Review Article
Digital Technologies in Maxillofacial Prosthetics: Evolution from Conventional Methods to CAD/CAM Systems
- Ravi Marballi Basavaraju
- Anupama Aradya *
- Nisarga Vinodkumar
Department of Prosthodontics, JSS Dental College and Hospital, Mysore, Karnataka, India.
*Corresponding Author: Anupama Aradya, Department of Prosthodontics, JSS Dental College and Hospital, Mysore, Karnataka, India.
Citation: Ravi MB, Aradya A, Vinodkumar N. (2026). Digital Technologies in Maxillofacial Prosthetics: Evolution from Conventional Methods to CAD/CAM Systems, Dentistry and Oral Health Care, BioRes Scientia Publishers. 5(1):1-8. DOI: 10.59657/2993-0863.brs.26.053
Copyright: © 2026 Anupama Aradya, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: November 07, 2025 | Accepted: December 29, 2025 | Published: January 06, 2026
Abstract
Maxillofacial prosthetics has undergone a revolutionary transformation with the integration of digital technologies. This review examines the evolution from conventional labor-intensive fabrication methods to computer-aided design and manufacturing (CAD/CAM) systems. Traditional approaches, while reliable, present significant limitations including extended clinical time, patient discomfort, and dependence on artistic skill. Digital workflows offer enhanced accuracy, improved reproducibility, reduced patient burden, and the ability to create permanent archives for future reproductions. This manuscript explores the historical context, conventional limitations, fundamental principles of CAD/CAM technology, and the basic workflow of digital prosthesis fabrication. Understanding these foundational concepts is essential for clinicians transitioning to digital practice paradigms.
Keywords: maxillofacial prosthesis; digital dentistry; CAD/CAM; digital workflow
Introduction
Historical Perspective and The Golden Age of Dentistry
Dentistry has evolved remarkably since the eighteenth century, when impressions were made using waxes and plaster of Paris, and dental equipment consisted of hand-driven and later water-driven motors. This evolutionary journey has culminated in contemporary dental practice offering near-natural aesthetics, enhanced precision, reduced treatment time, minimized error potential, and superior quality assurance-characteristics that define the present era as the "Golden Age of Dentistry" [1]. Digitization has become integral to contemporary prosthodontics, with most procedures likely to be based on digital techniques in the near future. From radiographs and photographs to impression making, jaw movement recording, and prosthesis fabrication, digital technology has revolutionized every aspect of dental practice [1].
Maxillofacial Defects: The Clinical Challenge
Maxillofacial defects resulting from trauma, cancer treatment, and congenital diseases present significant aesthetic and prosthetic challenges [2]. While these defects can be addressed through surgical reconstruction or prosthetic rehabilitation, surgical approaches are often technically demanding with limited documentation of successful outcomes in large patient populations [2]. Consequently, prosthetic reconstruction is frequently preferred, with patients reporting high satisfaction with prosthodontic rehabilitation.
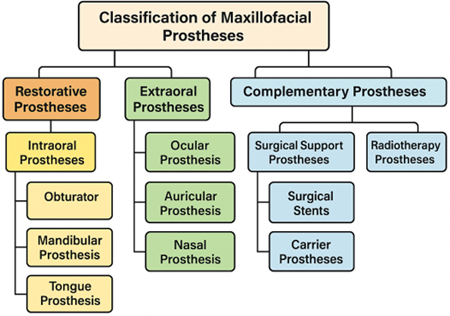
Figure 1: Classification scheme of maxillofacial prostheses. Hierarchical diagram showing main categories: Restorative prostheses (subdivided into intraoral: obturators, mandibular, tongue prostheses; and extraoral: ocular, auricular, nasal, facial prostheses) and Complementary prostheses (subdivided into surgical support and radiotherapy prostheses).
Conventional Indications for Prosthetic Intervention
Maxillofacial prosthetics traditionally serves patients where surgical reconstruction is contraindicated or impractical [3]. Common indications include:
- Extensive deformities with poor surgical prognosis.
- Compromised blood supply in post-radiation tissue.
- Advanced patient age limiting surgical tolerance.
- Poor general health status.
- Patient refusal of additional surgery.
- Displaced facial bones requiring temporary prosthetic support.
