Review Article
Combined Use of Cell Death Mechanisms as Plausible Therapeutic Targets with Canonical Therapies for Breast Cancer: Potentially Decreasing Dose, Toxicity, Expenditure and Improved Survival-A Narrative Review
1Scientific Director, Human Reproduction Scientific Director & Owner, G.T.B. Nagar, Jalandhar, Punjab, India.
2Scientific Director, Ex-Rotunda, A Centre for Human Reproduction, Kalpak Garden, Perry Cross Road, Near Otter’s Club, Bandra, Mumbai, India.
3Consultant Neurologist, Swami Satyanand Hospital, Near Nawi Kachehri, Baradri, Ladowali road, Jalandhar Punjab, India.
*Corresponding Author: Kulvinder Kochar Kaur, Scientific Director, Human Reproduction Scientific Director & Owner, G.T.B. Nagar, Jalandhar, Punjab, India.
Citation: Kaur KK, Allahbadia GN, Singh M. (2025). Combined Use of Cell Death Mechanisms as Plausible Therapeutic Targets with Canonical Therapies for Breast Cancer: Potentially Decreasing Dose, Toxicity, Expenditure and Improved Survival - A Narrative Review, Journal of Women Health Care and Gynaecology, BioRes Scientia Publishers. 5(6):1-26. DOI: 10.59657/2993-0871.brs.25.097
Copyright: © 2025 Kulvinder Kochar Kaur, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: April 08, 2025 | Accepted: September 15, 2025 | Published: September 23, 2025
Abstract
Breast Cancer (BC) has assumed the commonest mortality causing cancer in women globally which possesses plethora of subkinds, have bad prognosis as well as escalating mortality. In view of the molecular heterogeneity of BC restriction of effectiveness of canonical therapies exist in addition to pose hurdles basically in view of unanticipated clinical reactions, whose plausible exposition is the crosstalk along with changes of variable cell demise pathways. Nevertheless, maximum studies have concentrated just on one cell demise pathway, whereas rest of cell demise pathways are usually ignored, thus therapeutic chances correlated with cell demise pathways get missed. Thereby, taking into account isolation of pronounced kinds of cell demise, transitioning to variable cell demise designs at the time of mechanistic modes in addition to the controlling mechanistic modes behind BC. In this narrative review we have emphasized on the properties of diverse kinds cell demise inclusive of PANoptosis (apoptosis, pyroptosis, necroptosis) autophagy, ferroptosis along with cuproptosis, as well as detail their triggering factors signaling stepwise patterns in BC which might yield a baseline in reference to further scientific research with regards to pathogenesis of BC that might of contribute in generation of innovative approaches that might aid in decreasing dosages, thereby diminishing toxicity of chemotherapeutic agents, radiotherapy, thus expenditure & improved 5-year survival with overcoming resistance to agents like tamoxifen, apart from reverting resistance escalate chemosensitivity so much so that even metastasis might be conquered as well as triple negative breast cancer (TNBC) which posed substantial resistance to agents might get treated.
Keywords: breast cancer; apoptosis; pyroptosis; necroptosis; ferroptosis; cuproptosis
Introduction
Breast cancer (BC) got defined in the form of a Global Common Cancer by the International Agency for Research on cancer (IARC) in 2020. About 31% of new patients as well as 15% of demise is attributed to BC every year which makes it the second commonest etiology for demise in females [1]. As per a 2023 study it was illustrated that mortality occurred in 2.44 million BC cases in 81 countries, or areas, that portrayed a decrease of 3.2-11.6%in the last 2 decades in view of diagnosis at an earlier stage [2].
Classification of BC is made in the form of HER2 - rich kind (human epidermal growth factor receptor [EGFR] /HER2) (hormone receptor (HR), estrogen receptor (ER), progesterone receptor (PR- HER2+) basal-like kind (triple negative breast cancer, NBC (ER/PR/HER2-) normal mammary kind [3], in addition to luminal kind inclusive of luminal A (HR+ HER2-) along with luminal B (triple positive) [4]. Utilization of endocrine therapy as well as chemotherapy is frequently done for the treatment of BC [3]. Recently utilization of HER2 was done in the form of a target for the treatment of BC, however it was not efficacious in TNBC in view of absence of the target receptors.
