al., 2017).
Current treatment strategies for neonatal calf diarrhea rely primarily on fluid therapy and antimicrobial agents. However, the indiscriminate use of antimicrobials has contributed to the emergence of antimicrobial resistance, particularly among Salmonella isolates from pre-weaned dairy calves. This growing resistance poses a significant challenge to effective disease management and public health (Izzo et al., 2011).
Statement of The Problem
Diarrhea (scouring) is a complex multifactorial disease and the leading cause of neonatal calf mortality during the first weeks of life, with risk decreasing as calves age. Salmonella spp. is among the major bacterial pathogens responsible for calf diarrhea and cause substantial economic losses due to high morbidity and mortality in the cattle industry worldwide. In addition to Salmonella, several other infectious and non-infectious agents, including rotavirus, Escherichia coli, Giardia duodenalis, and Cryptosporidium spp., contribute to diarrheal disease in young livestock. Co-infections involving multiple pathogens are common, yet little is known about the interactions between these agents and their effects on disease severity and outcomes.
In Ethiopia, calf morbidity and mortality associated with diarrhea remain major challenges to dairy production. Despite this, coordinated surveillance systems and comprehensive assessments of the etiological complexes involving bacterial pathogens and protozoan parasites are limited. In particular, information on the isolation, identification, and antimicrobial susceptibility patterns of Salmonella spp., as well as their co-association with Cryptosporidium infection in diarrheic calves, is scarce. Therefore, research focusing on these pathogens is essential for improving disease control strategies and reducing economic losses during the critical early months of calf life.
Objectives
General Objectives: The overall objective of this study is to isolate and identify Salmonella spp., determine their antimicrobial susceptibility patterns, and assess their co-association with Cryptosporidium spp. in diarrheic and non-diarrheic calves.
Specific Objectives
- To isolate and identify Salmonella spp. from diarrheic and non-diarrheic calves.
- To determine the antimicrobial susceptibility patterns of Salmonella isolates.
- To assess the co-occurrence of Salmonella spp. and Cryptosporidium spp. in diarrheic and non-diarrheic calves.
- To identify risk factors associated with Salmonella infection in calves.
Literature Review
Bovine Salmonellosis
Etiology
Salmonellae are Gram negative, short plump shaped rods, nonperforming, no capsulated, aerobic and facultative anaerobic organisms and classified under the family Enterobacteriaceae (OIE Manual, 2006). There are 2 recognized species within the genus: S enteric and Salmonella bongori. S enteric can be further divided into 6 subspecies, S enterica subspecies enterica being the most relevant in dairy cattle (Smith, 2009). Ninety-nine percent of human and animal infections are caused by the subspecies enterica. Within this subspecies more than 2 600 serotypes have been described, based on serological reactions to cell wall lipopolysaccharide (O), flagellar (H) and capsular (VI) antigens (Sanderson & Nair, 2012).
Salmonella serotypes have different abilities to infect and cause disease, and are sometimes referred to as host generalists (causing infections in many hosts e.g. S. Typhimurium and S. Enteritidis), host-adapted (primarily associated with one host e.g. S. Dublin in cattle and S. Choleraesuis in pigs), and host-restricted (associated with one host only e.g. S. Typhi in man and S. Gallinarum in poultry) (Sanderson & Nair, 2012).Many serovars can infect cattle; Typhimurium and Dublin serovars are the most common. S. typhimurium is frequently associated with enteric disease in calves less than 2 months of age, and S. Dublin is associated with young and adult cattle and is more invasive than S. typhimurium (Mohler et al., 2009).
Salmonellae are chemoorganotrophic, with an ability to metabolize nutrients by the respiratory and fermentative pathway. An important characteristic of the Salmonella bacteria is that they are able to grow and multiply outside living host organisms, thus having greater survival chances than otherwise (Gray and Fedorka-Cray, 2002).
Epidemiology
The epidemiology of Salmonella is complex which often makes control of disease difficult. Epidemiological pattern of prevalence of infection and incidences of disease differ greatly between geographical area depending on climate, population density, land use farming practice, food harvesting and processing technologies and consumer habits. In addition, the biology of serovar differs so widely that Salmonella infection or Salmonella contamination are inevitably complex (Radostitis et al., 2007).
Salmonella Entritidis is the most prevalent species followed by S. typhimurium which are a worldwide distribution. Change in the relative frequency of serotypes can be observed over a short period of time. Some times within one or two years only limited number of serotypes is isolated from man or animals in a single region or country and the predominance of one or other can vary over a time. Some serotypes like S. entritidis and S. typhimurium are found worldwide in contrast to S. weltevreden which seems to be confined to Asia (Kemal, 2014). Sibhat et al., (2011) found the serovars Newport, Anatum and East bourne to be the most prevalent in Ethiopia. Salmonellae have a wide variety of domestic and wild animal hosts (Acha and Szyfres, 2001).
Pathogenesis
In both calves and adults, those factors that determine pathogenicity and whether or not clinical disease is seen include virulence of the serotype, dose of inoculum, degree of immunity (passive or adaptive) or previous exposure of host to the serotype, and other stressors currently affecting the host (Mohler et al .,2009).The most detailed studies of the pathogenesis of bovine salmonellosis infection come from the literature describing enteric infection via the oral route, mainly in calves(Mohler et al.,2008). Once ingested, Salmonella attaches to mucosal cells and is capable of destroying enterocytes. Attachment is increased if gastrointestinal stasis is present or the normal flora has been disturbed or is not yet established, as is the case in neonates (Smith, 2009).
The basic mechanism underlying Salmonella virulence includes the ability to invade the intestinal mucosa, multiply in lymphoid tissues, and evade host defense systems, leading to systemic disease. For Salmonella pathogenesis, the organism should be capable of invading Intestinal epithelial cells, surviving within macrophages, and causing enteropathogenicity (Reis BP et al., 2003). Salmonella colonizes M-cells, enterocytes, and tonsilar tissues following lymphoid tissue (e.g., tonsilar tissue) infection, Salmonella easily spreads throughout the whole body by invading mononuclear cells and phagocytes (Holt PS, 2000). Salmonella pathogenicity island 1 (SPI-1) and SPI-5 are known to influence the type III secretion system, and are mainly responsible for Salmonella-induced diarrhea in calves. SPI2 is involved in the second type III secretion system and is responsible for intracellular survival of the organism (Reis BP et al., 2003).
The virulence mechanisms of salmonellae are, therefore, composed of their ability to invade the intestinal mucosa, locate to and multiply within the lymphoid tissues, and to evade host defense mechanisms. Enterocolitis caused by Salmonella spp. is due to inflammation with subsequent maldigestion and malabsorption, and to a lesser extent from secretory mechanisms (Smith, 2009). Inflammation in the colon leads to the commonly observed fresh blood in the feces of both adults and calves. The diarrhea caused by Salmonella spp. is principally mediated by the host inflammatory reaction to the infection. To establish infection, enteropathogenesis such as Salmonella must first be able to overcome those host factors that resist colonization of the gut, principle among these being a fairly dense gut microbiota, which secrete a variety of bacteriocins, antibiotics, and colicins that hinder enteropathogen growth (Stecher, 2015).
Clinical Signs
One or more of the syndromes may occur simultaneously in an outbreak of salmonellosis in a heard, or even in a single affected animal. Salmonella Dublin is more often the cause of septicemia than Salmonella Typhimurium. In Calves clinical disease is most common 2-6 week of age. Calves are more likely than adult animals to suffer from septicemic infections and may develop per acute, acute or chronic salmonellosis. The per acute disease is usually a septicemic condition, it is often fatal and calves may die suddenly without premonitory sign. Some, however, develop enteritis and diarrhea in addition to septicemia. When the course of septicemia is protracted, sign of hepatitis, pneumonia, meningoencephalitis, polyarthritis and osteomyelitis may develop (Luciana et al., 2012).
Acute enteric salmonellosis is the most common syndrome of salmonellosis encountered in calves. Following an incubation period, which may vary from one to five days, calves develop high fever, in appetence, lethargy, diarrhea, and frequently a serious nasal discharge followed by a slight cough and scouring may only occur terminally. The feces of affected calves have foul smelling, putty-like consistency, and contained large amount of mucous, sloughed mucosa and flecks of blood. At this time body temperature may be normal or subnormal. The mortality rates are 5 to 10% and may reach as high as 75% in purchased calves (Wray and Davies, 2000).
Chronic salmonellosis in calves is characterized by un thriftiness, long and scruffy hair coats and stunting. Diarrhea is not always present and sign of chronic pneumonia with persistent coughing may occur. The calves that survive either per acute, acute or chronic disease may develop pneumonia, meningoencephalitis, purulent polyarthritis, and osteomyelitis of the vertebrae and bones of distal parts of limbs, resulting in lameness, paresis or even almost complete paraplegia. Dry gangrene of the skin of the lower limbs and the tips of ear and tail, resembling ergotism, is rarely encountered (Alemu, 2007).
Salmonellosis is very variable and, in some animals, particularly in very young animal’s rapid multiplication occurs both in intestine and systemically associated with poor absorption of specific immunoglobulin G from colostrum or with calves receiving in sufficient or no colostrum (L. Plym et al., 2006). More severe symptom may occur in people who are at high risk like those extreme age groups (the young because their immune system are immature and the elderly because the immune system are declining), person with decreased gastric acidity (because gastric acid is the first line of defense for the ingested Salmonella), person with altered gastric intestinal bacteria (including those taking broad spectrum antibiotics, purgatives or who have had bowl surgery) and person taking opiate drug in which the bowl movement is decreased. In these highest risk groups of people Salmonella may invade beyond the gastrointestinal tract (GIT) to cause severe systematic illness (Kemal, 2014).
