Research Article
Evaluation of Paclitaxel as a Non-Cross-Resistant Agent in Cisplatin-Resistant Small Cell Lung Cancer
- Fahima Tabassum 1,2*
1Internal Medicine Department, Shimane University Graduate School of Medicine, Izumo, Shimane, Japan.
2Department of Biological Science, The University of Osaka, Graduate School of Science, Suita city, Osaka, Japan.
*Corresponding Author: Fahima Tabassum, The University of Osaka, Graduate School of Science, Suita city, Osaka, Japan.
Citation: Tabassum F. (2025). Evaluation of Paclitaxel as a Non-Cross-Resistant Agent in Cisplatin-Resistant Small Cell Lung Cancer, Clinical Case Reports and Studies, BioRes Scientia Publishers. 10(6):1-6. DOI: 10.59657/2837-2565.brs.25.277
Copyright: © 2025 Fahima Tabassum, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: September 09, 2025 | Accepted: September 24, 2025 | Published: October 01, 2025
Abstract
Small cell lung cancer (SCLC) is an aggressive disease with early metastasis and poor prognosis. Etoposide and cisplatin/carboplatin are used in the first-line treatment of SCLC. Amrubicin is the only established second-line treatment for SCLC only in Japan but not in western world. With this because there is sever need of active agent for second-line treatment of therapy-resistant SCLC. Currently, paclitaxel has shown a degree of cytotoxic activity against SCLC only in phase II studies. This research aims to establish human SCLC cell lines resistant to cisplatin and evaluate the efficacy of paclitaxel on established cisplatin-resistant cell lines. I cultured human SCLC cell lines N417 and H82 and treated with different concentrations of cisplatin and performed WST-8 assay for comparing effectivity of cisplatin and paclitaxel. Parental N417 cells were passaged more than 50 times for establishing cisplatin-resistant cell line N417/CDDP; however, H82 cells were highly sensitive towards cisplatin. Although we did not obtain H82-derived resistant cell line, parental H82 cells started to show sensitivity from 0.3 μM, and we evaluate cell viability with 0.5 μM cisplatin-treated H82 cells for comparing. We found cisplatin decrease the cell viability only for parental N417 and H82 cell lines but not for N417/CDDP and 0.5 μM cisplatin-treated H82 cell line, but paclitaxel shows more effective response not only parental cell lines but also cisplatin resistant and treated cell lines.
Keywords: cisplatin-resistant cell line; SCLC; paclitaxel; cytotoxicity; paclitaxel; parental cells
Introduction
Small cell lung cancer (SCLC) is a type of lung cancer that has a high growth fraction, rapid doubling time, early development of extensive metastases, and an initial response to chemotherapy and radiation [1]. SCLC is prevalent in 15% of patients diagnosed with lung cancer, and develops from neuroendocrine precursor cells. At the time of initial diagnosis, about 10-25% of patients have brain metastases; an additional 40-50% will acquire them as their disease progresses [2]. Currently, platinum (cisplatin or carboplatin) plus etoposide, including programmed death ligand 1-targeted immune checkpoint inhibitors (atezolizumab or durvalumab), is the preferred first-line treatment for metastatic SCLC [4]. Unfortunately, though the rapid and high responses to initial therapy, a multifocal relapse is often occurred and limits on the efficacy of first-line therapies for many patients with SCLC [7]. There are very few drugs which are identically established for generating remission in relapsed SCLC [8]. Amrubicin is a drug for second-line therapy that has shown superiority in effectivity to topotecan in a Japanese population but was not considered superior in a study of western patients [9]. In this situation, it is a great need for new active agents in the setting of second line treatment for therapy-resistant in first-line therapy. Moreover, establishing of such drugs may lead to the development of more potent first-line regimens [3]. For this reason, it appears important to determine which tumor patients are sensitive to and resistant to the cytotoxic drugs utilized during the induction phase of their cancer [3].
