Research Article
Factors Influencing Treatment Success in Unexplained Infertility: A Systematic Review
1 Department of Obstetrics and Gynaecology, Mansa General Hospital, Mansa, Zambia.
2 The JBI (Joanna Briggs Institute) University of West Attica Evidence-Based Healthcare Center, Athens, Greece.
3 School of Medicine, European University of Cyprus, Nicosia, Cyprus.
4 First Department of Obstetrics and Gynecology, Maternity Hospital, Elena Venizelou, Athens, Greece.
5 School of Humanities, Social and Education Sciences, European University of Cyprus, Nicosia, Cyprus.
6 Department of Obstetrics and Gynaecology, Choma General Hospital, Choma, Zambia.
*Corresponding Author: Stylianos Sergios Chatziioannou, The JBI (Joanna Briggs Institute) University of West Attica Evidence-Based Healthcare Center, Athens, Greece.
Citation: Chalwe C; Stylianos S. Chatziioannou, Papasideri V., Siamalambwa Q. (2025). Factors Influencing Treatment Success in Unexplained Infertility: A Systematic Review, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 8(3):1-10. DOI: 10.59657/2837-4681.brs.25.186
Copyright: © 2025 Stylianos Sergios Chatziioannou, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: June 16, 2025 | Accepted: June 30, 2025 | Published: July 07, 2025
Abstract
Background: Unexplained infertility is a common diagnosis among couples struggling to conceive, accounting for approximately 30-40% of infertility cases. Despite advancements in reproductive medicine, predicting treatment success remains challenging.
Objective: This systematic review aimed to synthesize evidence on factors influencing treatment success in unexplained infertility and provide insights for optimizing clinical and policy approaches.
Methods: A systematic search was conducted on PubMed, Cochrane Database and Google Scholar to identify studies investigating prognostic factors and treatment outcomes in unexplained infertility.
Results: Seven studies involving over 1,800 couples were included in the systematic review. Female age, duration of infertility, history of miscarriage, number of treatment cycles attempted, duration of stimulation, pre-ovulatory follicle count, follicular size, endometrial thickness, endometrial pattern, total motile sperm count, smoking status, and socioeconomic status were significant predictors of success. Success rates plateaued after three intrauterine insemination (IUI) cycles, prompting a transition to in-vitro fertilization (IVF) for improved outcomes.
Conclusions: Treatment success in unexplained infertility is influenced by a combination of biological, lifestyle, and socioeconomic factors. Personalised treatment strategies and early intervention are critical for optimising outcomes.
Keywords: unexplained infertility; prognostic factors; treatment success; personalized fertility treatment; intrauterine insemination (IUI); iIn-vitro fertilization (IVF)
Introduction
Unexplained infertility is defined as the failure to achieve pregnancy after at least 12 months of regular, unprotected intercourse, despite normal results from standard fertility evaluations, including assessments of ovulation, tubal patency, uterine anatomy, and semen analysis (Raperport et al., 2024). It is considered a diagnosis of exclusion, meaning that common causes such as ovulatory disorders, tubal damage, and male factor infertility must be ruled out first. This lack of a specific cause complicates both clinical management and prognosis. Unexplained infertility accounts for approximately 30% of infertility cases globally (Penzias et al., 2020). Couples facing this diagnosis often undergo extensive testing without receiving definitive answers, contributing to emotional distress, anxiety, and frustration (Mostafa and Elashram, 2020). The absence of a clear etiology creates a challenging scenario for both patients and clinicians. Diagnosis is further limited by the inability of conventional tests to detect subtle abnormalities. For example, current diagnostic tools may fail to identify minor sperm dysfunctions, subclinical endometrial receptivity issues, or egg abnormalities that affect fertilisation or implantation (Carson and Kallen, 2021; Mansour, 2023). Conditions like early-stage endometriosis or diminished ovarian reserve may also remain undetected (Kamath and Deepti, 2016; Wang et al., 2023). While advanced tools such as endometrial receptivity arrays and embryo genetic testing may reveal hidden issues, they are costly and not routinely used (Buckett and Sierra, 2019; The Guideline Group on Unexplained Infertility et al., 2023). Furthermore, reproductive function can vary across menstrual cycles, so single-cycle testing may not accurately reflect fertility potential (Bayoumi et al., 2024).