Source of Infection and Transmission
The dominating route of infection is fecal to oral (La Ragione et al., 2013). Cattle herds constitute an integrated part of the environment (Wales & Davies, 2012). Surface water, pastures, and forage may be contaminated by slurry, sewage sludge, or irrigation water containing salmonella bacteria (Ruzante et al., 2010). The bacteria can survive and multiply in the environment, manure, and effluents under suitable conditions (Lahti, 2010).
For transmission between herds, purchase of infected animals is an important factor (van Schaik et al., 2002). Mixing of animals at dealers or on transports also pose a risk. Indirect spread between herds occurs, e.g. through shared pastures (van Schaik et al., 2002), spread of manure from other herds (Veling et al., 2002b), and shared equipment and visitors. Rodents, arthropods, birds and wild animals can serve as vectors, and occasionally as reservoirs, for salmonella (Wales & Davies, 2012).
Spread within herds largely depends on management routines and herd hygiene (Belluco et al., 2015). On-farm feed storages are often open and exposed to contamination by rodents, wildlife and even manure. For S. Dublin, the calving area has been identified as an important source of spread (Fossler et al., 2005b; House & Smith, 2004) due to excretion of bacteria from the cow or other adult cattle, leading to infection of the susceptible calves. Even though the dominating route of infection is oral, for S. Dublin, other less likely transmission routes identified are aero genic, intrauterine, and conjunctival. Occasionally, calves can also be infected by ingestion of milk from cows excreting salmonella into the milk (La Ragione et al., 2013).
Most Salmonella infection in farm animals are likely to acquire from animals of the same species, especially in the case of the host adapted serovars. In adult cattle there are important differences in the behavior of S. Dublin and S. typhimurium. Those animals which recover from S. Dublin infection may become persistent excretes, shedding up to 106 organisms per gram of faces daily. Other herd may harbor infection and excrete the organisms only when stressed particularly at parturition. Aerosol transmission has long been suggested as a means by which Salmonella may be transmitted and experimental infection of calves by aerosol has been reported recently. In addition, pasture contamination results when flooding occurs and there are many reports of clinical case in adult cattle arising from grazing recently flooded pasture (Kemal, 2014).
Diagnosis
Diagnosis is based on the isolation of the organism either from tissues collected aseptically at necropsy or from feces, milk, blood, rectal swabs or environmental samples (Acha and Szyfres, 2001; OIÉ, 2000). When infection of the reproductive organs or conceptus occurs, it is necessary to culture fetal stomach contents, placenta and vaginal swabs and, in the case of poultry, egg contents. However, salmonellosis is particularly difficult to determine in clinically normal carrier animals.
Salmonellae may be isolated by a variety of techniques, which may include pre-enrichment in non-selective medium to resuscitate sub-lethally damaged salmonellae, enrichment media that contain inhibitory substances to suppress non-Salmonella organisms, and selective plating agars to differentiate salmonellae from other Enterobacteriaceae (OIÉ, 2000; ISO 6579, 1998).
Various biochemical and serological tests can be applied to the pure culture to provide a definitive confirmation of an isolated strain. Salmonellae possess antigens designated somatic (O), flagellar (H) and virulent (Vi), which may be identified by specific typing sera, and the serovar may be determined by reference to the antigenic formulae in the Kauffman-White scheme. A number of serological tests, such as the serum agglutination test (OIÉ, 2000) and indirect enzyme-linked immunosorbent assays have been developed for diagnosis of Salmonella. Serological tests are useful for the identification of infected herds but are inadequate for the identification of persistently infected animals (OIÉ, 2000).
Treatment and Control
In animal treatment supportive treatment with intravenous fluid is necessary for patients that have anorexia, depression, significant dehydration. Individual patient may be treated aggressively following acid base and electrolyte assessment. Oral fluid and electrolyte may be somewhat helpful and much cheaper than IV fluid for cattle demand to be mildly or moderately dehydrated. The effectiveness of oral fluid may be somewhat compromised by malabsorption and maldigestion in salmonellosis patient but still should be considered useful. Cattle that are willing to drink can have specific electrolyte (NaCl, KCl) added to drinking water to help correcting electrolyte (Kemal, 2014).
The implementation of broad prophylactic strategies that are efficacious for all Salmonellae may be required in order to overcome the diversity of Salmonella serovars present on farms, and the potential for different serovars to possess different virulence factors (Mohler et al., 2009). Early treatment is essential for septicemic salmonellosis but there is controversy regarding the use of antimicrobial agent for intestinal salmonellosis. Oral antibiotic may alter the intestinal micro flora and interfere with competitive antagonism and prolong shading of the organism. There is also a concern that antibiotic resistance strain of Salmonella selected by oral antibiotic may subsequently infect human. Antibiotic such as ampicillin or cephalosporin led to lyses of bacteria with release of endotoxin. NSAID may be used to reduce the effect of endotoxemia (Davison, 2005).
Vaccination of calves at 1-3 weeks of age with a modified aromatic defendant S. Dublin bacterium have detectable anti-lipopolysaccharide immunoglobins after immunization. Safe live oral vaccine against S. Typhimurium and S. Dublin has been constructed and shown to control protection against experimental infection with virulent wide type strain of the organisms. A virulent S. Choleraesuis vaccine is efficacies experimentally against salmonellosis due to S. Dublinin calves to protect young calves. The best program is to vaccinate the cow during pregnancy which will give passive protection to calves for 6 weeks (Radostitis et al., 2007).
Generally, it is agreed that supportive therapy and good nursing are important. These include oral or parenteral re-hydration, correction of electrolyte balance and stabilization of acid base equilibrium. Both live and attenuated vaccines produced from rough strain in bacteria commercially. There is some evidence that inactivated bacterins can induce a lower level of protection (Danielle, 2006).
Cryptosporidiosis in Bovine
Etiology and Epidemiology
Cryptosporidia are protozoan parasites historically classified as belonging to phylum Apicomplexa, class Coccidea, together with e.g. Eimeria, Isospora and Toxoplasma. This classification was based on similarities in life cycles, such as invasion of host epithelial cells (Fayer, 2008). Cryptosporidium parvum affects many species of mammals that include human and it has considerable economic significance in farm animals, especially in the ruminant (Ryan and Hijjawi, 2015). Modern molecular methods are used to analyze the genetic profile of this parasite and new sub-species and genotypes of the parasite have been identified. Currently, 28 species have been recognized (Hotchkiss et al., 2015).
The prevalence of C. hominis and C. parvum varies in different regions of the world. Indeed, C. hominis by far more prevalent in North and South America, Australia and Africa, while C. parvum causes more human infections in Europe, especially in the UK. The only documented report of Cryptosporidium infection in Ethiopia is by that of Wudu (Wudu, 2004) who reported 6.7% prevalence in calves after a longitudinal study conducted to determine the cause of calf morbidity and mortality. However, full account on the epidemiology of the parasite and the species encountered were not provided (Learmonth et al., 2004).
Cryptosporidium oocyst is resistant to many unfavorable environmental conditions and can be well preserved under the cold and wet environment surrounding the calving grounds (Hoar et al., 2001). The presence of high concentrations of the parasite when cattle are overcrowded is also related to higher rates of infection. Another risk factor identified for increasing probability of calves shedding oocyst is frequent bedding changes as personnel and equipment used for removal of the bedding can actually become a vehicle for spreading the infection (Sischo et al., 2000).
Risk Factors
There is a significant association between age and risk of infection with Cryptosporidium. Cryptosporidiosis due to C. parvum is predominantly a problem of neonate animals with maximum rate of excretion of oocysts between the age of 4 and 21 days. According to (Nguyen et al. (2007), age appeared to be an important factor that influences the occurrence of the Cryptosporidium. The calves under 3 months are at higher risk of infection compared to the older ones. The higher prevalence in this age group can be attributed to the fact that these age groups are highly susceptible to the disease because of the immature immune system of the animal at this age. Kvac et al., (2006) explained that the animal is becoming resistant with age due to the immune development through time. Although exceptions occur, older animals generally develop poor infections, even when unexposed previously to this parasite (Xiao et al., 2004) age-related resistance, unrelated to prior exposure, has been observed in lambs but not calves (Radostits et al., 2006). The common occurrence of cryptosporidiosis in young animals reflects their susceptibility to infection with a low number of oocysts and common exposure to oocysts (Fayer, R. and L. Xiao, 2008).
Oocysts are resistant to most disinfectants and can reportedly remain viable for about 18 months in a cool, damp or wet environment, can survive for several months in soil and slurry, but are susceptible to desiccation and temperatures above 60°c. The infectivity of the oocysts can be destroyed by ammonia, formalin, freeze-drying and exposure to temperatures below O°C (32°F) and above 65°C (149°F). Ammonium hydroxide, hydrogen peroxide, chlorine dioxide, 10% formalin saline and 5% ammonia are effective in destroying the infectivity of the oocysts. The infectivity of oocysts in calf feces is reduced after 1-4 days of drying (Radostits et al., 2006).