A variety of solid malignancies, such as testicular, ovarian, head and neck, colorectal, bladder, and lung cancers, are treated with platinum-based medications, most notably cisplatin [5]. Cisplatin is a key drug with high anticancer activity for the systemic treatment of SCLC. The cytotoxic mechanism of cisplatin is triggered by its interaction with DNA to generate adducts that cause programmed cell death or apoptosis [6]. One hand, paclitaxel is an antimicrotubular agent which inhibit mitotic spindle assembly and only has shown a degree of activity in small phase II studies in small-cell lung cancer [3,9]. It was originally derived from the bark of the Pacific yew tree and can be partially synthesized from the precursor 10-deactylbaccatin III, derived from the needles of the European yew [2]. Paclitaxel binds to tubulin to stabilize the microtubule and inhibit its disassembly, ultimately leading to cell death by apoptosis. The clinical use of paclitaxel is limited by tumor resistance, risk of hypersensitivity reactions, and toxicity [11]. With a response rate of 29%, single-agent paclitaxel has shown an activity among patients with SCLC resistant to cyclophosphamide, doxorubicin, and etoposide in a clinical trial [3]. Moreover, paclitaxel is considered to have cytotoxic activity against several drug-resistant tumors such as platinum-resistant ovarian cancer and anthracycline-resistant breast cancer. (Rowinsky et al,1994). In a previous study, almost 40% of acquired cisplatin-resistant cells were not resistant to paclitaxel, and only 28% shows hypersensitivity to paclitaxel, suggesting that patients with cisplatin-resistant cancers can benefited from paclitaxel treatment because as developing resistance to one drug can be different to other drugs [12].
The purpose of this study was to evaluate the efficacy of paclitaxel, whether it can be considered as a non-cross-resistant agent to cisplatin-resistant small cell lung cancer.
Materials and Methods
Cell Culture: Two human small lung cancer cell lines, NCI-N417 (N417) and NCI-H82 (H82), were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA) and used as parental cell lines. The N417 cell line was isolated from the lungs of a patient with small cell lung cancer and the H82 cell line was derived from the pleural fluid of a patient with small cell lung cancer. For passage, 2.5×105 or 5×105 cells were seeded into T-25 tissue culture flasks. The cells were cultured in RPMI-1640 (Fujifilm Wako Pure Chemical Industries, Osaka, Japan) growth medium supplemented with 10 percentage fetal bovine serum and incubated at 37°C in a humidified incubator with 5% CO2. Paclitaxel and cisplatin were diluted with phosphate-buffered saline (PBS).
Establishment of Cisplatin-Resistant Human Cell Line: To establish cisplatin-resistant SCLC cell lines, N417 cells were cultured in T-25 tissue culture flasks in the continuously presence of cisplatin. The drug concentration was gradually increased by approximately 3-fold when the cell growth interval from seeding to ≥ 70% confluence was less than 6 days without obvious changes in the cell shape and increasing the number of floating cells after passage. The cells were confirmed to proliferate after the concentration of cisplatin was increased; otherwise, they were cultured with cisplatin at the same concentration used in the previous step.
Cell Viability Assay: Cultured cells were seeded in 96-well tissue culture plates (5000 cells/well) with 200 μL of medium per well. Cell viability was determined by the WST-8 assay using the Cell Counting Kit-8 (Dojindo, Kumamoto, Japan), according to the manufacturer’s instructions. Absorbance was measured at 450 nm using a Tecan Sunrise microplate reader (Tecan, Männedorf, Switzerland). To determine the relative cell viability, the absorbance of the samples was divided by that of the vehicle control.
Results
Cultured Cells of Parental and Cisplatin-Resistant N417 Cell Lines
To compare the sensitivity of cisplatin-resistant and parental N417 to cisplatin, we established cisplatin-resistant cell line, N417/CDDP from parental N417. There seems different condition of cells between the parental cell-seeded flask and drug-treated flasks because of the continuous treatment of cisplatin. For observing the drug response towards cells, the cells were passaged and treated at least 2-3 times for each concentration. The initial concentration was 1 μM which was increased by determining cell proliferation rate and condition. The cells were passaged 55 times to establish cisplatin-resistant N417/CDDP cell line.
Figure 1: Different cell condition after passaging into flask. A. Parental N417 cell confluency after passaging. B. Established cisplatin resistant N417/CDDP cell line.
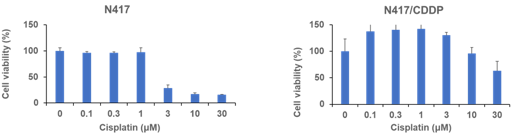
Comparison of Cisplatin Sensitivity Between Parental N417 and Cisplatin-Resistant N417/CDDP
To compare the sensitivity of cisplatin-resistant and parental N417 lines to cisplatin, we established cisplatin-resistant cell line, N417/CDDP from parental human N417 cell line. We can observe from the graph that cisplatin shows effectivity on parental N417 cell line but not on cisplatin-resistance N417/CDDP cell line. We evaluated the cell viability. Parental cells N417 started to show drug effectivity from 3 μM whereas if we observe the same drug concentration effect in cisplatin-resistant cell line we found that there seems not much effectivity for the same drug concentration towards resistant cells. Cell viability didn’t change as much as parental cell line.