Despite the diagnostic uncertainty, many couples eventually conceive without intervention. Studies show that up to 80% of couples with unexplained infertility achieve an ongoing pregnancy, with around 74% conceiving naturally (Brandes et al., 2011; Sadeghi, 2015). Younger age and shorter duration of infertility improve the chances of spontaneous conception, whereas older age and longer durations reduce them (Abdelazim et al., 2018). Treatment options range from expectant management to assisted reproductive technologies (ART). Expectant management may be suitable for younger couples with a favourable prognosis, especially if their chance of conceiving within six months is over 30% (Shingshetty et al., 2024). However, active treatments generally offer higher success rates (Wang et al., 2019; Wessel et al., 2022). The first-line treatment for unexplained infertility typically combines intrauterine insemination (IUI) with ovarian stimulation (OS), offering a balance between effectiveness, invasiveness, and cost (The Guideline Group on Unexplained Infertility et al., 2023; Homburg, 2022). Evidence shows that IUI-OS can result in satisfactory pregnancy rates within three cycles, though the risk of multiple pregnancies and ovarian hyperstimulation remains a concern (Osmanlıoğlu et al., 2022; Cohlen et al., 2018). When IUI-OS fails after three to six attempts, in vitro fertilisation (IVF) is usually recommended (Pandian et al., 2015; Homburg, 2022).
IVF offers higher success rates, particularly in older women or when rapid intervention is necessary. However, unexplained infertility is associated with an increased risk of total fertilisation failure (TFF), reported in 8.4–22.7% of IVF cycles (Sadeghi, 2015). Intracytoplasmic sperm injection (ICSI) is often used to reduce TFF, though its benefit in unexplained infertility remains debated (Qiu et al., 2024; Senapati et al., 2017; Iwamoto et al., 2024). Several factors affect treatment outcomes. Female age is consistently shown to be the strongest predictor, with significantly reduced success rates after age 35 (Somigliana et al., 2016; ESHRE Capri Workshop Group, 2005). The role of male age is less clear, with mixed findings (Elbardisi et al., 2021; Coban et al., 2019). Other factors include BMI, smoking, alcohol use, duration of infertility, and psychosocial stress (Sneed et al., 2008; Whynott et al., 2021; Rockhill et al., 2019; Lyngsø et al., 2021; Domar, 2004; Huang et al., 2024). Socioeconomic status and access to care also play critical roles in treatment success (Imrie et al., 2023). A prognosis-based approach to unexplained infertility is gaining interest, aiming to tailor treatments based on individual predictors such as age, fertility history, and lifestyle factors (Ramya et al., 2023; Shingshetty et al., 2024). Therefore, the purpose of this study is to systematically review the current literature on prognostic factors influencing treatment success in unexplained infertility, with the goal of guiding personalised, evidence-based clinical care and identifying gaps for future research.
Materials and Methods
Study Design
This systematic review was conducted to evaluate the prognostic factors influencing treatment success in couples with unexplained infertility. The review was structured using the PICO framework. The population consisted of couples diagnosed with unexplained infertility. Interventions included fertility treatments such as intrauterine insemination (IUI) with ovarian stimulation and/or in vitro fertilisation (IVF). There was no comparison group, and the primary outcomes of interest were pregnancy rates, live birth rates, and associated predictive factors. The review adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure methodological transparency and reproducibility (Page et al., 2021).
Eligibility Criteria
Studies were included if they examined couples with unexplained infertility, evaluated IUI, IVF, or controlled ovarian stimulation, and assessed predictors of treatment success. Eligible studies were limited to randomised controlled trials; prospective or retrospective cohort studies published in English between 2014 and 2024. Studies were excluded if they investigated infertility due to known causes such as tubal factor, male factor, endometriosis, or anovulation. Reviews, case reports, opinion articles, non-English publications, and studies with incomplete outcome data were also excluded.
Search Strategy
A comprehensive literature search was performed in PubMed, the Cochrane Library, and Google Scholar. The search strategy combined MeSH terms and keywords including “unexplained infertility,” “idiopathic infertility,” “prognostic factors,” “treatment success,” “pregnancy rate,” and “live birth rate,” using Boolean operators to refine results. Filters were applied to limit the search to English-language studies involving human participants and published within the last ten years. Citation tracking and manual searches of reference lists from included studies were also conducted to ensure coverage.
Study Selection
Search results were imported into Rayyan software, where duplicates were removed. Two reviewers independently screened the titles and abstracts, followed by full-text review of potentially relevant studies based on the predefined eligibility criteria. Any disagreements were resolved through discussion with a third reviewer. The PRISMA flow diagram was used to document the study selection process.