Diagnosis
Several methods exist to detect Cryptosporidium in fecal samples. Among them the most common method is microscopy for the detection of oocysts. Fecal samples can be examined directly on slides or after concentration either by flotation or sedimentation to remove fecal debris or to concentrate the number of oocysts; the detection of oocysts in animals with low numbers of oocysts is facilitated (Fayer and Xiao, 2007).
Visualization of Cryptosporidium oocysts by microscopy most commonly done by direct smear and without any staining and by the modified Ziehl-Neelsen stain under light microscopy, whereby the oocysts stain purple with blue background. Immunofluorescence staining techniques using monoclonal antibodies against the oocyst wall antigen under epifluorescence microscopy are also useful. Immunofluorescent antibody-based (IFA) procedures have a high sensitivity, but still the easier and cheaper traditional staining methods such as the Ziehl-Neelsen stain are widely used, despite their lower sensitivity (Caccio and Widmer, 2014).
A differential diagnosis list for cryptosporidiosis includes Giardia, Isospora, microsporidia, Salmonella, rotavirus and other diarrheal diseases. Cryptosporidiosis can be diagnosed by finding C. parvum after a fecal flotation in either sucrose or zinc sulfate solutions. The mature oocysts are 4-5 μm in diameter and contain four thin, flat, motile sporozoites. Cryptosporidium parvum can also be detected through an acid-fast staining. Oocysts are not shed continuously and repeated sampling may be necessary. The oocysts appear red after the staining. Immunofluorescence can also be used to detect Cryptosporidium in feces. Finally, cryptosporidiosis can be detected by stained biopsy/necropsy specimens or fresh intestinal scrapings (CFSP H, 2011).
Materials and Methods
Study Areas
The study was carried out in two selected areas of eastern Hararghe Ethiopia, which are Haramaya and Harar towns. Haramaya is located in Oromia Regional State of Eastern Hararghe Zone, at500 km East of Addis Ababa, the capital city of Ethiopia and 14 km from Harar town to west (HADB, 2009). Geographically, Haramaya district is situated at 410 51l 58ll N latitude and 90024l10llS longitude. The area is located at 2000 m altitude above sea level and receives 492 mm cube average annual rain fall ranging from 118-866 mm cube with a bimodal rain fall of short rain season ranging from February to May and long rain season ranging from June to September. The rainfall reaches peak in mid-August and April. The max and min temperatures are 24 0c and 90c respectively. Haramaya district has livestock population of 114062 cattle, 109765 goats and 730745 sheep (HWAO, 2018). Harar town is found at 525 Km East of Addis Ababa the capital city of Ethiopia. The town is located at 42.04 - 42.22 °E Longitude and 100-250 °N Latitude. It has an average altitude of 1780 m above sea levels and average temperature of 22.65 °c. The annual rain fall on average is 700 mm (HRAO, 2018).
Study Animals
The study was conducted on diarrheic and non-diarrheic calves which were managed under intensive and extensive management systems in study areas. The study animals include both sexes male and female aged up to one year. All selected farms were visited before and during the study period which enable us to observe health status, body condition and management systems. Any calve that was in antimicrobial treatment regiments (i.e., for diarrhea case) during the data collection period was excluded from sampling.
Sample Size Determination and Sampling
The sample size required for the current study was determined according to Thrusfield (2007) with the expected prevalence of 50%, 5 percentage absolute precision at 95 percentage confidence level as follows:
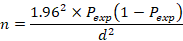
Where n = required sample size; Pexp= expected prevalence, d = desired absolute precision.
Therefore, 384 calves were selected from the target population in the study areas.
Study Design
Cross sectional study design was employed from November 2021 to May 2022 to achieve the objective of the study in the study area.
Isolation and Identification of Salmonella
Pre-enrichment in Non-Selective Media: In a first step, samples were diluted in a pre-enrichment medium, followed by homogenization in a stomacher for 1 minute. The reason for stomaching is that Salmonella spp. is not homogeneously distributed in faeces, especially when only low numbers of Salmonella spp. are shed (Funk et al.,2000). Processed samples in appropriate amount of buffered peptone water (BPW) (1:9) were incubated for 18h ± 2h at 37oC ± 1oC.
Enrichment in Selective Media: Enrichment in selective liquid media was done by transferring 0.1 ml of culture obtained from the non-selective pre-enrichment media to a tube containing 10ml of Rappaport vassiliads Salmonella enrichment broth then incubated at 42 OC for 24± 3hrs (ISO 6579; 1998: ISO 6579; 2002).
Plating Media: Xylose-lysine Deoxycholate agar (XLD) and Brilliant Green Agar (BGA) plates were used for plating out and identification purpose. A loop full of inoculums from Rappaport vassiliads Salmonella enrichment broth Culture was streaked on to XLD and BGA agar plates and the Table 1Table 2inoculated plates were incubated at 37OC for 24 ± 3h. After proper incubation, plates were examined for the presence of typical Salmonella colonies. Typical colonies of Salmonella grown on XLD medium produce hydrogen sulphide (H2S) and have black (H2S) centre and a lightly transparent zone of reddish color due to the color change of the indicator while on BGA medium they become Red due to non-lactose-fermenting. Salmonella H2S negative variants (e.g. S. Paratyphi A) grown on XLD agar are pink with a darker pink center where as lactose- positive Salmonella are yellow with or without blackening (ISO 6579; 2002).
Biochemical Tests: For confirmation, at least five presumptive colonies were selected from every selective plating media. Pure cultures obtained from nutrient agar were used for biochemical confirmation. Triple sugar iron agar (TSI) slants were inoculated from pure culture by streaking the slant and stubbing the butt incubated at 37OC ± 1OC for 24 ± 3h loosely capped to maintain aerobic condition and to prevent excessive production of H2S production. Typical Salmonella culture in TSI agar show alkaline (red) slants, and acid (yellow) butts with gas (bubbles) formation and (in about 90% of the cases) formation of hydrogen sulphide (blackening of the agar). Alkaline reaction (purple color) both in the slant and the butt superimposed with H2S after incubation indicates a typical positive reaction for Salmonella in lysine iron agar (ISO 6579; 2002). Pure isolates were inoculated on urea agar and Simmons’s citrate agar by streaking the slant. Both of the inoculated tubes were incubated at 37OC ± 1OC for 24 ± 3h (ISO 6579, 2002) and change in the incubated media were interpreted for Salmonella after the end of incubation following the guide line. Positive reaction in urea changes the color of phenol red to rose pink and latter to deep cerise due to liberation of ammonia as the result of splitting of urea. However, typical Salmonella colonies do not hydrolyse urea; therefore, the medium remain yellow (negative test) (ISO 6579; 2002). Salmonella colonies on Simmons’s citrate agar produce alkaline products using the medium as the sole carbon source hence deep blue color indicates positive reaction. The pure isolates were also inoculated in to tryptose soya broth (TSI) to determine the ability of an organism to spilt amino acid tryptophan to form the compound indole. For this an inoculated broth culture was incubated for 37OC ± 1OC for 24 ± 3h and Kovac’s reagent was added. In positive reaction pink colored ring is formed on the top of the broth due to reaction of indole with Kovac’s reagent. However typical Salmonella colonies due not hydrolyze amino acid tryptophan; therefore, the medium remain unchanged after addition of appropriate reagent (ISO 6579; 2002). Finally, pure cultures were inoculated in to MR- VP Medium (Buffered glucose broth) (glucose phosphate broth) incubated at 37OC ± 1OC for 24 ± 3h and broth culture in each incubated tube is divided in to two halves. One half is used for methyl-red test and the other is for Vogus-Proskauer test. Alpha-naphtol (5%) and potassium hydroxide (40%) were added to the medium prepared for vages-proskauer test to know where the organism utilize glucose though butylene glycol pathway and produce acetoin. If acetoin is present, the medium will turn pink-reddish in color. Methyl red reagent is added in another half. If the organism uses the mixed acid fermentation pathway and produces stable acidic end-products, the acids will overcome the buffers in the medium and produce an acidic environment in the medium. When methyl red is added, if acidic end products are present, the methyl red will stay red. Typical Salmonella isolates are VP negative and MR positive (ISO 6579; 2002).
Examination of Feces for Cryptosporidium Oocysts: Cryptosporidium oocysts will be detected by modified Ziehl-Neelsen staining method. Preparation of staining solutions and procedures will be according to the recommendations given by (OIE, 2O16). In this method a thin smear will be prepared directly from fresh facal samples and allowed to air dry. Then the slides are fixed with absolute methanol for 3minutes and stained with carbol fuchsine for 15 minutes. The slides then will be washed with tap water and decolorized with acid alcohol (1ml HCl and 99ml of absolute methanol) for 1015seconds. After washing the slides with tap water, the slides will be counter stained by 0.4% malachite green for 30 seconds, washed in tap water and allowed to air dry. The slides will then be examined for the presence of oocysts by scanning the slide systematically using the ×40 objective lens of a bright-field microscope. Confirmation of the presence of oocysts will be under the oil immersion objective lens with 100x magnification. Cryptosporidium spp. oocysts will appear red stained discoid or crescent shaped on a pale green background (Hendrix, 1998).