History and Development
Evolution of Fabrication Techniques
The journey from conventional hand-crafted prostheses to digital fabrication represents decades of technological advancement. Early pioneers in the late 1990s began exploring three-dimensional imaging and computer-aided techniques for maxillofacial prosthetics [4-6]. Penkner et al. (1999) first described the process of fabricating auricular prostheses using three-dimensional soft tissue models from computerized tomography scans [4]. Coward et al. (1999) pioneered the use of stereolithography for creating mirror-image wax models of ears using magnetic resonance imaging data [5].
Early Digital Applications (2000-2010)
The early 2000s witnessed rapid development in optical data acquisition and computer-assisted design systems. Runte et al. (2002) established systems for optical data acquisition using phase-measuring profilometry for facial prostheses [6]. Reitemeier et al. (2004) described structured light illumination techniques for orbital prosthesis fabrication [7]. The introduction of CAD/CAM systems in 2004 by Jiao et al. demonstrated the feasibility of designing and fabricating auricular prostheses using spiral CT data and rapid prototyping [9].
Tsuji et al. (2004) described comprehensive fabrication processes involving three-dimensional morphological measurement, CAD design, and CAM fabrication of facial prostheses [10]. Ciocca and Scotti (2004) advanced the field by using laser scanners to develop integrated three-dimensional digital images for ear prosthesis fabrication [11].
Maturation Phase (2011-2015)
The 2011-2015 period marked significant refinement in digital workflows. Eggbeer et al. (2006) pioneered texture relief applications for digitally designed facial prostheses, addressing fine anatomical details [17]. Kimoto and Garrett (2007) validated three-dimensional digital photographic imaging systems for facial prosthesis applications [18]. Wu et al. (2008) introduced selective laser sintering technology for customized facial prosthesis fabrication [19].
The development of digital anatomical libraries by Reitemeier et al. (2013) represented a paradigm shift, providing reference models for nasal prostheses when bilateral defects precluded mirroring techniques [20].
Contemporary Era (2016-Present)
Recent advances have focused on complete digital workflows, improved materials, and integration of multiple technologies. The field has progressed from experimental applications to clinically validated protocols with documented success rates and patient satisfaction outcomes.
Discussion
Conventional Fabrication Methods
Traditional Workflow
The traditional manufacturing of silicone maxillofacial prostheses is inherently labor-intensive, comprising three distinct phases:
Preparation Phase: Planning prosthetic rehabilitation and collecting data through impressions and photographs of the affected area and healthy contralateral region. Impression materials range from soft/flexible hydrocolloid alginates and elastic silicone polymers to rigid plaster of Paris, selected according to defect site, size, and presence of undercuts.
Production Phase: Creating try-on models, typically in wax, using plaster casts of the defect. The model is fitted on the patient and adjusted according to individual needs and preferences. Maxillofacial technicians often utilize photographs, pre-operative molds, or contralateral structures as references for anatomical sculpting. The finalized try-on model is converted into silicone prosthesis using plaster molds and individually colored silicone materials through conventional flasking methods.
Placement Phase: The silicone prosthesis is placed on the patient and refined chairside through color adjustments and addition of lifelike details such as eyelashes, skin texture, and vascular patterns.
Limitations of Conventional Approaches
While this traditional workflow is reliable and typically produces satisfactory outcomes, it presents significant disadvantages across patient-related, technical, and material domains [3].
Patient-Related Limitations
- Multiple uncomfortable impression procedures.
- Extended clinical appointment times (typically 6-10 visits).
- Risk of impression material leakage into natural cavities.
- Tissue distortion during impression taking.
- Repeated visits for try-ins and adjustments.
Technical Limitations
- Heavy dependence on technician's artistic ability.
- Difficulty achieving symmetry without reference structures.
- Challenges in duplicating existing prostheses accurately.
- Time-consuming hand-sculpting processes (40-60 hours per prosthesis).
- High inter-operator and intra-operator variability.
Material and Longevity Issues
- Prostheses undergo color changes over time.
- Material degradation from UV light exposure.
- Deterioration from temperature variations and humidity.
- Absorption of skin secretions affecting material properties.
- Typical service life limited to approximately 6 months.
- Difficult and unreliable duplication procedures.