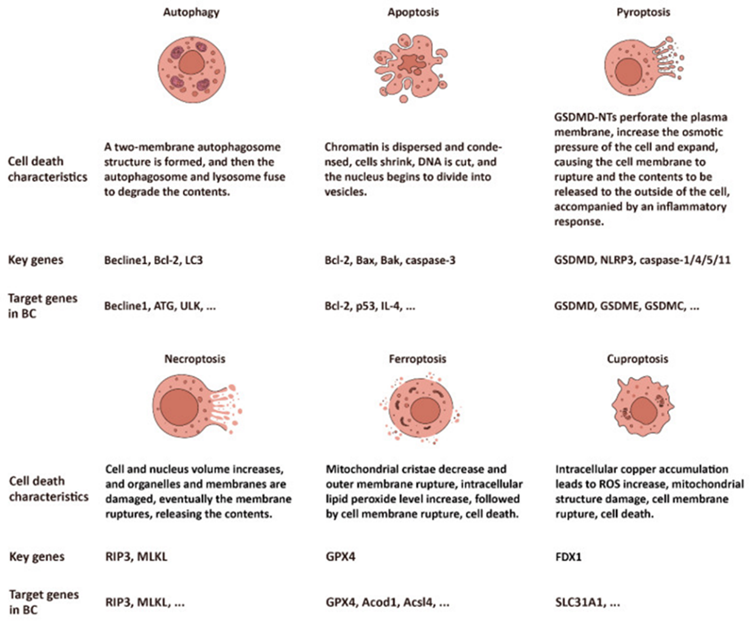
Recently studies have concentrated on cell demise in case of biological in addition to cancer therapy. Getting insight regarding the signaling pathways along with controlling mechanistic modes in case of cell demise might yield plausible therapeutic targets for BC as well as other kinds of cancer [5]. PANoptosis is the innovative term coined for the complex cell demise dependent in the interactions amongst pyroptosis, apoptosis in addition to necroptosis [6-8], based on or autonomous of autophagy that is implicated in the BC therapy or resistance to therapeutic agents. Ferroptosis along with cuproptosis portray innovative cell demise designs, stimulated as well as modulated by metal ions Fe in addition to Cu, respectively. Ferroptosis gets guided by lipid peroxide [9], whereas etiology of cuproptosis implicates accrual of copper ions resulting in cell poisoning [10]. Figure 1 depicts profiles of every kind of cell demise [11].
Figure 1: Courtesy reference no-11-The profiles of six subtypes of cell death: autophagy, apoptosis, pyroptosis, necroptosis, ferroptosis, and cuproptosis.
Autophagy
Autophagy: A programmed cell death (PCD) modulated by lysosomes
The idea of autophagy was posited initially by Christian de Duve subsequent to his invention that intraperitoneal injection of glucagon in rats stimulated generation of autophagic vacuoles along with influenced the lysosomes [12]. To start with scientific researchers found the breakdown of the cell structure as well as lysosomes appearance in the rat liver cells followed by finding autophagy in Saccharomyces cerevisiae [13]. Subsequent to that autophagy correlated genes were isolated [14]. Autophagy portrays a substantially preserved cellular physiological in addition to pathological events broadly observed in eukaryotic along with is a strictly controlled PCD. It possesses significant part in the sustenance of cellular homeostasis as well as elimination of injured organelles, bacteria, viruses in addition to other exogenous agents.
Three kinds of autophagy have been detected towards the lysosome namely i) macroautophagy ii) microautophagy besides iii) chaperone modulated autophagy (CMA). i) Macroautophagy is a frequently used term for autophagy. It represents the maximum functional autophagy along with possesses properties of kinds of autophagy responsible for the generation of autophagosomes, which portray double membrane structures whose fusion takes place with lysosomes as well as breakdown non wanted constituents. ii) CMA portrays an event where transportation of the substrate proteins possessing Lys-Phe-Glu-Arg-Gln (KFERQ) motif subsequent to acknowledgement takes place to the lysosomes for breakdown. Subsequent to binding to the chaperone heat shock cognate protein 70 (HSC 70), transportation of the substrate protein takes place to the lysosomes where after recognition their internalization takes place by lysosomal associated membrane protein 2A (LAMP2A) for breakdown by lysosomes [15]. iii) Microautophagy portrays an event of lysosomal/endosomal membrane self-engulfing cytoplasmic proteins in addition to their breakdown by lysosomal proteases [16]. Microautophagy is more particular in contrast to macroautophagy. Nanoparticular autophagy portrays an event where autophagosomes trap cytoplasmic constituents in a random fashion along with degrade them into cellular debris with regards to provision of energy in case of situations of starvation, whereas particular autophagy portrays an event where autophagosomes selectively result in phagocytosis of substance for instance injured or senescent organelles / other macromolecular substances [17]. Based on the selectivity of particular autophagy for substrates, autophagy further gets classified in the form of mitochondrial autophagy ii) peroxisomal autophagy iii) chloroplast autophagy or alternate labelling is for mitochondria, peroxisomes, lipids, as well as RNA the labelling is mitophagy, peroxophagy, lipophagy as well as ribophagy, ER-phagy etc., respectively.