Antimicrobial Susceptibility Test of Salmonella: The antibiotic susceptibility tests of the Salmonella isolates were performed according to the Clinical and Laboratory Standards Institute guidelines (CLSI, 2012). Each isolate is tested for a series of eight antimicrobials. Amoxicillin (AML) (10 μg), ampicillin (AMP) (20 μg), cefoxitin (FOX) (30 μg), chloramphenicol (C) (30 μg), streptomycin (S) (10 μg), nalidixic acid (NA) (30 μg), ciprofloxacin (CIP) (5 μg),) and Erythromycin E 5 μg. Four to five well isolated colonies grown on nutrient agar were transferred onto tubes containing 5 ml of typtic soy broth (Oxoid LTD, England). The broth culture was inoculated at 35OC for 4hrs until it achieves or exceeds the 0.5 McFarland turbidity standard (appendix II). For these tubes which exceeded the turbidity standard, adjustment was made by adding the sterile saline solution to obtain turbidity visually comparable to the standard. Within 15 minutes after adjusting the turbidity of the inoculum’s suspension, a sterile swab was immersed in each of dilution suspension and swabbed uniformly over surface of two plates of muller Hinton agar (Oxoid LTD, England) for each inoculum. The plates were held at room temperature for 30 minutes to allow drying. Using sterile forceps, discs impregnated with known concentration of antimicrobials were dispensed onto the surface of Muller Hinton agar plates. The plates were incubated at 37OC for 20 hrs and examined for zone of inhibition. The diameter of zone of inhibition was recorded to the nearest millimeter, and classified as resistance, intermediate, or susceptible according to published interpretive chart (CLSI, 2012). The type of tested antimicrobials, their concentration in the discs and the zone of inhibition in deciding susceptibility are given in (Table 1).
Table 1: Antimicrobials and concentrations used to test susceptibility of Salmonella isolates (CLSI, 2012).
| Antimicrobials | Disk potency | Resistant | Intermediate | Sensitive |
| Ampicillin | Amp 10 μg | 13or less | 14-16 | 17 or more |
| Amoxicillin | AMC (20 μg) | 13 or less | 14-16 | 17or more |
| Ciprofloxacin | CIP 5 μg | 14 or less | 15-17 | 21or more |
| Chloramphenicol | C 30 μg | 12or less | 13-17 | 18 or more |
| Cefoxitin | FOX (30 μg) | 41or less | 15-17 | 18 or more |
| Erythromycin | E 5 μg | 13 or less | 14-22 | 18 or more |
| Naldixic acid | 30 μg | 13 or less | 14-18 | 19 or more |
| Streptomycin | 10 μg | 11 or less | 12-14 | 15 or more |
Data Storage, Management and Analysis: The occurrence of Salmonella was determined by descriptive statistics such as frequency and percentage. Chi square (χ2) analysis was used to measure the association between the different risk factors and occurrence of Salmonella in studied animals. In addition, the multivariable logistic regression analysis was used to calculate the association of variables with the occurrence of Salmonella. Before running a multivariable logistic regression analysis, all the variables were first screened for the presence of association using univariable logistic regression analysis. Then, those having a p-value of 0.25 and less were considered for the multivariable logistic regression. The overall effects of variables on the Salmonella occurrence were reported as statistically significant if p-value is less than or equals to 5 % (p less than 0.05).
Results
The Over All Prevalence of Salmonella
In the present study, Salmonella was recorded in 16.4 % (63/384) of sampled animals. The isolates were higher in Haramaya University farm (24.2%), 8/33) followed by Harar town farms (22.6%), 35/155) and Haramaya District (10.3), 20/195 (Table 2).
Table 2: The Over All Prevalence of Salmonella.
| Study Area | No. of Animals Examined | Salmonella Positive Animals (%) | χ2 value | p-value |
| Haramaya University | 33 | 8 (24.2) | 10.944 | 0.004 |
| Harar Town | 156 | 35 (22.4) | ||
| Haramaya District | 195 | 20 (10.3) | ||
| Total | 384 | 63 (16.4) |
Salmonella Occurrence and Associated Risk Factors
Chi-square analysis shows that: age category (χ2=21.211, P=0.000), different body condition scores (χ2=11.551, P= 0.004), fecal consistency (χ2=60.741, P=0.000) and management systems (χ2=10.931, P=0.000) were significantly associated with the occurrence of salmonella. However, no significant association was observed between the prevalence of Salmonella and sexes (χ2=0.183, p=0.381) (Table 3).
Table 3: Chi square analysis of Salmonella and associated risk factors.
| Variables* | No. of Animals Examined | No. of animals with Salmonella (%) | Chi Square Analysis | ||
| χ2-Value | p-Value | ||||
| Management | Intensive | 189 | 43 (22.8) | χ2=10.93 | 0.001 |
| Extensive | 195 | 20 (10.3) | |||
| Sex | Female | 175 | 27 (15.4) | 0.183 | 10.38 |
| Male | 209 | 36 (17.2) | |||
| Age | 6-12 Months | 205 | 52 (25.3) | 21.2 | 0.000 |
| less than 6 Months | 179 | 11 (6.1) | |||
| Study Area | Haramaya Town | 195 | 20 (10.3) | 10.94 | 0.004 |
| Haramaya University | 33 | 8 (23.5) | |||
| Harar Town | 156 | 35 (22.6) | |||
| Body Condition | Good | 91 | 11 (11.7) | 11.55 | 0.003 |
| Medium | 163 | 19 (11.8) | |||
| Poor | 130 | 33 (25.4) | |||
| Fecal Consistency | Diarrheic | 116 | 45 (38.8) | 60.74 | 0.000 |
| Normal | 268 | 18 (6.7) | |||
Regression Analysis of Salmonella Occurrence
Multi-variable logistic regression analysis shows that, Salmonella infection was significantly associated with the management system, fecal consistency and age groups (Table 4). Accordingly, calves under six months of age had a higher likelihood (OR =5.2, 95% CI = 0.4–10.9, p less than 0.000) of harboring, salmonella compared to calves of 6 to 12 months of age. Diarrheic animals had harboring Salmonella more frequently than those calves with the normal fecal matter (OR =8.7, 95% CI =0.2-19.2, P less than 0.000). calves under intensive management system had a higher risk of infection as compared to extensive management system (OR = 2.9, 95% CI=0.7-5.8, p = 0.002) (Table 4).
Table 4: Regression analysis of Salmonella and associated risk factors.
| Variables* | No. of Animals with Salmonella (%) | Univariable Logistic Regression | Multivariable Logistic Regression | |||
| OR (95% CI) | p-value | OR (95% CI) | p-value | |||
| Management | Extensive | 20 (10.3) | Ref | 0.001 | Ref | 0.002 |
| Intensive | 43 (22.3) | 2.6 (0.7-4.6) | 2.9 (0.7-5.8) | |||
| Sex | Male | 36 (17.1) | 1.1(0.65-0.1.9) | 0.67 | - | - |
| Female | 27 (15.5) | Ref | - | |||
| Age | 6-12 Months | 11 (6.1) | Ref | 0.000 | Ref | 0.000 |
| less than 6 Months | 52 (25.3) | 4.5 (4.5-9.0) | 5.2 (0.4-10.9) | |||
| Study Area | Haramaya District | 20 (10.3) | Ref | Ref | - | - |
| Haramaya University | 8 (24.2) | 1.1 (2.3-2.5) | 0.91 | - | - | |
| Harar | 35 (22.6) | 2.5 (1.4-4.6) | 0.002 | - | - | |
| Body Condition | Good | 11 (11.7) | Ref | Ref | Ref | Ref |
| Medium | 19 (11.8) | 1(0.4.5-2.2) | 0.995 | - | - | |
| Poor | 33 (25.4) | 2.5 (0.8-5.3) | 0.014 | - | - | |
| Fecal Consistency | Normal | 18 (6.7) | Ref | 0.000 | Ref | 0.000 |
| Diarrheic | 45 (38.8) | 8.7 (0.2-16.1) | 9.8(0.2-19.2) | |||
OR= Odds Ratio; Ref= Reference Variable.
Salmonella Co-infection with Cryptosporidium Oocyst
In the current investigation from 63 (16.4%) salmonella positive animal; 40 (63.5%) were positive for Cryptosporidium oocyst, which accounts 10.4 % (40/384) out of total sample. The highest co-infection association was recorded in age categories of (less than 6 months) (69.2%) than calves 6-12 month (36.4%) (χ2 = 4.23, P = 0.045). Calves with poor body condition (81.8%) significantly higher co-infection than calves with medium (52.6%) and good body condition (27.2%) (χ2 = 11.97, P = 0.003). Statistically significant difference in prevalence of co- infection with fecal consistency. The result indicates that co-infection was highly in diarrheic calves (79.5%) than calves with normal feces (22.2%) (χ2=18.52, p=0.000). The revealed that, there was strong co-infection association between two management system (χ2 = 18.73, P= 0.000). Calves kept under intensive management system (81.4%) were found to be affected more than those kept under extensive management system (0.25). However, no significant co-infection was observed on sex of the animals (χ2=0.36, P =0.34) (Table 5).
Table 5: Salmonella Co-infection association with Cryptosporidium.
| Variables* | No. of Animals Positive for Salmonella | No. of Animals Co-Infection with Cryptosporidium (%) | Chi Square Analysis | ||
| χ2-Value | p-Value | ||||
| Management | Extensive | 20 | 5 (0.25) | 18.730 | 0.000 |
| Intensive | 43 | 35 (81.4) | |||
| Sex | Male | 36 | 24 (66.7) | 0.360 | 0.34 |
| Female | 27 | 16 (59.3) | |||
| Age | 6-12 Months | 11 | 4 (36.4) | 4.230 | 0.045 |
| <6> | 52 | 36 (69.2) | |||
| Study Area | Haramaya Town | 20 | 5 (0.25) | 18.90 | 0.000 |
| Haramaya University | 8 | 6 (0.75) | |||
| Harar Town | 35 | 29 (82.8) | |||
| Body Condition | Good | 11 | 3 (27.2) | 11.970 | 0.003 |
| Medium | 19 | 10 (52.6) | |||
| Poor | 35 | 27 (81.8) | |||
| Fecal Consistency | Normal | 18 | 4 (22.2) | 18.520 | 0.000 |
| Diarrheic | 45 | 36 (79.5) | |||
*Means values are based on Fisher’s exact test.