Data Extraction and Quality Assessment
Data were extracted using a standardised form, capturing study characteristics, sample size, intervention type, outcome measures, and significant predictors. The Cochrane Risk of Bias 2.0 tool was used for randomised trials, and the ROBINS-I tool was used for observational studies to assess the risk of bias across relevant domains.
Data Synthesis
Due to variability in study designs, interventions, and outcomes, a meta-analysis was not feasible. Instead, a narrative synthesis was undertaken to identify consistent patterns and significant predictors across studies. Results were summarised thematically, focusing on treatment outcomes and prognostic factors.
Ethics
As this review analysed previously published data, no primary data collection was involved. Ethical approval was obtained from the Faculty of Education and Life Sciences at the University of South Wales. All included studies were assumed to have received prior ethical approval.
Results
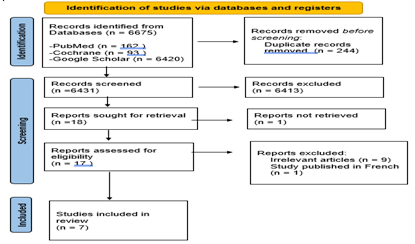
The database searches yielded a total of 6675 records, comprising 6420 records from Google Scholar, 162 records from PubMed, and 93 record from the Cochrane database The abstracts were uploaded to Rayyan software, which identified 244 duplicates that were subsequently removed. Screening was conducted on 6431 abstracts, leading to the manual exclusion of 6413 abstracts. Although 18 full articles were sought, only 17 were retrieved, as the full text of one article could not be accessed. After assessing the 17 reports for eligibility, 10 were excluded (9 irrelevant articles and 1 French-language study), leaving 7 studies that met the eligibility criteria and were included in the review. The 7 included studies consisted of 1 randomised controlled trial (RCT) and 6 observational studies. A detailed illustration of the study selection process is provided in the accompanying PRISMA flow diagram (Figure 1).
Figure 1: PRISMA 2020 Flow Diagram
Characteristics of Included Studies
The studies were published between 2016 and 2022, with sample sizes ranging from 50 to 900 participants. The included studies involved a total of 1,675 participants with unexplained infertility. One study employed a randomized controlled trial (RCT) design, while others were prospective cohort studies (n = 3) and retrospective analyses (n = 3). The treatment approaches varied across studies and included natural and stimulated IUI, controlled ovarian stimulation (COS) with IUI, and COS with IUI followed by IVF in cases where COS-IUI was unsuccessful. Table 1 provides an overview of the characteristics of the studies included in the analysis.
Table 1: Characteristics of included studies
| Author (Year) | Location | Study design, Sample Size | Interventions | Prognostic factors studied | Predictors of treatment success | |
| 1 | Ganguly et al. (2016) | India | Prospective observational study, 146 | Controlled ovarian stimulation (COS) with Intrauterine Insemination (IUI) | Age, duration of infertility, duration of ovarian stimulation, number of dominant follicles >14mm in diameter, endometrial thickness, number of cycles, body mass index (BMI) | Number of cycles and duration of stimulation. |
| 2 | Guan et al. (2021) | China | Retrospective study, 212 | COS with IUI | Female age, smoking, affected pregnancy, number of cycles, BMI, treatment regimen, type of infertility (primary or secondary), endometrium, and timing of insemination | Female age, smoking status and the number of cycles. |
| 3 | Hansen et al. (2016) | United States | Secondary analysis of data from a prospective, randomized, multicenter clinical trial, 900 | COS with IUI | Age, BMI, waist circumference, waist-to-hip ratio, race, ethnicity, smoking, alcohol consumption, income, education level, duration of infertility, total motile sperm count from screening semen analysis, history of conception, previous pregnancy loss, prior parity, past infertility treatments, serum Anti-Mullerian Hormone (AMH) levels, and emotional domain scores | Female age, duration of infertility, previous pregnancy loss and income level |
| 4 | Ohannessian et al. (2018) | France | Retrospective Study, 133 | COS with IUI, In vitro Fertilisation (IVF) | Age, BMI and smoking status, duration of infertility, type of infertility, antral follicle count (AFC), day 3 serum FSH level, serum AMH, female partner Chlamydia trachomatis serology | No prognostic factors were found. |