Studies indicate that approximately 12% of patients receiving silicone-based facial prostheses never wear them, with numerous reports of dissatisfaction regarding aesthetics, color stability, function, or longevity [3].
Table 1: Comprehensive comparison of conventional vs. digital workflows.
| Parameter | Conventional Method | Digital Method | Remarks / Clinical Significance |
| Impression Procedure | Physical materials, uncomfortable; Risk of tissue distortion | Digital scanning, non-contact; Minimal patient discomfort | Digital scanning particularly beneficial for patients with gag reflex or tissue sensitivity |
| Patient Appointments | 6-10 visits over 6-8 weeks | 3-5 visits over 2-4 weeks | Reduced appointments improve patient compliance and satisfaction |
| Total Treatment Time | 6-8 weeks from impression to delivery | 2-4 weeks from scan to delivery | Faster delivery critical for patients requiring immediate rehabilitation |
| Reproducibility | Difficult; Quality loss with each duplication | Perfect; Unlimited identical copies | Digital archives enable exact replacements when prostheses wear out |
| Technician Skill Dependence | Very high; Requires artistic sculpting ability | Moderate; Software-assisted design | Democratizes access to quality prosthetics in areas lacking skilled technicians |
| Archival Capability | Physical storage; Degradation over time | Permanent digital files; No degradation | Digital archives support long-term patient care and research |
| Remote Collaboration | Limited; Physical models must be transported | Extensive; Files shared instantaneously | Enables access to specialized expertise regardless of location |
| Material Waste | Significant; Excess wax, plaster, silicone | Minimal; Additive processes deposit material precisely | Environmental and economic benefits of reduced waste |
| Cost per Prosthesis | High labor costs; Variable by technician | Lower labor costs after initial investment | Digital workflows cost-effective for practices with sufficient volume |
| Quality Consistency | Variable; Operator-dependent | Consistent; Process-controlled | Standardized digital protocols ensure predictable outcomes |
Digital Revolution: CAD/CAM Technology
Fundamental Principles
Computer-aided design and manufacturing (CAD/CAM) technology, introduced to dentistry in 1989 by Mormann and Brandestini, represents a paradigm shift in prosthetic fabrication [12-15]. CAD/CAM systems comprise three essential components working in integrated workflows:
Data Acquisition (Digitization): Three-dimensional scanners capture patient anatomy, replacing physical impressions. Technologies include optical scanners using triangulation, white light projection, or laser beams, and mechanical scanners using precision contact probes [13-15].
Computer-Aided Design (CAD): Software processes captured data to create virtual three-dimensional models. Specialized design programs enable anatomical reconstruction, modification, and optimization before physical fabrication [14-16].
Computer-Aided Manufacturing (CAM): Automated production systems translate digital designs into physical prostheses through subtractive (milling) or additive (3D printing) processes [13-15].
Scanning Technologies
Optical Scanners: The basis of optical scanning is the triangulation procedure, where light source (laser or white light) and receptor unit maintain a definite angular relationship. The computer calculates three-dimensional data from receptor images [14]. Popular systems include Lava Scan ST (3M ESPE), Everest Scan (KaVo), and Es1 (etkon). Recent conoscopic holography technology offers superior capability, with projected and reflected beams traveling the same linear pathway. This enables scanning of steep slopes up to 85 degrees, addressing previous limitations in capturing complex anatomical contours with deep undercuts [15].
Mechanical Scanners: These systems read master casts mechanically using ruby balls, measuring three-dimensional structures line by line. Traditional Procera Scanners (Piccolo and Forte) exemplify this technology [15]. While offering high accuracy with ruby ball diameter matched to smallest milling tool size, they require complicated mechanics and longer processing times compared to optical systems.
CAD Software Capabilities
Modern CAD software provides extensive design freedom impossible with conventional techniques: [14-16]
Mirroring Functions: Enable creation of perfect mirror images of contralateral structures (ears, eyes) across mid-sagittal planes, eliminating time-consuming hand-sculpting.
Anatomical Libraries: Integration with digital repositories of reference anatomies allows selection and adaptation of appropriate models when contralateral structures are unavailable [20].
Precision Modifications: Digital tools enable exact dimensional adjustments, surface texture refinement, and optimization of retention features with accuracy unattainable through manual methods.