Antibiotics Susceptibility Profile of Salmonella Isolates
All the 63 isolates were tested against eight commonly used antimicrobials. All isolates were resistant at least to one or more antimicrobials. The antibiotic susceptibility profiles of the isolates showed that; the isolates were 95.2%, 80.9% and 73.0% resistant to Naldixic acid, Amoxicillin and Cefoxitin, respectively. However, the isolates were found to be susceptible to ciprofloxacin (100 %) and chloramphenicol (87.3%) (Table 6). Multiple antimicrobial resistances (resistance to two or more antimicrobials) were detected in 95.2% (60/63) of the isolates. A total of seven different antimicrobial resistance patterns were observed. The most frequent MDR pattern consisting three drugs were found against Naldixic acid, Amoxicillin and Cefoxitin with a resistance of (95.2%), (80.9%) and (73.0%) respectively. The isolates were found to be resistant to four antimicrobial drugs namely, Streptomycin (46.0%), Erythromycin (25.5%) Ampicillin (23.8%) and Chloramphenicol (7.9%) (Table 7).
Table 6: Antibiotic susceptibility result of Salmonella isolates.
| Antimicrobials | No. of Isolates (%) | No. of Susceptible Isolates (%) | No. of Intermediate Isolates (%) | No. of Resistant Isolates (%) |
| Ampicillin | 63 | 38 (60.3) | 10 (15.8) | 15 (23.8) |
| Ciprofloxacin | 63 | 63 (100) | 0 (0) | 0 (0) |
| Chloramphenicol | 63 | 55 (87.3) | 3 (4.7) | 5 (7.9) |
| Cefoxitin | 63 | 17 (26.9) | 0 (0) | 46 (73.0) |
| Erythromycin | 63 | 35 (55.5) | 12 (19.0) | 16 (25.3) |
| Naldixic acid | 63 | 0 (0.00) | 3 (4.8) | 60 (95.2) |
| Streptomycin | 63 | 25 (39.7) | 9 (14.3) | 29 (46.0) |
| Amoxicillin | 63 | 0 (0.00) | 12 (19.0) | 51 (80.9) |
Table 7: Multi drug resistance of Salmonella isolates.
| Isolates with The Same Pattern | Antimicrobial Resistance Pattern | No. of Antimicrobials Developed Resistance |
| 18 | NA, FOX | 2 |
| 11 | NA, FOX, AML, | 3 |
| 11 | NA, FOX, AML, S | 4 |
| 13 | NA, FOX, AML, S, ERY | 5 |
| 9 | NA, FOX, AML, S, ERY, AMP | 6 |
| 1 | NA, FOX, AML, S, ERY, AMP, C | 7 |
AML=Amoxicillin, AMP=Ampicillin, FOX=Cefoxitin, C= Chloramphenicol, S=Streptomycin, ERY = Erythromycin, NA= Naldixic Acid
Discussion
In the present study, from a total of 384 diarrheic and non-diarrheic calves, 16.4 % of the isolates was positive for Salmonella. This finding was relatively comparable with the report of (Sunday et al., 2016) (13%) and (Manickam et al., 2017) (18.33%). The current result was higher than that of (Dereje, 2012) (5%) and lower than that of (Abunna et al., 2017) (43.75%) and (Abdullah et al.,2013) (21.9%). The variations in the Salmonella isolates found in the present study in Ethiopia as well as in other countries could possibly be due to variations in climatic conditions, management practices, including hygienic conditions, time of first colostrum feeding and housing system of the farms.
Significant association was observed between the prevalence of Salmonella and faecal consistency; where diarrheic calves had harboring Salmonella more frequently than those calves with the normal faecal matter. Accordingly, animals with diarrheic fecal condition had 8.7 times the chance of harboring Salmonella (OR=8.7, p-value=0.000) than animals with normal fecal matter. This finding is in line with the report of (Moussa et al, 2010) (43.5%) and (27.69%) in diarrheic and apparently healthy contact calves respectively. The result also shows that the calves with severe diarrhea had highest percentage positivity to bacterial scours. This affirms the virulent ability of the organism to destroy the mucosa of the intestinal lining which affects the absorption and secretion ability of the gastro intestinal system and result in profuse watery foul smelling diarrhea The study revealed that the occurrence of the Salmonella infection was significantly associated with the age of the calf, indicating the calves under six months of age had a higher risk of infection as compared to the older ones (OR =4.5, p = 0.000). This finding was relatively comparable with the report of (Adeladlew,2020) and (Temesgen,2004) that calves aged between 0-3 months were at a higher risk of diarrhea, particularly during the first week of life and the risk decreased with age. This was well explained by (Godden ,2007), (Mellor and Stafferd,2004) who reported that the structure of the bovine placenta impedes easy acquision of immunoglobulins by unborn calves during pregnancy and therefore calves are born without circulating protective antibodies, therefore they are more susceptible to various pathogens. The present study showed that the occurrence of the Salmonella infection was significantly associated with the management system applied indicating that, calves under intensive management system had a higher risk of infection as compared to extensive management system (OR = 2.9, p = 0.002). (Charles et al.,2003) indicated that gaps in management includes inadequate nutrition, exposure to severe environment, insufficient attention to the new born calf, or a combination of these factors are often involved in scours outbreaks. The study identified that, Calves with good body condition had high probability of being infected by Salmonella as compared with calves of poor body condition but statistically not significant (P=0.995). Similarly, higher infection was recorded in male calves than female, but statistically not significant (p=0.670).
In the present study, all Salmonella positive animals were subjected for analysis of co infection association status with Cryptosporidium oocyst. Generally, 63.5% (40/63) of Salmonella positive animals were positive for Cryptosporidium oocyst. A statistically significant co-infection was observed on age less than 6 month (χ2 =4.23, P = 0.045), diarrheic calves (χ2=18.52, p=0.000), poor body condition scores (χ2 = 11.97, P = 0.003) and intensive management system (χ2 = 18.73, P= 0.000). However, no significant co-infection was observed on sex of the animals (χ2=0.36, P =0.34). A strong co-infection was observed on the types of fecal consistency, where, Positive sample were higher in diarrheic calf than non-diarrheic one which was statistically significant (χ2=18.52, p=0.000). This is in accordance with the reports of (Brar et al., 2017), who reports that, 40% of C. parvumpositive fecal samples were also positive for Salmonella spp. This is could be due to the fact that Cryptosporidium is an enteric parasite that causes the loss of epithelial cells and microvillus of the intestine as well as a reduction in the absorptive surface area of the intestine, resulting in diarrhea (Brook et al., 2008). Lorenz et al., (2011) reported that; this is probably due to free cycling of these bacteria between animals in a limited host environment once the pathogen gets access to the farm in animals kept indoors. It can be inferred that calves reared in extensive management have low risk of infection and are more resistant while those reared in intensive are exposed to causes of infection and less resistant to infection. Reasons for this could be poor hygiene in intensive, feeding animals with contaminated concentrates, hay and forages animal to make choice of what to eat could also be a contributing factor.
Improper use of antimicrobials in both human and veterinary medicine has contributed to development and dissemination of antimicrobial resistant pathogens (Tajbakhsh et al., 2012). Out of all 63 isolates tested 60 (95.2%) found to be resistant to one or more antimicrobials. This is in agreement with the report of (Abunna et al.,2017) (96.4), higher than the report of (Zewdu, 2009) (32.7%) and lower than the report of (Adeladlew, 2020) (100%) from Bahir Dar. In the current investigation, the resistance profile of ampicillin (23.8%) and cefoxitin (73.0) were lower than reported by (Adeladlew, 2020) (100%). Manickam et al., (2017) reported (75%) resistance to ampicillin which was higher than the result found in this study.
On the other hand, the sensitivity of Salmonella to ampicillin (23.8%) was lower than reports of (Mulaw, 2017) (94.4%) and cefoxitin (73.0%) was higher than reports of (Mulaw, 2017).
(36.6%). Study in Alexandria Egypt (Mohamed et al., 2011) reported that 85.7% of Salmonella species isolated from dairy cattle were sensitive to ampicillin. This result strongly disagrees with the current study, in which 23.8% of the isolates were resistant to ampicillin. Resistance rates to ampicillin were very high when compared to results documented in America (Blau et al., 2005) reported as 4.4 % resistance levels. Similarly, resistance level of Naldixic acid (95.2%) relatively comparable with result of (Abunna et al., 2017) (85.7%). The resistance profile of amoxicillin (80.9%) was relatively comparable with the study reported by (Abe Kebede et al., 2016) amoxicillin (77%).Therefore the difference in the level of antimicrobial resistance from the previous study might be described from the view of differences in frequency and type of antimicrobials used in an area where the animals originated, or might arise from differences in bacterial culture and antimicrobial susceptibility testing technique (Padungtod and Kaneene, 2006). The difference observed might be also due to differences in the number of antimicrobial drugs tested in the present study and as well as diversity in their usage. In addition, in Ethiopia, the indiscriminate usage of antimicrobials for human as well as animal treatment is increasing from time to time and this might have been contributing to drug resistance. However, the sensitivity of Salmonella isolates to chloramphenicol (87.3%), and ciprofloxacin (100%) agreed with the reports of (Mulaw, 2017). In present study the Salmonella isolates were most susceptible to Ciprofloxacin (100%). This result was similar with the result reported by (Adeladlew, 2020) (100%) from Bahir Dar.