| 5 | Prithivi et al. (2022) | India | prospective observational study, 124 | COS with IUI | Age, BMI, duration of infertility, type of infertility | No prognostic factors were found. |
| 6 | Raouf et al. (2020) | Iraq | Retrospective study, 110 | COS with IUI | Age, duration of infertility, and type of infertility (primary or secondary), sperm count, sperm motility, progressive motility, follicular size, number of follicles and endometrial thickness. | Duration of infertility, male and female age, type of infertility, sperm count, sperm motility, progressive motility, follicular size, number of follicles and endometrial thickness. |
| 7 | Shrivastava et al. (2016) | India | Prospective observational study, 50 | Natural IUI and COS with IUI | Age, duration of infertility, type of infertility (primary or secondary), stimulation protocol, AFC, endometrial thickness, endometrial pattern, number of preovulatory follicles and pre-post wash count and motility | Female age, number of preovulatory follicles, motile sperm count and endometrial pattern. |
The treatment outcomes varied across studies. Two studies, Hansen et al. (2016) and Ohannessian et al. (2018) reported the live birth rate (LBR) while the other 5 studies reported the pregnancy rate per cycle. The clinical pregnancy rate (CPR) per cycle varied across studies, ranging from 11.29% to 30.9%. Ganguly et al. (2016) reported a per-cycle pregnancy rate of 11.29%. Guan et al. (2021) observed a per-cycle pregnancy rate of 13.7%, with a cumulative success rate of 28.9 percentage across three cycles. Shrivastava et al. (2016) noted a cumulative pregnancy rate of 17.2% over two cycles. Raouf et al. (2020) reported the highest success, with a per-cycle pregnancy rate of 30.9%. Privithi et al. (2022) documented a per-cycle pregnancy rate of 16.1%, with most conceptions occurring within the first two cycles. Hansen et al. (2016) identified a cumulative live birth rate of approximately 25 percentage after four cycles. Ohannessian et al. (2018) observed a cumulative live birth rate of 37.6 (after an average of 2 cycles of IUI) and a cumulative live birth rate of 65.7 percentage when couples who underwent IUI followed by IVF if initial IUI attempts were unsuccessful are included. Table 2 presents a summary of the treatment outcomes from the included studies, with a focus on the success rates achieved.
Table 2: Treatment Outcomes of the included studies
| Study | Clinical Pregnancy Rate Per Cycle (%) | Cumulative Pregnancy Rate (%) | Cumulative Live Birth Rate (%) |
| Ganguly et al. (2016) | 11.29 | Not reported | Not reported |
| Guan et al. (2021) | 13.7 | 28.9 (up to 3 cycles) | Not reported |
| Hansen et al. (2016) | Not specified | Not specified | 25 (up to 4 cycles) |
| Ohannessian et al. (2018) | Not specified | Not specified | 37.6 (after an average of 2 cycles of IUI) 65.7 (including couples who underwent IVF after initial IUI attempts were unsuccessful). |
| Shrivastava et al. (2016) | 14 (first cycle), 20.93 (second cycle) | 17.2 | Not reported |
| Raouf et al. (2020) | 30.9 | Not reported | Not reported |
| Privithi et al. (2022) | 16.1 | 27.1 (all cycles combined) | Not reported |
Guan et al. (2021) observed that the clinical pregnancy rate (CPR) following intrauterine insemination (IUI) improved with the number of treatment cycles but plateaued after the third cycle, followed by a notable decline. Specifically, they reported a CPR of 63.9% in the first cycle, which dropped to 19.7% in the second, 14.8% in the third, and declined further to just 1.6% in subsequent cycles. Similarly, Ganguly et al. (2016) found that the highest CPR occurred during the first IUI cycle at 15.75%, decreasing to 5.88% in the second cycle, with no pregnancies observed in the third. This progressive decline across cycles was statistically significant (p = 0.045), suggesting that repeated attempts beyond the third cycle may not significantly improve outcomes. The duration of ovarian stimulation has also been shown to influence IUI success. According to Ganguly et al. (2016), women who conceived had a significantly longer average stimulation period (12.92 days) compared to those who did not conceive (11.39 days), with a p-value of 0.037, indicating a positive correlation between prolonged stimulation and successful outcomes.