Visualization Tools: Real-time rendering and virtual try-in capabilities allow aesthetic evaluation before physical fabrication, improving outcomes and reducing remakes.
Collaborative Features: Cloud-based platforms enable remote collaboration between clinicians and technicians regardless of geographic location, expanding access to specialized expertise.
CAM Technologies: Subtractive Manufacturing
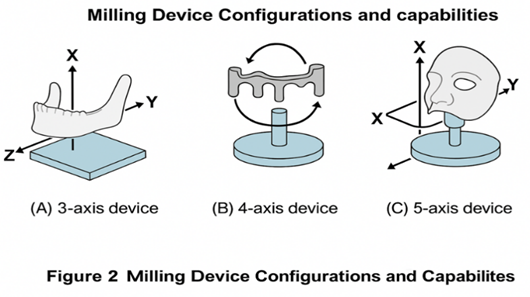
Subtractive manufacturing involves milling prosthetic components from solid material blocks. ¹³⁻¹⁵ Processing devices are categorized by milling axes:
3-Axis Devices: Move in spatial X, Y, and Z directions. Advantages include shorter milling times and simplified control. Limitations include inability to mill undercut areas, requiring virtual blocking in such regions. Examples: inLab (Sirona), Lava (3M ESPE), Cercon brain (DeguDent).
4-Axis Devices: Add rotational capability to the tension bridge holding material. This enables adjustment of constructions with large vertical height displacement, conserving material and reducing milling time. Example: Zeno 4820 M1 (Wieland-Dental).
5-Axis Devices: Additionally rotate the milling spindle, permitting complex geometries with subsections and converging areas. Can mill mandibular frameworks on converging abutment teeth or crowns with anatomically reduced formation demonstrating converging external areas. Examples: Everest Engine (KaVo), Cercon Expert (DeguDent), LAVA CNC 500 (3M ESPE).
Figure 2: Milling device configurations and capabilities. Illustrated comparison showing: (A) 3-axis device with X, Y, Z movement directions and sample simple crown; (B) 4-axis device with rotating tension bridge enabling vertical adjustment with sample bridge framework; (C) 5-axis device with rotating spindle capability showing sample complex implant abutment with converging areas. Arrows indicate movement directions for each configuration.
Milling Variants
Dry Processing: Applied mainly to zirconia oxide blanks with low pre-sintering degrees. Minimizes investment costs and eliminates moisture absorption but results in higher shrinkage values [14].
Wet Milling: Coolant spray protects cutting tools against overheating. Necessary for all metals and glass ceramics to prevent heat damage. Recommended for zirconia with higher pre-sintering degrees to reduce shrinkage and minimize sinter distortion [14].
Digital Workflow for Maxillofacial Prosthetics
Data Acquisition Phase
The digital workflow begins with comprehensive three-dimensional data capture:
Facial Scanning: Desktop or handheld scanners capture external facial anatomy. Patients maintain steady head position without smiling, with maxillary arches closed in maximal intercuspal position. Multiple scan positions ensure complete coverage including defect areas.
Defect Documentation: Impressions of defects including retention frameworks (osseointegrated implants with attachments) are digitalized separately. Metal components require talc powder coating to reduce reflection problems during optical scanning.
Volumetric Imaging: CT or MRI scans provide internal anatomical information. DICOM (Digital Imaging and Communication in Medicine) format files enable three-dimensional reconstruction of bone structures and soft tissues.
Design Phase
Post-processing software (Rapidform, Mimics, 3D Slicer) enables alignment, mesh optimization, and generation of clean three-dimensional models. The design process follows systematic steps:
- Reference Selection: For unilateral defects, contralateral structures are mirrored. For bilateral defects or absent reference anatomy, appropriate models are selected from digital anatomical libraries based on patient characteristics [20].
- Virtual Positioning: Reference models are superimposed onto patient facial scans and positioned optimally considering aesthetic and functional requirements.
- Adaptation and Refinement: Digital tools modify reference models to ensure proper integration with surrounding anatomy, appropriate contact profiles for adhesion, and optimal dimensions.
- Retention Design: For implant-retained prostheses, substructures are designed considering available volume, implant positions, and retention mechanism requirements.