Figure 2: Cell viability of human SCLC cell line parental N417 and established cisplatin resistant N417/CDDP cell line determined using WST-8 assay. Results represent the mean ±SD. (A) Viability of N417 cells treated with indicated concentrations of cisplatin for 120h. (B) Viability of N417/CDDP cells with same drug concentration and treatment time.
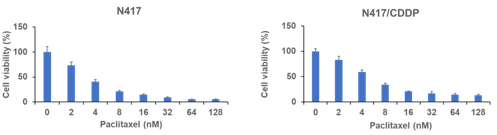
Comparison of Paclitaxel Sensitivity Between Parental N417 and Cisplatin-Resistant N417/CDDP
We also performed the WST-8 assay of paclitaxel towards parental N417 and cisplatin-resistant N417/CDDP for evaluating the effectivity comparing to cisplatin. Here we found that paclitaxel shows not only effectivity against parental cells, but also cisplatin-resistant cells compared to cisplatin effectivity against cisplatin-resistant cells. From the graph of parental N417 cell line, the drug started to show effective response from 8 nM and similar response with the same drug concentration against cisplatin-resistant cell line. From comparing our observation of drug effectivity towards parental and cisplatin-resistant cell line, cisplatin did not affect to resistant cell line N417/CDDP, but paclitaxel showed effective response against N417/CDDP.
Figure 3: Cell viability of human SCLC cell line parental N417 and established cisplatin resistant N417/CDDP cell line determined using WST-8 assay. Results represent the mean ±SD. (A) Viability of N417 cells treated with indicated concentrations of paclitaxel for 72h. (B) Viability of N417/CDDP cells with same drug concentration and treatment time.
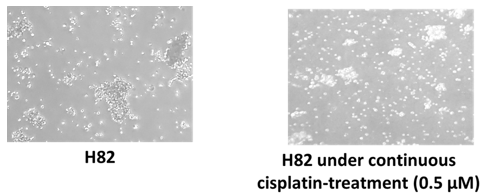
Cultured Cells of Parental and Cisplatin-Treated H82 Cell Lines
For establishing cisplatin-resistant H82/CDDP cell line, we started cisplatin treatment to parental H82 cell line from 0.1 μM and gradually increase and observe that cells started to show sensitivity within 0.3 μM drug concentration and the growth rate of cells were 3 times slower than N417 cell line. We treated the cells at least 3 times with same drug concentration before increasing. For sensitivity towards cisplatin, we can increase drug concentration up to 0.5 μM. When we tried to increase drug concentration from 0.5 μM to 0.7 μM we found dead cells floating inside the flask for cisplatin high sensitivity. In 0.5 μM CDDP treated flask, we found better condition of cells comparing to 0.7 μM CDDP treated flask. Considering the condition of cells, it was hard to obtain cisplatin-resistance cell line H82/CDDP from parental H82.
Figure 4: Different cell condition after passaging into flask. A. Parental H82 cell confluency in flask after passaging. B. 0.5 μM cisplatin treated H82 cell line.
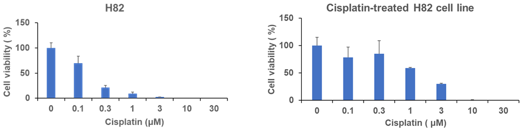
Comparison of Cisplatin Sensitivity Between Parental H82 Cell Line and Cisplatin-Treated 0.5 μM H82 Cell Line
For H82 cell line, we also performed cell viability assay for comparing the paclitaxel and cisplatin effectivity. In these experiments, we can see that in parental H82 cell line cisplatin started to show effective response from 0.3 μM. The cells changed the cell shape and showed sensitivity response. For evaluating the effect of cisplatin on cisplatin-treated cells we perform cell viability assay with the maximum drug concentration 0.5 μM H82 cell line and similarly compare the cell viability. In this case, we observed that, cisplatin shows effective response on parental cell line from 0.3 μM but for drug treated cells effective response starts from 3 μM, which indicates that cisplatin shows not much effective response to 0.5 μM cisplatin treated cells comparing to parental cells.