Endometrial and follicular characteristics also appear to be important predictors of treatment success. Raouf et al. (2020) reported that women who achieved pregnancy had a thicker endometrial lining (mean 10.3 mm) than those who did not (mean 8.1 mm). In addition, endometrial pattern was associated with outcome, as Shrivastava et al. (2016) found that a trilaminar endometrial appearance correlated with a higher pregnancy rate (33.33%) compared to an isoechoic pattern (8.12%), a statistically significant difference (p = 0.004). Follicular size was another important factor. Raouf et al. (2020) found that the mean follicular size in successful cycles was 20.73 mm, compared to 19.3 mm in unsuccessful ones (p less than 0.001), suggesting that optimal follicular development is associated with increased likelihood of conception. Similarly, the number of preovulatory follicles was found to influence outcomes. Shrivastava et al. (2016) showed that higher CPRs were observed in women with more follicles, peaking at 41.67% for those with three mature follicles (p = 0.046). However, the increased number of follicles also raises the risk of multiple gestations, underlining the need for cautious monitoring during stimulation.
Finally, semen parameters—particularly total motile sperm count (TMSC)—play a critical role in predicting IUI success. Studies by Raouf et al. (2020) and Shrivastava et al. (2016) highlighted the importance of TMSC, with Raouf et al. reporting that couples with TMSC greater than 10 million had significantly higher CPRs (18.6%) compared to those with lower counts (7.2%), with a p-value of less than 0.01. The probability of achieving pregnancy declined sharply when TMSC fell below 5 million, indicating insufficient sperm availability for effective fertilization.
Risk of bias
The risk of bias was low in one study, moderate in 3 studies, and high in 3 studies due to incomplete outcome reporting. The risk-of-bias tables are shown in figure 2 and figure 3 below.
Non-Randomized Studies
Figure 2: Risk of bias table for non-randomized studies
Randomized Controlled Trial
Figure 3: Risk of bias table for the RCT
Discussion
This systematic review synthesized data from multiple studies investigating prognostic factors associated with treatment success in unexplained infertility. Cumulative pregnancy rates (CPRs) were highest within the first two to three cycles of intrauterine insemination (IUI), with rates ranging from 11.3% to 30.9% per cycle. Beyond three cycles, success rates declined significantly, suggesting a limited window for optimal IUI outcomes. Controlled ovarian stimulation (COS) combined with IUI emerged as an effective initial approach. Key predictors of success included female age, infertility duration, previous miscarriage, total motile sperm count (TMSC), stimulation duration, follicular development, endometrial thickness and pattern, smoking status, and socioeconomic factors.
The findings reinforce well-established predictors of fertility treatment success, while also offering nuanced insights specific to unexplained infertility.
Patient Characteristics and Clinical Predictors
Female age remains the most critical determinant of treatment success, with consistently higher pregnancy rates observed in women under 35 years. This is supported by prior studies (e.g., Hansen et al., 2016; Maheshwari et al., 2008), which have shown that increasing age negatively affects ovarian reserve and oocyte quality. The data strongly support early intervention for older women to avoid the compounding effects of diminished reproductive potential.
Infertility duration also emerged as a significant prognostic factor. Couples with infertility durations under five years demonstrated better outcomes, a finding consistent with retrospective analyses such as Huang et al. (2024). This trend suggests that delayed treatment may be associated with underlying reproductive pathology that becomes more pronounced over time, particularly in women over 35. Although body mass index (BMI) has been broadly linked to reduced fertility outcomes, this review found a limited effect of BMI on success rates in unexplained infertility. This contrasts with evidence from Supramaniam et al. (2018), where elevated BMI was associated with reduced live birth rates. The discrepancy may be explained by variability in study populations or differences in stimulation protocols that compensate for BMI-related reproductive changes. A history of miscarriage was associated with improved outcomes, aligning with findings by Cameron et al. (2017). This may reflect preserved reproductive potential in individuals who have previously conceived, even if pregnancy was not sustained. Similarly, higher success rates among couples with secondary infertility point toward milder or resolved fertility barriers.
Lifestyle and Socioeconomic Factors
Smoking was consistently associated with lower pregnancy rates, though it did not appear to influence live birth rates once pregnancy was established. These results align with evidence showing that smoking impairs endometrial receptivity and sperm quality. The findings support integrating smoking cessation programs into fertility care. Socioeconomic status was a strong predictor of treatment success. Couples with higher incomes had better access to advanced treatments and were more likely to complete multiple cycles, increasing their cumulative success rates. This underscores the importance of addressing health disparities and expanding insurance coverage and public fertility funding, especially in lower-income settings (Hansen et al., 2016; Smith et al., 2011).