Autophagy event comprises of 5 stages i) initiation ii) nucleation iii) extension of vesicles membrane iv) fusion of vesicles in addition to v) breakdown of intravesiculal contents i) at the time of initiation phase once hampering of mammalian target of rapamycin complex(mTORC1) takes place in case of starvation, there is existence of deficiency of cellular energy situations, FIP 200 along with autophagy correlated gene 13 (ATG13) undergo phosphorylation by the phosphorylated UNC51 kinase (ULK1), thus causing activation of ULK complex. AMP-activated protein kinase (AMPK), activation might further result in ULK complex to assume the excitation form. ULK complex is comprised of FIP 200, ATG13 in addition to ULK1/2 [18].
ii) at the time of nucleation stage binding of ULK complex which has been already activated, takes place with phosphoinositol 3-kinase (PI3K) complex for generating: phosphatidylinositol 3,4,5-trisphosphate (PIP3) which possesses the capacity of enrollment of greater autophagy correlated proteins subsequent to placement in autophagosomes precursor, facilitating formation of autophagosomes.
In addition to iii) extension of vesicle membranes. The PI3K complex comprises of beclin1, VPS34 along with ATG14. At the time of stage of extension of vesicle membranes, combination of ATG5, ATG12, as well as ATG16 produce E3 ubiquitin like ligase. The microtubule correlated protein 1A/1B light chain 3B (LC3) portrays the homolog of the yeast autophagy gene ATG8 in case of humans along with is imperative for extension of membrane [19]. Transformation of LC3 takes place into soluble LC3-I with the existence of ATG 4. Subsequently conversion of LC3-I takes place into lipoylated LC3-II with the existence of E1, E2 as well as E3. Binding of LC3-II takes place with phosphatidyl ethanolamine (PE) in addition to its insertion into membrane of autophagosomes where it aids in membrane extension [20]. At the time of membrane extension for generation of autophagosomes, particular receptor proteins subsequent to recognition along with binding molecules with particular breakdown signals for generating complexes which get encapsulated into autophagosomes for their breakdown by lysosomes [21].
Decontrolling of Autophagy - correlated gene along with treatment of BC
Autophagy possesses a double part regarding tumor propagation as well as treatment. On one side, it facilitates tumor survival by provision of nutrients in addition to energy for the pacey proliferation of cancer cells along with breakdown of organelles as well as macromolecules. In the meantime, escalation of autophagic actions takes place subsequent to tumor radiotherapy helping in the depletion of injured organelles along with macromolecules which take place as a result of therapy [22]. Conversely, autophagy hampers tumor survival. At the time of replicative crisis, autophagy aids in clearance of cancerous as well as compound which are mutated amongst cells which aids in avoidance of normal cells getting transformed into cancer cells. By definition replication crisis portrays cell demise which takes place as a result of shortening of telomeres that works in the form of last hurdle for deleting precancerous cells getting converted into cancer cells [23]. Occasional, precancerous cells might evade replication crisis; thereby it might work in the form of an efficacious tumor suppressor. Autophagy possesses a key part at the time of replication crisis by activation of demise of precancerous cells. In case of hampering of autophagy, a greater probability is present precancerous cells would evade replication crisis in addition to this might facilitate proliferation of cells along with accrual of genomic instability in precancerous cells. In the case of correlated cancer, generally there is association with elimination of autophagy working [24].
At present for the treatment of BC chemotherapy, endocrine therapy, targeted therapy, radiotherapy as well as immunotherapy are utilized [25]. Autophagy possesses an important part in the generation of melanoma, correlated cancer, pancreatic cancer in addition to neurodegenerative diseases [26]. Changes in autophagy influence the production along with therapy of BC in plethora of manners.