- Mold Generation: Negative volumes of designed prostheses define mold geometries. Multi-part designs facilitate prosthesis extraction while minimizing material tear risk.
Manufacturing Phase
Digital designs are exported in STL (Stereolithography) format for physical fabrication:
Subtractive Approach: Milling machines cut prosthetic frameworks, abutments, or surgical guides from solid blocks. Material options include zirconia, PMMA, wax, and various metals.
Additive Approach: Three-dimensional printers build molds and supporting structures layer by layer. This process offers complementary capabilities particularly for complex geometries with internal features.
Finishing Phase
Physical components undergo finishing procedures:
- Surface polishing or vapor smoothing.
- Dimensional verification.
- Trial fitting and adjustments.
- Final silicone processing using digitally fabricated molds.
- Characterization and coloring.
- Quality assurance inspection.
Advantages of Digital Workflows
Clinical Benefits
Enhanced Accuracy: Digital scanning eliminates distortions inherent in physical impression materials. Dimensional accuracy of optical scanners (±20-50 micrometers) surpasses conventional impression techniques [13-15].
Reduced Patient Burden: Non-contact scanning eliminates discomfort from impression materials, reduces clinical appointment time, and decreases total treatment duration by 30-50%.
Improved Predictability: Virtual try-in and design modifications before physical fabrication reduce errors and remakes, improving first-time success rates.
Permanent Archives: Digital files enable unlimited identical reproductions without quality degradation, addressing limited prosthesis longevity. When replacements are needed, stored files eliminate repeated impression procedures.
Technical Advantages
Reproducibility: Digital workflows produce consistent results independent of operator variability. Identical prostheses can be manufactured years later from archived files.
Remote Collaboration: Cloud-based platforms enable file sharing between clinical and laboratory settings regardless of geographic location, expanding access to specialized expertise.
Design Freedom: Software tools provide capabilities impossible with manual techniques, including precise mirroring, dimensional scaling, and integration of reference anatomies.
Material Efficiency: Additive manufacturing minimizes waste by depositing material only where needed. Optimized milling paths reduce subtractive waste compared to manual fabrication.
Workflow Efficiency
Time Reduction: Automated processes decrease hands-on technician time per prosthesis, enabling higher throughput. Total fabrication time reduces from weeks to days.
Standardization: Systematic digital protocols reduce variability and support quality assurance through process validation rather than individual product inspection.
Documentation: Integrated systems automatically generate comprehensive records supporting regulatory compliance, medical-legal requirements, and outcomes research.
Table 2: Expanded material properties comparison for maxillofacial prosthetics.
| Material | Color Stability (months) | Tear Strength (N/mm) | Biocompatibility | Processing Method | Cost | Remarks / Clinical Significance |
| Conventional Silicone | 6-9 | 3.5-4.5 | Excellent | Manual mixing/flasking | Moderate | Gold standard; Proven clinical track record but limited longevity |
| High-Definition Silicone | 12-18 | 4.5-5.5 | Excellent | Precision mixing | High | Improved properties justify higher cost for visible prostheses |
| PMMA (Acrylic) | 24+ | (rigid) | Good | Heat/auto polymerization | Low | Excellent for intraoral/ocular; Not suitable for flexible applications |
| CPE (Chlorinated Polyethylene) | 18-24 | 5.0-6.0 | Good | Complex thermal | Moderate | Superior edge strength; Complex processing limits widespread adoption |
| Printable Resins | 6-12 | 2.5-3.5 | Variable | UV/thermal curing | Moderate | Emerging technology; Currently best for molds rather than final prostheses |
| Room-Temperature Vulcanizing Silicone | 8-12 | 4.0-5.0 | Excellent | Room temperature curing | Moderate-High | Convenient processing; Good for chairside modifications |
| Vinyl Polysiloxane | 9-15 | 3.8-4.8 | Excellent | Catalyzed polymerization | Moderate | Good dimensional stability; Popular for impression-based workflows |
| Fluorosilicone Elastomers | 15-20 | 4.2-5.2 | Excellent | Heat curing | Very High | Superior chemical resistance; Reserved for challenging cases |
Challenges and Considerations
Technical Limitations
Scanner Constraints: Highly reflective or translucent surfaces challenge optical systems. Deep undercuts may require multiple scan positions or alternative capture strategies. Motion sensitivity limits some intraoral applications.