Treatment-Related Factors
Success rates peaked within the first three IUI cycles, after which diminishing returns were consistently observed. This supports the recommendation for early transition to IVF in cases of repeated IUI failure (van Rumste et al., 2008; Guan et al., 2021). COS with IUI was identified as an effective first-line treatment in younger women, consistent with findings from Lai et al. (2024). Stimulation duration also played a role in predicting success, with longer stimulation periods associated with improved outcomes, provided that ovarian hyperstimulation is avoided. Studies such as Tian et al. (2022) suggest a balance is needed to optimise follicular development while minimising risk. The development of multiple mature follicles was positively correlated with pregnancy outcomes, though this benefit must be weighed against the risk of multiple gestations. Follicular size, especially within the range of 18–22 mm, was also associated with higher pregnancy rates, reinforcing the importance of precise monitoring and timing of ovulation triggers. Endometrial thickness and pattern were significant predictors of implantation success. A trilaminar pattern and EMT between 8-14mm were associated with optimal outcomes, consistent with prior studies (Hamdi et al., 2018; Zhao et al., 2014). While some research supports acceptable outcomes even with thinner EMT, clinicians should remain vigilant in assessing and managing endometrial parameters. Total motile sperm count (TMSC), even within “normal” semen analysis parameters, significantly influenced IUI outcomes. Success rates were highest when post-preparation TMSC exceeded 10 million/mL. These findings align with evidence from Raouf et al. (2020) and support the routine assessment of TMSC in unexplained infertility cases.
Strengths and Limitations
This review’s strength lies in its comprehensive synthesis of diverse studies from various geographic and clinical settings, providing a robust overview of current knowledge. Adherence to PRISMA guidelines and use of validated bias assessment tools enhance the reliability of the findings. However, the review is limited by the heterogeneity of included studies in terms of design, population characteristics, outcome measures, and intervention protocols. The predominance of observational studies limits causal inference, and language restrictions may have excluded relevant non-English research. Additionally, a narrative synthesis was used due to methodological variability, preventing quantitative pooling of results.
Clinical and Policy Implications
Clinically, these findings support a personalised, prognosis-driven approach to managing unexplained infertility. IUI with COS should be limited to three cycles, particularly in women under 35 with shorter infertility duration. For patients with poor prognostic factors such as advanced age or prolonged infertility, early transition to IVF is recommended to maximise outcomes. From a policy perspective, disparities in access to fertility treatment remain a critical concern. Expanding public funding and insurance coverage, particularly for diagnostics and first-line treatments, could improve equity in fertility care. Integration of lifestyle counselling and psychosocial support may further enhance patient outcomes and quality of life.
Heterogeinety Among Studies
Significant heterogeneity was observed across the included studies, driven by differences in population characteristics, study design, treatment protocols, healthcare access, and outcome measures. Variability in age, BMI, and infertility duration likely influenced success rates—for instance, younger participants in Raouf et al. (2020) (mean age 23.8) had higher outcomes than those in Privithi et al. (2022) (mean age 30.4). Study designs ranged from high-quality randomized controlled trials (e.g., Hansen et al., 2016) to retrospective analyses (e.g., Raouf et al., 2020), affecting the reliability of findings. Intervention protocols also varied, particularly in the choice of ovarian stimulation agents and luteal phase support. Differences in healthcare systems influenced access to treatment. Studies from resource-limited settings emphasized cost-effective strategies, while others, like Hansen et al. (2016), highlighted the role of insurance and socioeconomic factors. Additionally, outcomes were inconsistently reported—some focusing on clinical pregnancy rates, others on live birth rates—limiting comparability. Sample size discrepancies, such as 50 participants in Shrivastava et al. (2016) versus over 100 in larger trials, further contributed to variability. Cultural and lifestyle differences, especially in smoking prevalence and public health practices, also impacted treatment outcomes and may explain inconsistent findings across populations.
Conclusion
This systematic review underscores the multifactorial nature of treatment success in unexplained infertility, highlighting key predictors such as female age, infertility duration, prior miscarriage, stimulation duration, follicular and endometrial parameters, TMSC, smoking status, and socioeconomic factors. The findings support a personalized, evidence-based approach to optimize outcomes and reduce unnecessary interventions. While the review draws from diverse clinical contexts and offers a comprehensive synthesis of prognostic factors, limitations include heterogeneity in study design, small sample sizes, and potential language bias. Nonetheless, the results provide valuable guidance for clinical decision-making and policy development. Future research should focus on advanced diagnostics, equitable access to care, and AI-driven predictive tools to improve stratification, personalize treatment, and enhance patient outcomes.