Figure 5: Cell viability assay of parental H82 cell line determined by WST-8 assay. Results represent the mean ± SD. (A) Viability of H82 cells treated with indicated concentrations of cisplatin for 120h. (B) Viability of H82 cells treated with indicated concentrations of cisplatin for 72h.
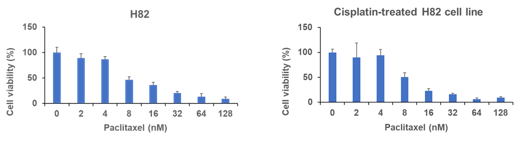
Comparison of Paclitaxel Effectivity Between Parental H82 Cell Line and Cisplatin-Treated 0.5 μM H82 Cell Line
Evaluating the paclitaxel effectivity on parental H82 and 0.5 μM cisplatin treated H82 cell line we performed similar cell viability assay with same drug concentration and treatment time as previous experiments. For paclitaxel treatment, the cells started to show sensitivity from 16 nM, in parental H82 cell line and for 0.5 μM cisplatin treated cells the effective response starts also from 16 nM which can also confirm the effective response of paclitaxel against parental H82 cell line.
Figure 6: Cell viability assay of H82 cell line determined by WST-8 assay. Results represent the mean ± SD. (A) Viability of H82 cells treated with indicated concentrations of cisplatin for 120h. (B) Viability of 0.5 μM cisplatin-treated H82 cells treated with indicated concentrations of cisplatin for 72h.
Discussion
One of the major problems in the management of SCLC is relapse due to drug resistance. Generally, the mode of action of cisplatin involves the generation of DNA lesions, followed by activation of the DNA damage response and induction of mitochondrial apoptosis [3]. Although the mechanisms of cisplatin sensitivity in our cell lines were not determined, differences in cisplatin sensitivity and mechanisms may be due to the mode of action of cisplatin, and several factors associated with cisplatin resistance have been previously reported. Cisplatin causes DNA-cisplatin adducts and DNA damage and inactivates the tumor-suppressive protein TP53, followed by activation of the DNA damage response and induction of mitochondrial apoptosis. ERCC1 is a protein that forms with ERCC4 and incises DNA on the 5’ side of bulky lesions, such as cisplatin adducts. ERCC1 expression is negatively correlated with cisplatin response in multiple human cancers. One of the copper transporters, CTR1 is a major influx transporter that plays an important role in mediating the uptake of platinum compounds. Cisplatin uptake is mediated by CTR1 in both yeast and mammals. Cells with increased CTR1 expression exhibited increased platinum accumulation and, in most instances, increased sensitivity to cisplatin. Additionally, MRP2 is overexpressed in several cisplatin-resistant cell lines. MRP2 expression levels affect the efficacy of cisplatin regimens in esophageal squamous cell carcinoma and hepatocellular carcinoma [5].
Only a few drugs that can be used in combination with platinum-based drugs are effective in SCLC treatment. N417 cells were passaged more than 50 times to establish the cisplatin-resistant cell line N417/CDDP, but an H82-derived cisplatin-resistant cell line has not yet been established. We found that the parental H82 cell line was much more sensitive to cisplatin than the parental N417 cell line, and it was difficult to obtain a cisplatin-resistant cell line from the H82 cell line because cells started to show sensitivity within 0.3 μM. Comparing the cell viability assay of parental H82, N417, 0.5 μM cisplatin treated H82, and cisplatin-resistant N417/CDDP, we observe the cell viability and found that paclitaxel shows much more effectivity for not only parental N417, H82 cells but also and cisplatin-resistant N417/CDDP and 0.5 μM cisplatin-treated H82. Although cisplatin is the first-line treatment for SCLC, it is not sufficiently effective for therapy-resistant SCLC. In this study, we observed effective responses to paclitaxel treatment in the cisplatin-naïve parental cell lines N417 and H82, cisplatin-resistant N417/CDDP cells, and cisplatin-treated H82 cells. In conclusion, we have considered paclitaxel as a candidate drug for cisplatin-resistant SCLC.
Subsequently, it is planned to establish cell lines resistant to other platinum-based drugs (carboplatin and etoposide) to evaluate the efficacy of paclitaxel against these drug-resistant cell lines. In addition, animal experiments using a xenograft mouse model would be useful for measuring the in vivo paclitaxel activity in human SCLC tumors [13,14].