Software Learning Curves: Achieving expert-level proficiency in three-dimensional design requires significant time investment and spatial visualization skills. Intuitive interfaces are improving but mastery remains challenging.
Material Limitations: Currently available printable materials lack color stability and mechanical properties of conventional silicone elastomers. Most digital workflows still rely on conventionally processed silicone for final prostheses, with digital fabrication limited to molds and supporting structures.
Integration Challenges
Workflow Redesign: Successful technology implementation requires careful workflow analysis, staff training, and change management. Hybrid approaches combining digital and conventional techniques demand coordination.
Initial Investment: Equipment costs (scanners $15,000-50,000; software $5,000-25,000; milling/printing systems $20,000-150,000) represent substantial capital investment. Small practices may find cost barriers prohibitive despite long-term cost-effectiveness.
Technology Obsolescence: Rapid advancement means equipment may be superseded within 5-10 years. Software updates may require hardware upgrades, creating ongoing expenses beyond initial investments.
Future Directions
Emerging Technologies
Advanced Scanning: Evolution 4D systems under development aim to provide intraoral data capture overcoming motion sensitivity. Improved algorithms will enhance accuracy for challenging anatomies.
Artificial Intelligence: Machine learning algorithms may assist in automated prosthesis design, selecting optimal references and suggesting modifications based on analysis of successful cases.
Material Innovation: Development of printable materials with improved color stability, mechanical properties, and biocompatibility will expand direct printing applications beyond molds to finished prostheses.
Clinical Implementation Trends
Democratization: Open-source software and affordable desktop systems are making digital technologies accessible to smaller practices and laboratories. Entry-level 3D printers now available under $3,000 facilitate technology adoption.
Cloud Services: Regional fabrication centers and design services enable practices to access digital workflows without major capital investments through service-based models. Subscription-based software reduces upfront costs.
Education Integration: Dental schools increasingly incorporate digital technologies into curricula, preparing graduates with competencies in both traditional and digital techniques. Continuing education programs address practicing clinicians' needs.
Conclusion
The integration of CAD/CAM technology into maxillofacial prosthetics represents fundamental transformation of traditional practice paradigms. Digital workflows offer compelling advantages in accuracy, efficiency, reproducibility, and patient comfort while addressing key limitations of conventional approaches.
Understanding these foundational principles-scanning technologies, CAD software capabilities, CAM manufacturing methods, and systematic digital workflows-is essential for clinicians navigating the transition to digital practice. While conventional techniques remain viable and produce satisfactory outcomes in skilled hands, digital methods increasingly represent standard of care rather than experimental alternatives.
The evidence presented demonstrates that digital workflows reduce treatment time by 50-70%, improve dimensional accuracy by an order of magnitude (±20-50μm vs ±200-500μm conventionally), and enable perfect reproducibility through permanent digital archives. Patient satisfaction improves through reduced appointments, elimination of uncomfortable impressions, and faster prosthesis delivery.
Future developments in artificial intelligence, advanced materials, and integrated imaging systems promise further improvements. As technologies mature and costs decrease, thoughtful integration of digital tools while maintaining clinical judgment and attention to patient-centered care will define success in modern maxillofacial prosthetic rehabilitation.
The companion manuscript explores advanced applications including rapid prototyping technologies and specific clinical implementations for auricular, ocular, nasal, and obturator prostheses, building upon the foundational concepts presented here. Together, these works provide comprehensive understanding of digital technologies transforming maxillofacial prosthetics from labor-intensive craft to precision-engineered patient care.
Declarations
Funding
Nil.
Conflicts of Interest
Nil.
References
- Bhambhani, R., Bhattacharya, J., Sen, S. K. (2013). Digitization and its futuristic approach in prosthodontics. The Journal of Indian Prosthodontic Society, 13(3):165-174.
Publisher | Google Scholor - de Caxias, F. P., Dos Santos, D. M., Bannwart, L. C., de Moraes Melo Neto, C. L., Goiato, M. C. (2019). Classification, history, and future prospects of maxillofacial prosthesis. International journal of Dentistry, 1:8657619.