List of abbreviations
AFC: Antral Follicle Count;
AMH: Anti-Müllerian Hormone;
ART: Assisted Reproductive Technology;
ASRM: American Society for Reproductive Medicine;
BMI: Body Mass Index; CI: Confidence Interval;
CLBR: Cumulative Live Birth Rate;
COH: Controlled Ovarian Hyperstimulation;
CPR: Clinical Pregnancy Rate;
DOR: Diminished Ovarian Reserve;
EMT: Endometrial Thickness;
ESHRE: European Society of Human Reproduction and Embryology;
FSH: Follicle-Stimulating Hormone;
hCG: Human Chorionic Gonadotropin;
hMG: Human Menopausal Gonadotropin;
HSG: Hysterosalpingogram;
ICSI: Intracytoplasmic Sperm Injection;
IUI: Intrauterine Insemination;
IVF: In Vitro Fertilization;
LBR: Live Birth Rate;
LH: Luteinizing Hormone;
MeSH: Medical Subject Headings;
OR: Odds Ratio;
PR: Pregnancy Rate;
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses;
RCT: Randomized Controlled Trial;
TMSC: Total Motile Sperm Count;
WHO: World Health Organization
References
- Abdelazim, I., Purohit, P., Farag, R., et al. (2018). Unexplained infertility: Prevalence, possible causes and treatment options. Journal of Obstetrics and Gynaecology, 113(1).
Publisher | Google Scholor - Bayoumi, R. R., Hurt, L., Zhang, N., Law, Y. J., Venetis, C., Fatem, H. M., Serour, G. I., van der Poel, S., & Boivin, J. (2024). A critical systematic review and meta-analyses of risk factors for fertility problems in a globalized world. Reproductive BioMedicine Online, 48(3):103217.
Publisher | Google Scholor - Buckett, W., & Sierra, S. (2019). The management of unexplained infertility: An evidence-based guideline from the Canadian Fertility and Andrology Society. Reproductive BioMedicine Online, 39(4):633–640.
Publisher | Google Scholor - Carson, S. A., & Kallen, A. N. (2021). Diagnosis and management of infertility: A review. JAMA, 326(1):65.
Publisher | Google Scholor - Cohlen, B., Bijkerk, A., Van Der Poel, S., & Ombelet, W. (2018). IUI: Review and systematic assessment of the evidence that supports global recommendations. Human Reproduction Update, 24(3):300–319.
Publisher | Google Scholor - Domar, A. D. (2004). Impact of psychological factors on dropout rates in insured infertility patients. Fertility and Sterility, 81(2):271–273.
Publisher | Google Scholor - ESHRE Capri Workshop Group. (2005). Fertility and ageing. Human Reproduction Update, 11(3):261–276.
Publisher | Google Scholor - Homburg, R. (2022). IUI is a better alternative than IVF as the first-line treatment of unexplained infertility. Reproductive BioMedicine Online.
Publisher | Google Scholor - Huang, C., Shi, Q., Xing, J., Yan, Y., Shen, X., Shan, H., et al. (2024). The relationship between duration of infertility and clinical outcomes of intrauterine insemination for younger women: A retrospective clinical study. BMC Pregnancy and Childbirth, 24(1):199.
Publisher | Google Scholor - Iwamoto, A., Summers, K. M., Sparks, A., & Mancuso, A. C. (2024). Intracytoplasmic sperm injection versus conventional in vitro fertilization in unexplained infertility. F&S Reports, 5(3):263–271.
Publisher | Google Scholor - Kamath, M., & Deepti, M. (2016). Unexplained infertility: An approach to diagnosis and management. Current Medical Issues, 14(4):94.
Publisher | Google Scholor - Lyngsø, J., Ramlau-Hansen, C. H., Bay, B., Ingerslev, H. J., Strandberg-Larsen, K., & Kesmodel, U. S. (2019). Low-to-moderate alcohol consumption and success in fertility treatment: A Danish cohort study. Human Reproduction, 34(7):1334–1344.
Publisher | Google Scholor - Maheshwari, A., Hamilton, M., & Bhattacharya, S. (2008). Effect of female age on the diagnostic categories of infertility. Human Reproduction, 23(3):538–542.
Publisher | Google Scholor - Mansour, H. A. E. (2023). Infertility diagnosis and management. Beni-Suef University Journal of Basic and Applied Sciences, 12(1):81.