Conclusion
We found cisplatin decrease the cell viability only for parental N417 and H82 cell lines but not for N417/CDDP and 0.5 μM cisplatin-treated H82 cell line, but paclitaxel shows more effective response not only parental cell lines but also cisplatin resistant and treated cell lines.
Declarations
Acknowledgement
I want to thank Almighty, my parents and The University of Osaka, for supporting to complete the research.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest
No conflict of interest to declare.
Ethical Statement
The author is accountable of all aspects of the work in ensuring that the question related to the accuracy and integrity of any part of the work are appropriately investigated and solved.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request. A copy of the thesis will also be deposited in the Shimane University library repository following institutional requirements.
References
- Govindan, R., Page, N., Morgensztern, D., Read, W., Tierney, R., et al. (2006). Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. Journal of Clinical Oncology, 24(28):4539-4544.
Publisher | Google Scholor - Hiddinga, B. I., Raskin, J., Janssens, A., Pauwels, P., Van Meerbeeck, J. P. (2021). Recent developments in the treatment of small cell lung cancer. European Respiratory Review, 30(161).
Publisher | Google Scholor - Smit, E. F., Fokkema, E., Biesma, B., Groen, H. J. M., Snoek, W., et al. (1998). A phase II study of paclitaxel in heavily pretreated patients with small-cell lung cancer. British Journal of Cancer, 77(2):347-351.
Publisher | Google Scholor - Mascaux, C., Paesmans, M., Berghmans, T., Branle, F., Lafitte, J. J., et al. (2000). A systematic review of the role of etoposide and cisplatin in the chemotherapy of small cell lung cancer with methodology assessment and meta-analysis. Lung Cancer, 30(1):23-36.
Publisher | Google Scholor - Galluzzi, L., Senovilla, L., Vitale, I., Michels, J., Martins, I., et al. (2012). Molecular mechanisms of cisplatin resistance. Oncogene, 31(15):1869-1883.
Publisher | Google Scholor - Brown, A., Kumar, S., Tchounwou, P. B. (2019). Cisplatin-based chemotherapy of human cancers. Journal of Cancer Science & Therapy, 11(4):97.
Publisher | Google Scholor - Gong, J., Salgia, R. (2018). Managing patients with relapsed small-cell lung cancer. Journal of Oncology Practice, 14(6):359-366.
Publisher | Google Scholor - Kosmas, C., Tsavaris, N. B., Malamos, N. A., Vadiaka, M., Koufos, C. (2001). Phase II study of paclitaxel, ifosfamide, and cisplatin as second-line treatment in relapsed small-cell lung cancer. Journal of Clinical Oncology, 19(1):119-126.
Publisher | Google Scholor - Asai, N., Ohkuni, Y., Kaneko, N., Yamaguchi, E., Kubo, A. (2014). Relapsed small cell lung cancer: treatment options and latest developments. Therapeutic Advances in Medical Oncology, 6(2):69-82.
Publisher | Google Scholor - Kampan, N. C., Madondo, M. T., McNally, O. M., Quinn, M., Plebanski, M. (2015). Paclitaxel and its evolving role in the management of ovarian cancer. BioMed Research International, 1:413076.
Publisher | Google Scholor - Morris, P. G., Fornier, M. N. (2008). Microtubule active agents: beyond the taxane frontier. Clinical Cancer Research, 14(22):7167-7172.
Publisher | Google Scholor - Stordal, B., Hamon, M., McEneaney, V., Roche, S., Gillet, J. P., et al. (2012). Resistance to paclitaxel in a cisplatin-resistant ovarian cancer cell line is mediated by P-glycoprotein. PloS One, 7(7):e40717.
Publisher | Google Scholor - Isobe, T., Onn, A., Morgensztern, D., Jacoby, J. J., Wu, W., et al. (2013). Evaluation of novel orthotopic nude mouse models for human small-cell lung cancer. Journal of Thoracic Oncology, 8(2):140-146.
Publisher | Google Scholor - Tanino, R., Amano, Y., Tong, X., Sun, R., Tsubata, Y., et al. (2020). Anticancer activity of ZnO nanoparticles against human small-cell lung cancer in an orthotopic mouse model. Molecular Cancer Therapeutics, 19(2):502-512.
Publisher | Google Scholor