Research Article
Evaluation Of Response and Toxicity of Pemetrexed-Carboplatin Versus Paclitaxel-Carboplatin as First-Line Treatment in Metastatic Non-Squamous Non-Small Cell Lung Cancer
- Tasnim Mahmud *
Department of Epidemiology, North South University, Dhaka, Bangladesh.
*Corresponding Author: Tasnim Mahmud, Department of Epidemiology, North South University, Dhaka, Bangladesh.
Citation: Mahmud T. (2025). Evaluation Of Response and Toxicity of Pemetrexed-Carboplatin Versus Paclitaxel-Carboplatin as First-Line Treatment in Metastatic Non-Squamous Non-Small Cell Lung Cancer. Journal of Clinical Medicine and Practice, BioRes Scientia Publishers. 2(1):1-7. DOI: 10.59657/3065-5668.brs.25.019
Copyright: © 2025 Tasnim Mahmud, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: February 17, 2025 | Accepted: March 04, 2025 | Published: March 08, 2025
Abstract
Non-small cell lung cancer (NSCLC) accounts for 80-85% of all lung cancer approximately. The paclitaxel-carboplatin combination is the established standard regimen of choice in metastatic NSCLC. Pemetrexed, a folate antimetabolite is also effective against non-small cell lung cancer. To compare the response and toxicity of the Pemetrexed-Carboplatin regimen with Paclitaxel-Carboplatin in the treatment of metastatic NSCLC. This Quasi-experimental study was conducted from three centers of Dhaka city. 80 patients (40 patients on each arm) who met the inclusion criteria of the study were enrolled. Arm-A received 500mg/m2 Pemetrexed (Day 1) plus Carboplatin: AUC=5 (Day 1) IV, in another arm, 175mg/m2 Paclitaxel (Day 1) plus Carboplatin AUC=6; (Day 1) IV, dexamethasone was given 12 mg on night before and on the morning of chemotherapy of each cycle & repeated every 21 days for 6 cycles; were given. Both outcome and toxicities were evaluated. Regarding the tumor control, there was no statistically significant difference in both arms at the follow-up after 6 weeks of completion of chemotherapy [Partial response was seen in 24 (60.00%) patients in Arm-A and in 22 (55.00%) patients in Arm-B, p= 0.58]. Grade ≥3 neutropenia was seen in 09 (22.50%) patients of the Arm-A and 20 (50.00%) patients of Arm-B, p<0.005. Treatment-emergent alopecia was significantly higher in Arm-B [Arm-A, 07 (17.50%) vs Arm-B, 22 (55.00%) p<0.05]. Other non-hematological toxicity was also assessed in both arms and there was no significant difference in the frequency of adverse events. This study supports the fact that Pemetrexed-Carboplatin-based chemotherapy may be equally effective with less haematologic & non-haematologic toxicity than the Paclitaxel-Carboplatin-based chemotherapy regimen.
Keywords: response; toxicity; paclitaxel; carboplatin; nsclc
Introduction
Cancer is a group of diseases involving abnormal cell growth, which tends to proliferate uncontrolled and potentially invade surrounding structures or spread to other body parts. Due to lifestyle factors and aging of the population cancer is now an emerging health problem. According to GLOBOCON 2020, almost 19.3 million new cancer cases and 10 million deaths occurred in 2020 [1]. Lung cancer is one of the most frequently diagnosed cancers and is the leading cause of death worldwide [2]. More than 95% of cases are associated with smoking. Other risk factors included genetic predisposition, and occupational or environmental exposure to carcinogens, like arsenic, asbestos, beryllium, cadmium, chromium, nickel, radon, and vinyl chloride; asbestos is the most common [3]. In Bangladesh, the prevalence of SCLC is 10.20% and NSCLC is 84.80
Methods
This Quasi-Experimental study was conducted from October 2022 to September 2023. It was a multi-centered study conducted in the Department of Clinical Oncology, Bangabandhu Sheikh Mujib Medical University (BSMMU), Shahbagh, National Institute of Cancer Research & Hospital (NICRH), Mohakhali, and Delta Hospital Limited, Mirpur, Dhaka. A purposive sampling technique was applied. A total of 80 patients were selected according to inclusion and exclusion criteria. The patients of Arm A were treated with 4 cycles of the Pemetrexed plus Carboplatin-based regimen, whereas the patients of Arm B were treated with Paclitaxel plus Carboplatin-based regimen. Findings of observation were recorded in a semi-structured data collection form. Data were edited and analyzed according to the objectives and variables of the study by using the SPSS software program for Windows, version 23. Data were edited and analyzed according to the objectives and variables of the study by using the SPSS software program for Windows, version 23. Differences between the two means were assessed by student’s t-test. All outcomes were compared by chi-square test in the case of qualitative variables. Fisher’s exact test was done when grater than 20% of cells in the cross table had an expected frequency of less than 5. A p-value of less than 0.05 in the tailed test was considered statistically significant.