Beclin-1 possesses a substantially significant part in autophagy, therefore beclin 1 might get utilized in the form of a therapeutic target in BC. Beclin-1 possesses a double part in the production of BC by i) hampering tumor growth as well as ii) facilitating tumor survival. In the case of maximum BC carcinogenesis, there is diminished expression of beclin 1 [27]. Additionally, heterozygous mice with beclin 1 deficiency illustrated greater risk of generating breast, ovarian in addition to prostate cancer [28]. On the other hand, mice with overexpressed beclin 1 had a diminished risk of BC, specifically with overexpression of HER2 BC, in view of escalated autophagy [29]. Nevertheless, subsequent to beclin 1 knockout (KO) enhancement, of proliferation of BC takes place that facilitates the propagation of BC [29], despite certain studies have illustrated that the expression of beclin 1 escalates autophagy actions that in turn hampers BC growth. Nevertheless, certain workers have illustrated that a greater expression quantity of beclin 1 in vitro escalates the quantities of cellular autophagy, leading to improvement of survival of BC in the situations of starvation along with hypoxia [28]. Specifically in case of HER2 positive BC as well as TNBC, overexpression of beclin 1 correlated with greater antagonistic characteristics [30].
MicroRNAs (miRNAs) represent endogenous small non coding RNAs (snc RNAs), single stranded molecules constituted of about 19-23 nucleotides (mean 22n) which possess the capacity of controlling the expression of plethora of autophagy genes. Decontrolled miRNA expression has been observed in BC [31], pointing that miRNA might yield innovative strategies for the therapy of BC. Esparza-Garridoet al. [32], isolated 18 miRNAs which possess the capability of hampering autophagic actions via downregulation of the expression of ULK1 as well as hampering the phosphorylation of beclin 1 in the BC cell lines MDA-MB-231, subsequent to B-cell lymphoma 2 (BCL2)-interacting killer (apoptosis inducing) (BIK) interference. Such 18 miRNAs further possessed the capability of hampering autophagy by hampering the mitogen activated protein kinase (MAPK)/ janus kinase (JAK) for the dephosphorylation of Bcl2, who’s binding therefore takes place with beclin 1 with greater ease [32]. miR-489 is capable of decreasing the expression of ULK1 in different BC cell lines, therefore diminishing autophagy actions in addition to hampering tumor growth. Additionally, miR-489 further possesses the capability of hampering autophagy by escalating the sensitivity of BC to adriamycin, pointing to diminish their resistance to adriamycin, therefore escalating the efficacy of BC therapy [33]. miR-27a is capable of hampering autophagy blockade of substrate administration to autophagosomes, therefore diminishing the quantities of LC3-II along with SQSTM1. Furthermore, it is capable of escalating the resistance of BC cells in vitro [34]. miR-20a possesses the capacity of negatively controlling the quantities of beclin 1, ATG 16L1 along with SQSTM1, as well as it further facilitates the generation of BC by diminishing the autophagy actions [35].
Long non coding RNAs (lnc RNAs) represent non coding RNA molecule kinds with the absence of the open reading frames (ORFs) as well as possess greater than 200 nucleotides as per length, having expression in BC. Lnc RNAs possess the capacity of directly controlling autophagy in addition to their overexpression in BC influences the cancer metastasis along with resistance to agents [36]. Concomitantly lnc RNAs might work in the form of therapeutic targets for affecting the propagation of BC. The initial lnc RNA which got isolated In reference to BC propagation was HOX antisense inter RNA(HOTAIR) [37], that significantly resulted in upregulation of target RNA molecules that get expressed in BC [38]. Li et al. [39], observed that knockdown of lnc RNA HOTAIR hampered PI3K/ AKT/mTOR signaling pathway resulting in diminishing their BC resistance to Adriamycin [39]. One further lnc RNA correlated with BC is NAMPT-AS which has its placement in the nicotinamide phosphoribosyl transferase (NAMPT) promoter area as well as its overexpression take place in TNBC. Overexpression of NAMPT-AS results in escalated viability, capability of proliferation, along with oncogenic characteristics of TNBC cells, in addition to are correlated with bad prognosis for the patients. NAMPT-AS controls propagation of TNBC by manipulating the transcriptional actions of NAMPT. Subsequent to downregulation of NAMPT expression, escalated expression of ATG 5, ATG 7 along with beclin 1 takes place. This is correlated with escalated aberrant autophagy actions, therefore hampering cell survival as well as invasion magnitude [40]. Overexpression of one further lnc RNA, namely differentiation antagonizing non protein coding RNA(DANCR), takes place in BC, in addition to knockdown results in diminished cell proliferation. LC3B along with ATG 5, escalated autophagy, that hampers malignant propagation of BC [41].