Multi drug resistances of seven out of the sixty-three Salmonella isolates obtained in this study showed MDR to naldixic acd (95.2%), amoxicillin (80.9%), cefoxitin (73.3%), streptomycin (46.0), erythromycin (25.3) ampicillin (23.8), and chloramphenicol (7.9) which could be the consequence of indiscriminate use of antibacterial in clinical practice. The possible reason for high rate of AMR level of Salmonella might be due to the increasing rate of irrational use of antimicrobials in the dairy farms, frequent usage both in livestock and public health, use of counterfeit drugs in animal husbandry self-medication due to easy access to antimicrobials without prescription in public health sector and administration of sub therapeutic dose of antimicrobials to livestock for prophylactic or nutritional purpose in food animals ( Tadesse Birhanu et al., 2014). Most cases of calf diarrhea are likely to be mixed infection, where more than one pathogenic agent is present. In this study, we recognized the following limitations: The study was a cross-sectional study, which could not determine the causal effect relationships between independent and dependent factors. The study did not involve species identification, molecular characterization and seasonal investigation, which may limit the value of the report.
Conclusions and Recommendations
Salmonella is one of the health problems of calves in and around Haramaya District and Harar town, West Hararghe zone. Out of 384 samples, 63 (16.4 %) tested positive for Salmonella. In the current investigation from 63 (16.4%) salmonella positive animal; 40 (63.5%) were positive for Cryptosporidium oocyst. Our findings confirmed that risk factors such as types of farm management, age, body condition scores and fecal consistency were found to have a significant effect on the occurrence of salmonella and co-infection association. Sixty of the 63 isolates were resistant to two or more antimicrobials. The antibiotic susceptibility profiles of the isolates showed that the isolates were 96.8%, 80.9% and 73.0% resistant to Naldixic acid, Amoxicillin and Cefoxitin, respectively. However, the isolates were found to be susceptible to ciprofloxacin (100 %) and chloramphenicol (87.3). In general, pathogenic characteristics of two agents, continuous multidrug resistance patterns of salmonella, absence of effective drug for Cryptosporidium make them difficult to control and this condition makes continuous and persistency infection in the dairy farm. However, Awareness creation, adequate management and hygienic practices are paramount importance in the control and prevention of the disease. Based on the above conclusion the following recommendations are forwarded:
- Better management practices like stream-bank fencing, use all-in, all-out management system, isolate infected animals, cleaning and disinfection activities to limit environmental contamination with fecal matter and use protective materials when handling animals should be implemented.
- Use of effective drugs and treatment of animals by professionals in order to minimize the impact of infection.
- Regular antimicrobial susceptibility tests against Salmonella should be conducted in order to monitor emergence of new MDR strains.
- Veterinarians should be advised to use ciprofloxacin and chloramphenicol in their farms against diarrheal diseases of Salmonella.
- Collaboration is needed among government, professional organizations and interest groups on control and prevention of the disease.
- While the molecular-based study is required to be carried out aiming at species identification and the epidemiology of the salmonella, particular attention should be paid to the control of the disease in the study area.
Abbreviations
BGA: Brilliant Green Agar; BPW: Buffered Peptone Water; CDC: Center for Disease Control; CFSPH: Center for Food Security and Public Health; CSA: Central Statistical Agency; ELISA: Enzyme Linked Immuno Sorbent Assay; FAO: Food and Agricultural Organization; H: Flagellar Antigen; IMViC: Indole, Methyl Red, Voges-Proskauer and Citrate Test; ISO: International Organization for Standardization; MR: Methyl Red; NCD: Neonatal Calf Diarrhea; O: Somatic Antigen; OIE: Office International des Epizooties; OR: Odds Ratio; TSI: Triple Sugar Iron; VP: Vogus-Proskauer; WHO: World Health Organization; XLD: Xylose-lysine-deoxycholate
Acknowledgements
We would like to express our heartfelt appreciation and sincere gratitude to our advisor, Dr. Bruk Abraha, for his invaluable and unwavering support throughout the study period. His material assistance, guidance, intellectual advice, careful manuscript review, constructive criticism, and constant encouragement were essential to the successful completion of this work.
We also extend our sincere appreciation to Sisay Alamu from the Department of Parasitology for his valuable guidance and intellectual support during the study period.
Our sincere thanks go to the Agricultural Office of the Harari Region and its staff for their assistance and cooperation during sample collection. We are also grateful to the dairy farm owners who permitted sampling on their farms, as well as the farm attendants for their cooperation during sample collection.
We would like to extend our sincere gratitude to the Hirna Regional Veterinary Laboratory for their willingness to support our research by providing the necessary culture media, disk diffusion materials, and other laboratory supplies.
We also express our heartfelt thanks to Dr. Hassen Yusuf, Yasin Usman, Dr. Darge Lulu, and Dr. Mohammed Goro Gutu for their laboratory assistance and valuable intellectual advice throughout the study period.
We find it difficult to express in words our deepest sense of gratitude and sincere appreciation to our brother Jamal Abdule, sisters Shamsi Abdule and Fatuma Abdule, as well as Mohammed Hassen, Nasiha Quxusu Shek Umer, Adame Shek Nuure, and the community of Chiro Town as a whole for their invaluable moral and financial support in our education and overall life.
Finally, we would like to express our sincere appreciation to the workers at Haramaya University Farms, the College of Veterinary Medicine, and the laboratory staff for their cooperation and support during fieldwork and laboratory activities.
References
- Abdullah, M., Akter, M. R., Kabir, S. L., Khan, M. A. S., Abdulaziz, M. (2013). Characterization of Bacterial Pathogens Isolated from Calf Diarrhoea in Panchagarh District of Bangladesh. J. Agric. Food. Tech, 3(6):8-13.
Publisher | Google Scholor - Kebede, A., Kemal, J., Alemayehu, H., Habte Mariam, S. (2016). Isolation, Identification, and Antibiotic Susceptibility Testing of Salmonella from Slaughtered Bovines and Ovines in Addis Ababa Abattoir Enterprise, Ethiopia: A Cross‐Sectional Study. International Journal of Bacteriology, 1:3714785.
Publisher | Google Scholor - Acha, P.N., Szyfres, B. (2003). Zoonoses and Communicable Diseases Common to Man and Animals. 3rd ed., Pan American Health Organization, 1:233-246.
Publisher | Google Scholor - Alemu, S. (2007). Salmonella Serovars in Apparently Healthy Slaughtered Cattle at Bahir Dar Abattoir, Ethiopia. MSc Thesis, Addis Ababa University School of Graduate Studies.
Publisher | Google Scholor - Belluco, S., Cibin, V., Davies, R., Ricci, A., Wales, A. (2015). A Review of The Scientific Literature on The Control of Salmonella Spp. in Food-Producing Animals Other Than Poultry. 81.
Publisher | Google Scholor - Birhanu, T., Abda, S., Gebrezgabiher, G. (2014). Prevalence of Salmonella on Sheep Carcasses Slaughtered at Adama Municipal Abattoir, South Eastern Ethiopia. Journal of Science, Technology and Arts Research, 3(3):107-111.
Publisher | Google Scholor - Blau, D. M., McCluskey, B. J., Ladely, S. R., Dargatz, D. A., Fedorka-Cray, P. J., et al. (2005). Salmonella in Dairy Operations in The United States: Prevalence and Antimicrobial Drug Susceptibility. Journal of Food Protection, 68(4):696-702.
Publisher | Google Scholor - Brar, A. P. S., Sood, N. K., Kaur, P., Singla, L. D., Sandhu, B. S., et al. (2017). Periurban Outbreaks of Bovine Calf Scours in Northern India Caused by Cryptosporidium in Association with Other Enteropathogens. Epidemiology & Infection, 145(13):2717-2726.
Publisher | Google Scholor - Brook, E., Hart, C. A., French, N., Christley, R. (2008). Prevalence and Risk Factors for Cryptosporidium Spp. Infection in Young Calves. Veterinary Parasitology, 152(1-2):46-52.
Publisher | Google Scholor - Cacciò, S. M., Widmer, G. (Eds.). (2013). Cryptosporidium: Parasite and Disease. Springer Science & Business Media.
Publisher | Google Scholor - CFSPH, (2005), Cryptosporidiosis, The Center for Food Security and Public Health, Lowa State University.
Publisher | Google Scholor - Charles, L., Stoltenow, L. L., Vincent, M. S. (2003). Calf Scour: Cause, Prevention and Treatment. Extension Service, North Dakota State University.
Publisher | Google Scholor - Cho, Y. I., Yoon, K. J. (2014). An Overview of Calf Diarrhea-Infectious Etiology, Diagnosis, and Intervention. Journal of Veterinary Science, 15(1):1.