Inclusion Criteria
Clinically diagnosed and histopathologically proven non-squamous non-small cell carcinoma of the lung.
Histopathologically or cytopathologically proven metastatic disease (stage IV diseases).
Exclusion Criteria
Those who were not willing. Age less than 18 & less than 73 years. Patients with a history of prior chemotherapy or radiotherapy. Eastern Cooperative Oncology Group (ECOG) performance status more than 2. Initial surgery (excluding diagnostic biopsy) of the primary site. Patients with double primaries. Pregnant or lactating woman. Very serious co-morbidity.
Results
Table 1 resembles the baseline characteristic of the study population. It is evident that, there was homogeneous distribution of the study sample and that none of the baseline characteristics were significant.
Table 1: Baseline characteristics of the study population (n=80)
| Variables | Arm-A (n=40) | Arm-B (n=40) | T-test | p-value |
| Age (years) | 58.88 ± 6.56 | 59.75 ± 6.73 | -.589 | .558 |
| Weight (kg) | 59.90 ± 8.42 | 58.88 ± 6.97 | .593 | .555 |
| Height (cm) | 163.60 ± 7.52 | 162.35 ± 8.36 | .703 | .484 |
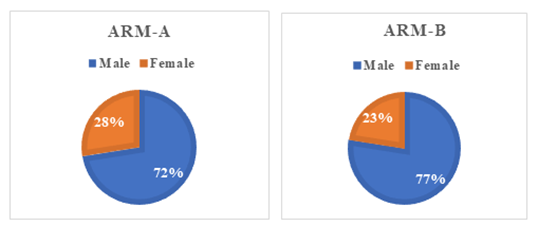
Figure 2: Distribution of patients according to gender in Arm A and Arm B (n=80)
Figure 2 shows the distribution of the patients according to gender in both arm A and arm B. Above Pie chart shows the distribution of the patients by gender in both Arms. 80 patients were included in Arm A and Arm B. They were divided into male and female groups, out of which in Arm A, 29 (72%) were male and 11 (28%) were female, and in Arm B, 31 were male (77%), and 9 (23%) were female. (p= 0.61). Table 2 illustrates the distribution of patients according to the risk factors. According to the table, 29 (72.5%) patients in Arm A and 27 (67.5%) patients in Arm B were smokers. A good number of patients were also associated with various lung diseases such as COPD, Asthma, TB etc in both arms.
Table 2: Distribution of patients according to the risk factors (n=80)
| Risk factors | Arm-A | Arm-B | Total |
p– value | ||||
| (n = 40) | % | (n = 40) | % | (n = 80) | % | |||
| TobaccoRelated | Smoking | 29 | 72.50 | 27 | 67.50 | 56 | 70.00 | 0.62 |
| Jarda | 21 | 52.50 | 23 | 57.50 | 44 | 55.00 | 0.65 | |
| Lung disease | COPD | 09 | 22.50 | 10 | 25.00 | 19 | 23.80 | 0.79 |
| Tuberculosis | 07 | 17.50 | 05 | 12.50 | 12 | 15.00 | 0.53 | |
| Co-Morbidities | HTN & DM | 19 | 47.50 | 17 | 42.50 | 36 | 45.00 | 0.65 |
| Occupation | Factory Worker | 07 | 17.50 | 09 | 22.50 | 16 | 20.00 | 0.44 |
| Firewood user | 14 | 35.00 | 11 | 27.50 | 25 | 31.25 | ||
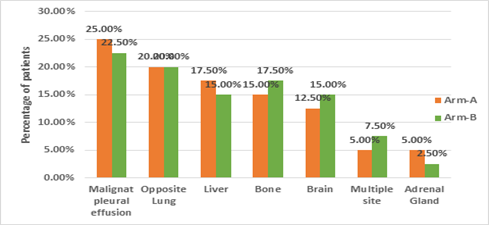
Figure 2: Distribution of patients according to site of metastasis.