PANoptosis: A Cross Controlled Mechanistic Mode of Cell Demise
PANoptosis is the innovative term coined in 2019 for the complex cell demise dependent on the interactions amongst pyroptosis, apoptosis in addition to necroptosis for generating a complementary circuit that reimburses for evading cell demise [42]. Prior to invention of PANoptosis scientists, did not possess the capacity of fathoming the manner such cell events might be associated by a complex which works in the form of augmenting cell demise mechanistic modes. Such invention pointed that PANoptosis possesses the capability of controlling the full cell demise events, enabling it to be a robust manner of hampering cancer. Nevertheless, it is significant to appreciate that whereas PANoptosis has the capacity of hampering cancer, mutations in genes result in propagation of tumor. Considerable shared mechanistic modes take place amongst pyroptosis, apoptosis in addition to necroptosis. For instance, death receptors (DRs), as well as death ligands (DLs), are capable of causing activation of apoptosis along with necroptosis. Subsequent to the binding of tumor necrosis factor receptor type 1(TNFR1) with its ligand tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), enrolment of complex that comprises of tumor necrosis factor- associated death domain (TRADD), receptor interacting protein 1(RIP1), in addition to E3 ubiquitin ligase. Following that deubiquitination of RIP1 takes place, stimulating the generation of complex IIa/IIb. The complex IIa is capable of activating caspase-8 along with stimulating extrinsic apoptosis. Subsequent to hampering caspase-8 the generation of complex IIb followed by activation of necroptosis takes place [11]. Additionally, the caspase- family might be observed in apoptosis in addition to pyroptosis. The caspase-1/4/5/11have the capacity of performing cleavage of GSDME along with stimulating pyroptosis [43,44], whereas caspase-3/8 have the capacity of performing cleavage of caspase-3 as well as stimulating apoptosis [45,46]. In the meantime, caspase-3 further possesses the capability of performing cleavage of GSDME along with stimulating pyroptosis, that is based on the GSDME activation [47]. The reactive oxygen species (ROS) further are capable of influencing pyroptosis, apoptosis in addition to necroptosis. The ROS further facilitates nuclear placement as well as transcriptional activation of nuclear factor κB(NFκB) [48]. NFκB possesses the capability of facilitating the expression of B cell lymphoma extra-large (Bcl-xL), that is capable of hampering apoptosis [49]. The ROS further have the capacity of facilitating the expression of nucleotide-binding domain, leucine-rich-repeat containing family, pyrin domain-containing (NLRP3) inflammasome, facilitate caspase-1 as well as stimulating pyroptosis [50]. ROS facilitates necrosome generation, along with necroptosis by influencing RIP1 phosphorylation [51].
Apoptosis
Apoptosis: A PCD Stimulated by Caspases (Alias the Cysteine Aspartyl Specific Protease)
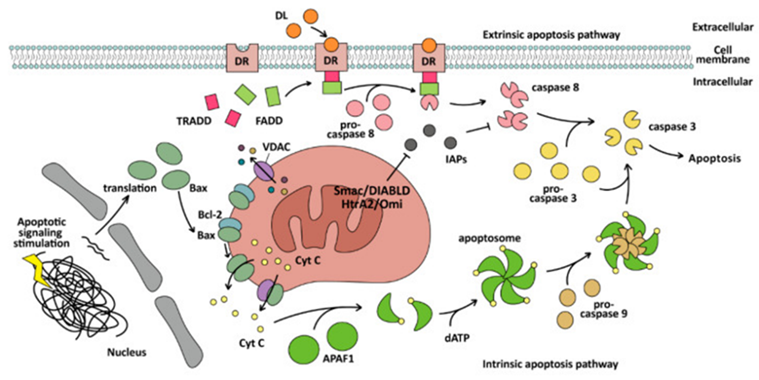
Apoptosis in the form of cell demise possesses akin history like that of autophagy. This word apoptosis got coined by Kerr et al. [52], in 1972 for detailing a morphologically distinctive kind of cell demise. Apoptosis possesses the properties of cell shrinking, morphology chromatin condensation, DNA fragmentation, nuclear fragmentation, as well as blebbing of cell membrane that is controlled by apoptosomes, that further undergo phagocytosis in addition to get degraded by macrophages or the adjacent cells [52,53]. Apoptosis basically gets modulated by caspase family proteins along with might be categorized into intrinsic as well as extrinsic pathway [54]. Implementation of apoptosis is basically brought about by caspase-3, which it accomplishes by resulting in cell demise through cleavage of α tubulin [55], myeloid leukemia 1(Mcl-1) [56], actin [55], fodrin [57], lamin [58] poly ADP ribose polymerase (PARP) [59], inhibitor of caspase activated deoxy ribonuclase (ICAD) [60], in addition to other substrate proteins. The intrinsic pathway aliases the mitochondrial pathway, is