Publisher | Google Scholor - Osmanlıoğlu, Ş., Şükür, Y. E., Tokgöz, V. Y., Özmen, B., Sönmezer, M., Berker, B., Aytaç, R., & Atabekoğlu, C. S. (2022). Intrauterine insemination with ovarian stimulation is a successful step prior to assisted reproductive technology for couples with unexplained infertility. Journal of Obstetrics and Gynaecology, 42(3):472–477.
Publisher | Google Scholor - Page, M. J., McKenzie, J. E., Bossuyt, P. M., et al. (2021). The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ, n71.
Publisher | Google Scholor - Pandian, Z., Gibreel, A., & Bhattacharya, S. (2015). In vitro fertilisation for unexplained subfertility. Cochrane Database of Systematic Reviews.
Publisher | Google Scholor - Penzias, A., Bendikson, K., Falcone, T., et al. (2020). Evidence-based treatments for couples with unexplained infertility: A guideline. Fertility and Sterility, 113(2):305–322.
Publisher | Google Scholor - Qiu, F., Zuo, Y., Xue, H., & Zhang, R. (2024). Fertilization, pregnancy, and neonatal outcomes after IVF, rescue ICSI, and ICSI in unexplained infertility: A retrospective study. Molecular Reproduction and Development, 91(2):e23734.
Publisher | Google Scholor - Raperport, C., Desai, J., Qureshi, D., et al. (2024). The definition of unexplained infertility: A systematic review. BJOG: An International Journal of Obstetrics & Gynaecology, 131(7):880–897.
Publisher | Google Scholor - Rockhill, K., Tong, V. T., Boulet, S. L., et al. (2019). Smoking and clinical outcomes of assisted reproductive technologies. Journal of Women’s Health, 28(3):314–322.
Publisher | Google Scholor - Sadeghi, M. R. (2015). Unexplained infertility: The controversial matter in management of infertile couples. Journal of Reproduction & Infertility, 16(1):1–2.
Publisher | Google Scholor - Senapati, S., Koelper, N. C., Sammel, M. D., Johnson, L., & Dokras, A. (2017). ICSI in unexplained infertility cycles: A linked cycle analysis of the SART database. Fertility and Sterility, 108(3):e331–e332.
Publisher | Google Scholor - Shingshetty, L., Wang, R., Feng, Q., Maheshwari, A., & Mol, B. W. (2024). Prognosis-based management of unexplained infertility—Why not? Human Reproduction Open, 2024(2):hoae015.
Publisher | Google Scholor - Smith, J. F., Eisenberg, M. L., Glidden, D., et al. (2011). Socioeconomic disparities in the use and success of fertility treatments: Analysis of data from a prospective cohort in the United States. Fertility and Sterility, 96(1):95–101.
Publisher | Google Scholor - Sneed, M. L., Uhler, M. L., Grotjan, H. E., et al. (2008). Body mass index: Impact on IVF success appears age-related. Human Reproduction, 23(8):1835–1839.
Publisher | Google Scholor - Somigliana, E., Paffoni, A., Busnelli, A., et al. (2016). Age-related infertility and unexplained infertility: An intricate clinical dilemma. Human Reproduction, 31(7):1390–1396.
Publisher | Google Scholor - Supramaniam, P. R., Mittal, M., & Lim, L. N. (2017). The impact of body mass index on assisted reproductive treatments. Open Journal of Obstetrics and Gynecology, 7(5):562–570.
Publisher | Google Scholor - The Guideline Group on Unexplained Infertility. (2023). Evidence-based guideline: Unexplained infertility. Human Reproduction, 38(10):1881–1890.
Publisher | Google Scholor - Wang, Q., Gu, X., Chen, Y., et al. (2023). The effect of sperm DNA fragmentation on in vitro fertilization outcomes of unexplained infertility. Clinics, 78:100261.
Publisher | Google Scholor - Wang, R., Danhof, N. A., Tjon-Kon-Fat, R. I., et al. (2019). Interventions for unexplained infertility: A systematic review and network meta-analysis. Cochrane Database of Systematic Reviews, 2019(9).
Publisher | Google Scholor - Wessel, J. A., Mochtar, M. H., Besselink, D. E., et al. (2022). Expectant management versus IUI in unexplained subfertility and a poor pregnancy prognosis (EXIUI study): A randomized controlled trial. Human Reproduction, 37(12):2808–2816.
Publisher | Google Scholor - Zhao, J., Zhang, Q., Wang, Y., & Li, Y. (2014). Endometrial pattern, thickness and growth in predicting pregnancy outcome following 3319 IVF cycle. Reproductive BioMedicine Online, 29(3):291–298.
Publisher | Google Scholor