Publisher | Google Scholor - Cho, Y. I., Han, J. I., Wang, C., Cooper, V., Schwartz, K., et al. (2013). Case-Control Study of Microbiological Etiology Associated with Calf Diarrhea. Veterinary Microbiology, 166(3-4):375-385.
Publisher | Google Scholor - Wayne, P. A. (2011). Performance Standards for Antimicrobial Susceptibility Testing. Clinical and Laboratory Standards Institute.
Publisher | Google Scholor - CSA, (2013), The Federal Democratic Republic of Ethiopia Central Statistical Agency. Ethiopian Statistical Service.
Publisher | Google Scholor - Debela, E. (2013), Epidemiology of Gastrointestinal Helminthiasis of Rift Valley Goats Under Traditional Husbandry System in Adami Tulu District, Ethiopia. SINET: Ethiop J Sci., 25:35-44.
Publisher | Google Scholor - Danielle, A. B. (2006). Deadly Diseases and Epidemics: Salmonella. David Heymann; Communicable Diseases Section. World Health Organization, Geneva, Switzerland. Chelsea House Publishers, 8-74.
Publisher | Google Scholor - Davison, S. (2005), Salmonellosis. Merck Veterinary Manual 10th Edition. Edited By Cynthia, M. Kahn. Merik And Co. (Inc. White House Station, N.J U.S. A). E.A., 2017. Bovine Cryptosporidiosis: Impact, Host-Parasite Interaction and Control Strategies. Vet. Res. 48:42.
Publisher | Google Scholor - Dereje, W. (2012). Isolation and Identification of Enterobacteria Species from Diarrheic Calves in and Around Addis Ababa, Ethiopia, 6(1):2474-7637.
Publisher | Google Scholor - Fayer, R., Xiao, L. (Eds.). (2007). Cryptosporidium and Cryptosporidiosis. CRC Press.
Publisher | Google Scholor - Fayer, R., Xiao, L. (2008): Cryptosporidium and Cryptosporidiosis, 2nd Edition. CRC Press.
Publisher | Google Scholor - Fossler, C. P., Wells, S. J., Kaneene, J. B., Ruegg, P. L., Warnick, L. D., et al. (2005). Herd-Level Factors Associated with Isolation of Salmonella in A Multi-State Study of Conventional and Organic Dairy Farms: I. Salmonella Shedding in Cows. Preventive Veterinary Medicine, 70(3-4):257-277.
Publisher | Google Scholor - Foster, D. M., Smith, G. W. (2009). Pathophysiology of Diarrhea in Calves. Veterinary Clinics of North America: Food Animal Practice, 25(1):13-36.
Publisher | Google Scholor - Fox L.S. (2007), The 5C’s of Calf Raising. Tri-State Dairy Nutrition Conference. 43-49.
Publisher | Google Scholor - Abunna, F., Ashenafi, D., Beyene, T., Ayana, D., Mamo, B., et al. (2017). Isolation, Identification and Antimicrobial Susceptibility Profiles of Salmonella Isolates from Dairy Farms in and Around Modjo Town, Ethiopia. Ethiopian Veterinary Journal, 21(2):92-108.
Publisher | Google Scholor - Giles, M., Chalmers, R., Pritchard, G., Elwin, K., Mueller-Doblies, D., et al. (2009). Cryptosporidium Hominis in A Goat and A Sheep in the UK. The Veterinary Record, 164(1):24.
Publisher | Google Scholor - Godden, S. (2007). Colostrum’s Management for Dairy Calves in Proceedings from The Conference Calf Management Siolheim Hansen.
Publisher | Google Scholor - Gray, T., Fedorka-Cray, J. (2002), Salmonella. In: Dean, O. and Riemann, P. (eds): Foodborne Diseases. Academic Press, San Francisco, 55-68.
Publisher | Google Scholor - Guesh Mulaw, (2017). Prevalence and antimicrobial susceptibility of Salmonella species from lactating cows in dairy farm of Bahir Dar Town, Ethiopia. 11(43):1578-1585.
Publisher | Google Scholor - HADB. (2009). Haramaya wereda Agricultural Development Bureau. Haramaya, Ethiopia.
Publisher | Google Scholor - Haramaya Wereda Agricultural office (HWAO), Agricultural report of 2018 Harari Regional Agricultural Office (HRAO), Agricultural Report of 2018.
Publisher | Google Scholor - Hendrix, C. (1998). Diagnostic Veterinary Parasitology, 2nd ed., Alabama: University of Auburn, 15-27.
Publisher | Google Scholor - Hijjawi, N.S., B.P. Meloni, M. Nganzo, U.M. Ryan, M.E. Olson, et al, (2015). Complete Development of Cryptosporidium Parvum in Host Cell-Free Culture. International Journal for Parasitology, 34:769-777.
Publisher | Google Scholor - Hoar, B.R., Atwill, E.R., Elmi, C., Farver, T.B. (2001): An Examination of Risk Factors Associated with Beef Cattle Shedding Pathogens of Potential Zoonotic Concern, Epidemiol Infect., 127:147-155.
Publisher | Google Scholor - Holt PS 2000: Host susceptibility, resistance and immunity to Salmonella in animals. In: Wray C, Wary W (eds.). Salmonella in Domestic Animals. pp. 73-88. CABI Publishing, New York.
Publisher | Google Scholor - House, J.K., Smith, B.P. (2004). Profitable Strategies to Control Salmonellosis in Dairy Cattle. Medecin Veterinaire du Quebec, 34(1/2):42-44.
Publisher | Google Scholor - ISO (International Organization for Standardization). (1998). Microbiology of food and animal feeding staff-horizontal method for the detection of Salmonella, ISO, and 6579 Geneva.
Publisher | Google Scholor - ISO (International Organization for Standardization). (2002). Microbiology of food and animal feeding staff-horizontal method for the detection of Salmonella, 4th ed., ISO, 6579 Geneva.
Publisher | Google Scholor - Izzo M, Mohler V, House J. (2011). Antimicrobial susceptibility of Salmonella isolates recovered from calves with diarrhoea in Australia. Aust Vet. 89:402-408.
Publisher | Google Scholor - Jay LS, Davos D, Dundas M, Frankish E, Light Foot, D. (2003). Salmonella Ch 8 In: Hocking AD (Ed) Food borne microorganisms of public health significance. 6th Ed, Australian Institute of Food Science and Technology (NSW Branch), Sydney, 207-266.
Publisher | Google Scholor - Kemal J (2014). A Review on the Public Health Importance of Bovine Salmonellosis. J Veterinar Sci Techno. 5:175.
Publisher | Google Scholor - Klein-Jöbstl D., Iwersen M., Drillich M. (2014). Farm characteristics and calf management practices on dairy farms with and without diarrhea: A case-control study to investigate risk factors for calf diarrhea. J. Dairy Sci, 97:1-10.
Publisher | Google Scholor - Kvac, M., Kouba, M., Vitovec, J. (2006): Age-related and housing-dependence of Cryptosporidium infection of calves from dairy and beef herds in South Bohemia, Czech Republic. Veterinary Parasitology, 137:202-209.
Publisher | Google Scholor - La Ragione, R., Metcalfe, H.J., Villarreal-Ramos, B. Werling, D. (2012): Salmonella Infections in Cattle. In: Salmonella in Domestic Animals, 2nd Edition, 233-262.
Publisher | Google Scholor - Lahti, E. (2010). A prolonged outbreak of Salmonella Reading affecting humans and animals in Sweden. In: Proceedings of International Symposium on Salmonella and Salmonellosis, St Malo, France, 333-337.
Publisher | Google Scholor - Learmonth, J.J., Ionas, G., Ebbett, K.A., Kwan, E.S. (2004). Genetic characterization and transmission cycles of Cryptosporidium species isolated from humans in New Zealand. Appl Environ Microbiol, 70:3973-3978.
Publisher | Google Scholor - Lorenz, I., Mee J.F., Earley, B., More, S.J. (2011): Calf health from birth to weaning. I. General aspects of disease prevention. Irish Vet Jour. 64:10.
Publisher | Google Scholor - Lorino, T., Daudin, J., Robin, S. (2005). Factors associated with time to neonatal diarrhea in French beef calves. Prev Vet Med. 68:91-102.
Publisher | Google Scholor - Luciana, F., Tatiana, A., Renee, M., Andrease, J., Renato, L. (2012). Salmonellosis in cattle: Advantages of being an experimental model. Research in Veterinary Science, 93(1):1-6.
Publisher | Google Scholor - Manickam, R. and Ponnusamy, P. (2017): Bacterial species isolated from diarrheic calves and its antibiotic sensitivity pattern. International Journal of Science, Environment and Technology, 6(4):2202-2211.
Publisher | Google Scholor - Mellado, M., Lopez, E., Veliz, F.G., De Santiago, M.A., Macias-Cruz, U., et al. (2014). Factors associated with neonatal dairy calf mortality in a hot-arid environment. Livestock Science, 159:149-155.
Publisher | Google Scholor - Mellor, D., Stafford, K. (2004): Animal welfare implications of neonatal mortality and morbidity in farm animals. Vet J. 168(2):118-133.
Publisher | Google Scholor - Mohamed Zahran, Farid A, Abaza A, Faltas R. 2011. Fecal shedding of non-typhoidal Salmonella species in dairy cattle and their attendants in Alexandria suburbs. J Am Sci. 7(9):623-631.