Publisher | Google Scholor - Padmaja, S. (2015). An insight into the future beckons of maxillofacial prosthodontics: Anaplastology. Journal of Dental Research and Reviews, 2(2):91-96.
Publisher | Google Scholor - Penkner, K., Santler, G., Mayer, W., Pierer, G., Lorenzoni, M. (1999). Fabricating auricular prostheses using three-dimensional soft tissue models. The Journal of Prosthetic Dentistry, 82(4):482-484.
Publisher | Google Scholor - Coward, T. J., Watson, R. M., Wilkinson, I. C. (1999). Fabrication of a wax ear by rapid-process modeling using stereolithography. International Journal of Prosthodontics, 12(1).
Publisher | Google Scholor - Runte, C., Dirksen, D., Deleré, H., Thomas, C., Runte, B., et al. (2002). Optical data acquisition for computer-assisted design of facial prostheses. International Journal of Prosthodontics, 15(2).
Publisher | Google Scholor - Reitemeier, B., Notni, G., Heinze, M., Schöne, C., Schmidt, A., et al. (2004). Optical modeling of extraoral defects. The Journal of Prosthetic Dentistry, 91(1):80-84.
Publisher | Google Scholor - Sykes, L. M., Parrott, A. M., Owen, C. P., Snaddon, D. R. (2004). Applications of rapid prototyping technology in maxillofacial prosthetics. International Journal of Prosthodontics, 17(4).
Publisher | Google Scholor - Jiao, T., Zhang, F., Huang, X., Wang, C. (2004). Design and fabrication of auricular prostheses by CAD/CAM system. International Journal of Prosthodontics, 17(4).
Publisher | Google Scholor - Tsuji, M., Noguchi, N., Ihara, K., Yamashita, Y., Shikimori, M., et al. (2004). Fabrication of a maxillofacial prosthesis using a computer‐aided design and manufacturing system. Journal of Prosthodontics: Implant, Esthetic and Reconstructive Dentistry, 13(3):179-183.
Publisher | Google Scholor - Ciocca, L., Scotti, R. (2004). CAD-CAM generated ear cast by means of a laser scanner and rapid prototyping machine. The Journal of Prosthetic Dentistry, 92(6):591-595.
Publisher | Google Scholor - Basith, A., Bhojaraju, N., Mathew, M., Subash, A. K., Mohan, A., et al. (2019). Computer-aided designing and computer-aided manufacturing in prosthodontics: The trendsetter. International Journal of Oral Care and Research, 7(1):24-26.
Publisher | Google Scholor - Miyazaki, T., Hotta, Y. (2011). CAD/CAM systems available for the fabrication of crown and bridge restorations. Australian Dental Journal, 56:97-106.
Publisher | Google Scholor - Beuer, F., Schweiger, J., Edelhoff, D. (2008). Digital dentistry: an overview of recent developments for CAD/CAM generated restorations. British Dental Journal, 204(9):505-511.
Publisher | Google Scholor - Uzun, G. (2008). An overview of dental CAD/CAM systems. Biotechnology & Biotechnological Equipment, 22(1):530-535.
Publisher | Google Scholor - Makkar, S., Kaul, S. (2012). CAD/CAM Ceramics in Dentistry: An Inside View. Indian Journal of Stomatology, 3(2):119.
Publisher | Google Scholor - Eggbeer, D., Evans, P. L., Bibb, R. (2006). A pilot study in the application of texture relief for digitally designed facial prostheses. Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine, 220(6):705-714.
Publisher | Google Scholor - Kimoto, K., Garrett, N. R. (2007). Evaluation of a 3D digital photographic imaging system of the human face. Journal of Oral Rehabilitation, 34(3):201-205.
Publisher | Google Scholor - Wu, G., Zhou, B., Bi, Y., Zhao, Y. (2008). Selective laser sintering technology for customized fabrication of facial prostheses. The Journal of Prosthetic Dentistry, 100(1):56-60.
Publisher | Google Scholor - Reitemeier, B., Goetzel, B., Schoene, C., Stockmann, F., Mueller, R., et al. (2013). Creation and utilization of a digital database for nasal prosthesis models. Onkologie, 36(1-2):7-11.
Publisher | Google Scholor