Figure 2 shows the distribution of patients according to the site of metastasis. The above figure demonstrated that the most commonly observed site of metastasis in both arms was metastatic pleural effusion followed by opposite lung, bone, liver, multiple sites, and adrenal gland accordingly. p-value=0.99 (insignificant) displayed that the distribution of patients according to the site of metastasis was homogenous. Table 3 shows response evaluation after the 2nd, and 3rd cycles & 6 weeks after completion of chemotherapy. above table showed that, at mid-term evaluation after the completion of 2nd & 3rd cycle chemotherapy, no patients had a complete response, 23 (57.50%) patients had a partial response and 17 (42.50%) had stable disease in Arm A. Whereas in Arm B, no patients had complete response, 21 (52.50%) and 19 (47.50%) patients had partial response & stable disease respectively. On the other hand, there was no complete response in both arms. Partial response was 24 (60%) in Arm A and 22(55%) in Arm B. 15 (37.50%) patients presented with stable disease in Arm A whereas 15 (37.50%) in Arm B. Progressive disease was reported in 01 (02.50%) in Arm A and 03 (7.50%) in Arm B.
Table 3: Response evaluation after 2nd & 3rd Cycle &6 weeks after completion of chemotherapy (n=80)
| Response (2nd &3rd cycle) | Arm-A | Arm-B | Total | Chi-square Value | p – value | |||
| (n=40) | % | (n=40) | % | (n=80) | % | |||
| Complete response (CR) | - | - | - | - | - | - | ||
| Partial response (PR) | 23 | 57.50 | 21 | 52.50 | 44 | 55.00 | 0.202 | 0.65 |
| Stable disease (SD) | 17 | 42.50 | 19 | 47.50 | 36 | 45.00 | ||
| Response (6th week) | Arm A | Arm B | Total | Chi-Square Value | p-value | |||
| (n=4) | % | (n=4) | % | (n=80) | % | |||
| Complete response (CR) | - | - | - | - | - | - | ||
| Partial response (PR) | 24 | 60.00 | 22 | 55.00 | 46 | 57.50 | 1.08 | 0.58 |
| Stable disease (SD) | 15 | 37.50 | 15 | 37.50 | 30 | 37.50 | ||
| Progressive disease (PD) | 01 | 02.50 | 03 | 07.50 | 04 | 05.00 | ||
Table 4 shows overall acute hematological toxicities in both arms. It can be seen that none of the patients were spared from anemia. The severity of anemia was slightly higher in Arm B compared to Arm A 11(27.5%) patients developed Grade 2 or more anemia in Arm-A, whereas 14 (35%) patients in Arm-B. This finding was statistically insignificant between the two arms (p >0.05). Leucopenia of various grades was predominant in both the Arms. It was seen that Grade 3 or more leucopenia was seen in 09 patients (22.50%) vs 21 patients (52.50%) among Arm A and B respectively. The finding was statistically significant (p <0>p <0>p >0.05). Grade 3 febrile neutropenia was seen to be more in Arm B than Arm A. 05 (12.50%) patients in Arm A, whereas 08 (20.00%) in Arm B developed grade 3 febrile neutropenia respectively. The finding was statistically insignificant (p >0.05).
Table 4: Overall acute hematological toxicities in both arms (n=80)
| Hematological toxicities | Arm A | Arm B | Total | Chi-Square Value | p – value | |||
| n=40 | % | n=40 | % | n=80 | % | |||
| Anemia | ||||||||
| Grade 0 | 04 | 10.00 | 03 | 7.50 | 07 | 8.75 | ||
| Grade 1 | 25 | 62.50 | 23 | 57.50 | 48 | 60.00 | ||
| Grade 2 | 08 | 20.00 | 10 | 25.00 | 18 | 22.50 | 0.648 | 0.95 |
| Grade 3 | 02 | 5.00 | 03 | 7.50 | 05 | 6.25 | ||
| Grade 4 | 01 | 2.50 | 01 | 2.50 | 02 | 2.25 | ||
| Leucopenia | ||||||||
| Grade 0 | 06 | 15.00 | 02 | 5.00 | 08 | 10.00 | ||
| Grade 1 | 16 | 40.00 | 07 | 17.50 | 23 | 28.75 | 10.48 | 0.033 |
| Grade 2 | 09 | 22.50 | 10 | 25.00 | 19 | 23.75 | ||
| Grade 3 | 07 | 17.50 | 15 | 37.50 | 22 | 27.50 | ||
| Grade 4 | 02 | 5.00 | 06 | 15.00 | 08 | 10.00 | ||
Table 5 illustrates overall acute non-hematological (alimentary) toxicities observed during chemotherapy. Incidence of nausea was almost similar on both arms. 12 (30.00%) and 01 (02.50%) patients in Arm A, whereas 10 (25%) and 01 (2.50%) patients in Arm B developed grade 2 and grade 3 nausea. The incidence of vomiting and diarrhea were also similar in both arms. Grade 2/3 mucositis was similar in Arm A and Arm B, 04 (10.00%) and 04(10.00%).