Publisher | Google Scholor - Mohler, V.L., Heithof, D.M., Maham, M.J., et al. (2008): Cross protective immunity conferred by a DNA adenine methylase deficient Salmonella eneterica serovar Typhimurium vaccine in calves challenged with Salmonella serovar Newport. Vaccine, 26(14):1751-1758.
Publisher | Google Scholor - Mohler V.L., Izzo, M.M., House, J.K. (2009): Salmonella in calves. Vet Clin North Am Food Anim Pract. 25:37-54.
Publisher | Google Scholor - Moussa, M. H., Ashgan, M. S., Mohamed, K. H. F., A. A. Al-Doss (2010). Rapid detection of Salmonella species in newborn calves by polymerase chain reaction. International Journal of Genetics and Molecular Biology, 2(4):062-066.
Publisher | Google Scholor - Nasir, A., Avais, M., Khan, M. S., Ahmad, N. (2009). Prevalence of Cryptosporidium parvum infection in Lahore (Pakistan) and its association with diarrhea in dairy calves. Int. J. Agric. Biol, 11:221.
Publisher | Google Scholor - Nguyen, S. T., Nguyen, D. T., Quyet Le, D., Le Hua, L. N., Nguyen, T. V., et al. (2007). Prevalence and first genetic identification of Cryptosporidium spp. In cattle in central Viet Nam. Veterinary Parasitology, 150:357-361.
Publisher | Google Scholor - Nielsen L. (2014). Review of pathogenesis and diagnostic methods of immediate relevance for epidemiology and control of Salmonella Dublin in cattle. Veterinary Microbiology, Barcelona, 162(1):1-9.
Publisher | Google Scholor - OIE (2006). Salmonellosis. Office International des Epizooties.
Publisher | Google Scholor - OIÉ (2000). Salmonellosis. In: Manual standards for diagnostic test and vaccines, 4thed. France, Paris. 1-18.
Publisher | Google Scholor - Padungtod, P, Kaneene, B.J. (2006). Salmonella in food animals and humans in northern Thailand. International Journal of Food Microbiology, 108:346-354.
Publisher | Google Scholor - Plym L., Forshell, Wierup M. (2006). Salmonella contamination: a significant challenge to the global marketing of animal food products. Rev. Sci. Tech. Off. Int. Epiz. 25(2):541-554.
Publisher | Google Scholor - Radiostitis, O.M., Gay, C., Constable, P. D., Hinchliff K.W. (2007). Disease associated with protozoa, veterinary medicine a text book of the disease of horse, sheep, pig, and goat. 10th ed. London: Harcourt Publishers Ltd. 1498-1506.
Publisher | Google Scholor - Razzaque M.A., Bedair M. and Abbas S. (2009). Performance of pre-weaned female calves confined in housing and open environment hutches in Kuwait. Pak. Vet. J, 29:1-4.
Publisher | Google Scholor - Ruzante, J.M., Lombard, J.E., Wagner, B., Fossler, C.P., Karns, J.S., et al. (2010). Factors Associated with Salmonella Presence in Environmental Samples and Bulk Tank Milk from US Dairies. Zoonoses and Public Health, 57(7-8):E217-E225.
Publisher | Google Scholor - Sanderson, K.E., Nair, S. (2012). Taxonomy and Species Concepts in the Genus Salmonella. In: Barrow, P.A. & Methner, and U. (Eds) Salmonella in Domestic Animals. 2. ed. United Kingdom: CAB International.
Publisher | Google Scholor - Santin, M., Trout, J.M., Fayer, R. (2008). A longitudinal study of cryptosporidiosis in dairy cattle from birth to 2 years of age. Vet Parasitol., 155.
Publisher | Google Scholor - Sibhat B, Molla B, Zerihun A, Muckle A, Cole L, et al. (2011). Salmonella Serovars and Antimicrobial Resistance Profiles in Beef Cattle, Slaughterhouse Personnel and Slaughterhouse Environment in Ethiopia. Zoonoses Public Health, 58:102-109.
Publisher | Google Scholor - Sischo, W. M., Atwill, E. R., Lanyon, L. E., George, J. (2000). Cryptosporidia on dairy farms and the role these farms may have in contaminating surface water supplies in the northeastern United States. Preventive Veterinary Medicine, 43(4),253-267.
Publisher | Google Scholor - Wudu, T. (2004). Calf morbidity and mortality in dairy farms in Debre Zeit and its envirions, Ethiopia. Faculty of Veterinary Medicine, Addis Ababa University, Addis Ababa, Ethiopia.
Publisher | Google Scholor - Smith, B.P. (2009). Salmonellosis in ruminants. In: Smith BP, editor. Large animal internal medicine. 4th edition. St. Louis (MO): Mosby; 877-881.
Publisher | Google Scholor - Stecher, B. (2015). The roles of inflammation, nutrient availability and the commensal microbiota in enteric pathogen infection. Microbiol Spectr, 3(3).
Publisher | Google Scholor - Sunday Olaogun, C., Olalekan Jeremiah, T., Afusat Jubril, J., Olaoluwa Adewuyi O. (2016). Calf Diarrhea: Epidemiological Prevalence and Bacterial Load in Oyo and Ogun States, Nigeria, 90-96.
Publisher | Google Scholor - Szonyi, B., Chang, Y.F., Wade, S.E., Mohammed, H.O. (2012). Evaluation of factors associated with the risk of infection with Cryptosporidium parvum in dairy calves. Am J Vet Res. 73:76-85.
Publisher | Google Scholor - Tajbakhsh, F., Tajbakhsh, E., Momeni, M., Rahimi, E., Sohrabi, R. (2012). Occurrence and Antibiotic Resistance of Salmonella spp isolated from raw Cow’s Milk from Shahahrekord Iran. Inter. J. Microbiol. Res, 3:242-245.
Publisher | Google Scholor - Tegegne A, Crawford TW (2000). Draft animal power use in Ethiopia. Draft Animal News. 33.
Publisher | Google Scholor - Tesfaye Adeladlew (2020). Isolation, identification and antimicrobial Susceptibility patterns of Salmonella isolates from diarrheic calves in Bahir Dar city dairy farms, North West Ethiopia: VET; 5(4): 03-10.
Publisher | Google Scholor - Thomson, S., Hamilton, C.A., Hope, J.C., Katzer, F., Mabbott, N.A., et al. (2017): Bovine cryptosporidiosis: impact, host-parasite interaction and control strategies. Vet Res. 48(1):42.
Publisher | Google Scholor - Thrusfield, M. (2007). Veterinary epidemiology. 3rd edition London: Blackwell Science. Pp. 227-247.
Publisher | Google Scholor - Van Schaik, G., Schukken, Y.H., Nielen, M., Dijkhuizen, A.A., Barkema, H.W., et al. (2002). Probability of and risk factors for introduction of infectious diseases into Dutch SPF dairy farms: a cohort study. Preventive Veterinary Medicine, 54(3):279-289.
Publisher | Google Scholor - Veling, J., Wilpshaar, H., Frankena, K., Bartels, C., Barkema, H.W. (2002b). Risk factors for clinical Salmonella enterica subsp enterica serovar Typhimurium infection on Dutch dairy farms. Preventive Veterinary Medicine, 54(2):157-168.
Publisher | Google Scholor - Wales, A., Davies, R.H. (2012). Environmental aspects of Salmonella. In: Barrow, P. & Methner, and U. (Eds) Salmonella in Domestic Animals. 2. ed. United Kingdom: CAB International.
Publisher | Google Scholor - Wang, Y., Feng, Y., Cui, B., Jian, F., Ning, C., et al. (2010). Cervine genotype is the major Cryptosporidium Genotype.
Publisher | Google Scholor - Warnick, L.D., Kanistanon, K., McDonough, P.L., Power, L. (2003). Effect of previous antimicrobial treatment on fecal shedding of Salmonella enterica subsp. Enterica serogroup b in NewYork dairy herds with recent clinical salmonellosis. Prev Vet Med (Praha), 56:85-97.
Publisher | Google Scholor - Wondmu Temesgen (2004). Calf morbidity and mortality in small holder dairy farms in Adaa Liben district of Oromia, Ethiopia. Tropic Anim Health Prod, 40:369-376.
Publisher | Google Scholor - Wray C, Davies, R.H. (2000). Salmonella Infections in Cattle. In: Wray, C. and A. Wray. (Eds.). Salmonella in Domestic Animals. New York, CABI Publishing. 169-190.
Publisher | Google Scholor - Wray, C. and Davies, R.H. (2003). The epidemiology and ecology of Salmonella in meat producing animals. In: Torrence, M.E., Isaacson, R.E. (eds). Microbial food safety in animal agriculture. Current Topics 1st Ed USA, Blackwell Publishing. 73-82.
Publisher | Google Scholor - Wudu, T. (2004). Calf morbidity and mortality in dairy farms in Debre Zeit and its envirions, Ethiopia. MSc Thesis. Faculty of Veterinary Medicine, Addis Ababa University, Ethiopia.
Publisher | Google Scholor - Xiao, L., Fayer, R., Ryan, U., Upton S.J. (2004). Cryptosporidium taxonomy: recent advances and implications for public health. Clin. Microbiol. Rev., 17:72-97.
Publisher | Google Scholor - Zewudu, E., Cornelius, P. (2009). Antimicrobial resistance pattern of Salmonella serotypes isolated from food items and personnel in Addis Ababa. Ethiopian Tropical Animal Health and Production, 41(2):241-249.
Publisher | Google Scholor





