Table 5: Overall acute non-hematological (alimentary) toxicities observed during Chemotherapy (n=80)
| Toxicities | Arm A | Arm B | Total | Chi-square value | p – value | |||
| n=40 | % | n=40 | % | n=80 | % | |||
| Nausea | ||||||||
| Grade 0 | 04 | 10.00 | 08 | 20.00 | 12 | 15.00 | ||
| Grade 1 | 23 | 57.50 | 21 | 52.50 | 44 | 55.00 | 1.6 | 0.65 |
| Grade 2 | 12 | 30.00 | 10 | 25.00 | 22 | 27.50 | ||
| Grade 3 | 01 | 2.50 | 01 | 2.50 | 02 | 02 2.50 | ||
| Vomiting | ||||||||
| Grade 0 | 26 | 65.00 | 25 | 62.50 | 51 | 63.75 | ||
| Grade 1 | 11 | 27.50 | 12 | 30.00 | 23 | 28.75 | 0.06 | 0.96 |
| Grade 2 | 03 | 7.50 | 03 | 7.50 | 06 | 7.50 | ||
| Diarhoea | ||||||||
| Grade 0 | 26 | 65.00 | 25 | 62.50 | 51 | 63.75 | ||
| Grade 1 | 11 | 27.50 | 13 | 32.50 | 24 | 30.00 | 0.52 | 0.91 |
| Grade 2 | 02 | 5.00 | 01 | 2.50 | 03 | 3.75 | ||
| Grade 3 | 01 | 2.50 | 01 | 2.50 | 02 | 2.50 | ||
| Mucositis | ||||||||
| Grade 0 | 25 | 62.50 | 26 | 65.00 | 51 | 63.75 | ||
| Grade 1 | 11 | 27.50 | 10 | 25.00 | 21 | 26.25 | 1.2 | 0.75 |
| Grade 2 | 03 | 7.50 | 04 | 10.00 | 07 | 8.75 | ||
| Grade 3 | 01 | 2.50 | 00 | 00 | 01 | 1.25 | ||
Discussion
Patients from various medical centers across Dhaka who met the study's specific criteria were included in this research endeavor. In this study, the average age of patients at the time of diagnosis was 59.32±6.70. Tao Chen et al. (2019) revealed the average age of patients at diagnosis of NSCLC was 68 years [12]. Interestingly, the result of this study closely mirrors the data from the Cancer registry report (2015-2017) of NICRH, Dhaka, which reported the average age of patients at diagnosis was 58.85±12.14. This alignment between this study's findings and the registry's data adds further depth to our understanding of the patient demographics in this context.
Out of the 80 patients under scrutiny, a substantial majority, comprising 60 (75%) individuals were male, while the remaining 20 (25%) patients were female. The overarching gender ratio, therefore, stands at an appreciable 3:1, firmly indicating a pronounced male predominance within this patient cohort. Notably, this observation echoes the findings of Ruquiya Afrose et al. (2015) in their study, which discerned a higher incidence among males as compared to females, with a distribution of 81% and 19% respectively. Cancer Registry Report (2015-2017) of NICRH observed higher incidence of Lung cancer among males 84.50% than females 14.50% [13].
The analysis encompassed an assessment of multiple risk factors, with smoking emerging as the foremost global contributor to lung cancer. Culminating in a total of 56 patients (70%) among the entire study population who had a history of smoking. It is noteworthy that smoking has been unequivocally established as the most significant risk factor for non-small cell lung cancer, as delineated by the American Cancer Society in 2019 [14].
The partial response rate for large cell carcinoma was 0% in Arm A and 66.70% in Arm B. In Arm A, 50.00% of patients exhibited stable disease, while in Arm B, this percentage was 33.30%. Progressive disease was observed in 50.00% of patients in Arm A and 0% in Arm B. Jules et al. (2017) observed in their research that Paclitaxel based chemotherapy showed superior overall survival than Pemetrexed based chemotherapy [15].
In the assessment of hematological toxicities, the study found that various acute hematological toxicities were commonly observed, including anemia, leucopenia, neutropenia, and thrombocytopenia. In particular, when comparing Arm-B to Arm-A, there were statistically significant differences in the incidence of grade 3/4 leucopenia (52.50% in Arm-B compared to 22.50% in Arm-A, p= less than 0.05) and neutropenia (50% in Arm-B compared to 22.50% in Arm-A, p= <0>
Conclusion
According to this study it may be concluded that Pemetrexed plus Carboplatin-based chemotherapy regimen was equally effective with less haematologic as well as non-haematologic toxicity in comparison to Paclitaxel plus Carboplatin based chemotherapy regimen.
References
- Sung H, Ferlay J, Siegel RLb, Laversanne M, Soerjomataram I, et al. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA a cancer journal for clinicians, 71(3):209-249.
Publisher | Google Scholor - Witschi H. (2001). A short history of lung cancer. Toxicological sciences, 64(1):4-6.
Publisher | Google Scholor - Ganie FA, Wani ML, Lone H, Wani SN, Hussain SA. (2013). Carcinoma lung: Clinical presentation, diagnosis, and its surgical management. The Journal of Association of Chest Physicians, 1(2):38-43.
Publisher | Google Scholor - Stoddard N, Heil JR, Lowery DR. (2019). Anatomy, thorax, mediastinum. StatPearls,
Publisher | Google Scholor - Dietel M, Bubendorf L, Dingemans AM, Dooms C, Elmberger G, et al. (2016). Diagnostic procedures for non-small-cell lung cancer (NSCLC): recommendations of the European Expert Group. Thorax, 71(2):177-184.
Publisher | Google Scholor - Dietel M, Bubendorf L, Dingemans AM, Dooms C, Elmberger G, et al. (2016). Diagnostic procedures for non-small-cell lung cancer (NSCLC): recommendations of the European Expert Group. Thorax, 71(2):177-184.
Publisher | Google Scholor - Casal-Mouriño A, Ruano-Ravina A, Lorenzo-González M, Rodríguez-Martínez Á, Giraldo-Osorio A, et al. (2021). Epidemiology of stage III lung cancer: frequency, diagnostic characteristics, and survival. Translational Lung Cancer Research, 10(1):506.
Publisher | Google Scholor - Aarts MJ, van den Borne BE, Biesma B, Kloover JS, Aerts JG, et al. (2015). Improvement in population‐based survival of stage IV NSCLC due to increased use of chemotherapy. International Journal of Cancer, 136(5):387-395.
Publisher | Google Scholor - Aarts MJ, van den Borne BE, Biesma B, Kloover JS, Aerts JG, et al. (2015). Improvement in population‐based survival of stage IV NSCLC due to increased use of chemotherapy. International Journal of Cancer, 136(5):387-395.
Publisher | Google Scholor - Horiuchi K, Sato T, Kuno T, Takagi H, Hirsch FR, rt al. (2021). Platinum-doublet chemotherapy as second-line treatment for relapsed patients with small-cell lung cancer: A systematic review and meta-analysis. Lung Cancer, 156:59-67.
Publisher | Google Scholor - Azzoli C.G, Baker Jr S, Temin S, Pao W, Aliff T, et al. (2009). American Society of Clinical Oncology clinical practice guideline update on chemotherapy for stage IV non–small-cell lung cancer. Journal of clinical oncology, 27(36):6251.
Publisher | Google Scholor - Chen T, Zhou F, Jiang W, Mao R, Zheng H, et al. (2019). Age at diagnosis is a heterogeneous factor for non-small cell lung cancer patients. Journal of Thoracic Disease, 11(6):2251.
Publisher | Google Scholor - Afrose R, Akram M, Karimi AM, Siddiqui SA. (2015). Correlation of age and gender with different histological subtypes of primary lung cancer. Medical Journal of Dr. DY Patil University, 8(4):447-451.
Publisher | Google Scholor - Schabath MB, Cote ML. (2019). Cancer progress and priorities: lung cancer. Cancer epidemiology, biomarkers & prevention, 28(10):1563-1579.
Publisher | Google Scholor - Derks JL, Van Suylen RJ, Thunnissen E, Den Bakker MA, Groen HJ, et al. (2017). Chemotherapy for pulmonary large cell neuroendocrine carcinomas: does the regimen matter? European Respiratory Journal, 49(6).
Publisher | Google Scholor - Gaafar RM, Surmont VF, Scagliotti GV, Van Klaveren RJ, Papamichael D, et al. (2011). EORTC Lung Cancer Group. A double-blind, randomised, placebo-controlled phase III intergroup study of gefitinib in patients with advanced NSCLC, non-progressing after first line platinum-based chemotherapy (EORTC 08021/ILCP 01/03). European Journal of Cancer, 47(15):2331-2340.
Publisher | Google Scholor