based on caspase-9 as well as Bcl2 family proteins. Bcl2 family proteins might be categorized into 2 conflicting kinds- for instance i) anti apoptotic, that implicates the Bcl homology (BH) 1/2/3 domain inclusive of Bcl2, B cell lymphoma extra-large (Bcl-xL), Bcl-W, Mcl-1 whereas ii) the proteins belonging to the proapoptotic class are ones with just BH 3 domain for instance Bcl2 correlated X protein (Bax), Bcl2antagonistic killer1(Bak1), Bad, Bcl-xS, Bak, Bid, Bik, Bim along with Hrk [61,62]. Mitochondrial pathway controlling, is indivisible from the BH domain. For instance, Bax, Bcl2, with BH 1/2/3 possess the capability of generating a heterodimer with Bax with just BH 3, thereby hampering apoptosis [63]. In the absence of apoptotic stress, they produce Bcl2 in addition to Bcl-xL (pro-survival) a heterodimer with Bax/ Bak for the sustenance of outer mitochondrial membrane (OMM) intactness. On stimulation of cells by proapoptotic signals, Bax expression gets escalated, that aids in preventing binding with the Bcl2 family proteins. Its oligomerization takes place along with it getting placed on OMM in addition to generating channels [56], or its binding takes place with voltage dependent anion channel (VDAC) for generating larger channels on the OMM [64]. Subsequent to that liberation of cytochrome (CytC) might take place through the mitochondrial membrane gap into the cytoplasm [65]. Placement of Bak further is in the OMM. Subsequent to the apoptotic signals getting produced activation of Bak that was dormant earlier takes place by the BH3 domain of the BH3 interacting domain death agonist (BID), Subsequent to which activated Bak possesses the capability of enrolment of along with activating the dormant Bak for generating floating Bak for generating a cascade reaction .Oligomerization of such Bak takes place for generating holes in the OMM, resulting in mitochondrial outer membrane permeabilization (MOMP) [66]. On the liberation of CytC in the cytoplasm, it possesses the capability of binding apoptosis protease activating factor (Apaf-1)monomers for altering its configuration, leading to exposure of the WD domain, resulting in variable Apaf-1for assembly of apoptosomes by binding dATP [46], followed by exposure of the caspase activation and recruitment domain (CARD) of Apaf-1for enrolment of as well as activation of caspase-9, catalyzed the caspase-3 maturation in addition to stimulating apoptosis .Additionally, in the form activation of a transcription factor p53 is capable of stimulating mitochondrial pathway apoptosis by facilitating PUMA (a p53 upregulated modulator of apoptosis) expression, Particularly, of PUMAα along with PUMAβ that possess BH3 domain [67].
ii)The extrinsic pathway alias the DR modulated pathway is based on binding of natural killer cells (NK cells) to the DRs along with activating caspase-8 for stimulating apoptosis. Such DRs are inclusive of Fas receptors, death receptor4(DR4), DR5, TNFR1, as well as TRAIL receptor (TRAIL-R), all of which have existence on the cell surface [68]. Subsequent to the binding of extracellular DL(TNF) with DRs (TNF R1) on the cell membrane, the adaptor protein TRADD collects in addition to results in enrolment of Fas associated death domain (FADD), getting followed by enrolment of monomeric procaspase-8 for generating death induced signaling complex (DISC) via the DED domain of FADD. Procaspase-8 crosstalk’s with DISC along with facilitates dimerization of procaspase-8 as well as gets activated by caspase-8 [6,45]. Apoptosis activation hat takes place through caspase-8 gets impacted by 2 kinds of cells. In the ‘’type II’’ cells start of apoptosis has the requirement of activating mitochondria, whereas ‘’type I’’ cells start does not [45]. in case of “type I’’ cells caspase-8 has the capability of directly activating caspase-3, whereas in “type II’’ cells, mitochondria have the requirement of liberating second mitochondrial activator of caspases (Smac)/DIABLO [53], in addition to Omi/ HtrA2 [69], for hampering the inhibitor of apoptosis proteins (IAPs) or IAPs would hamper caspase-8 from activating other caspases along with avoidance of apoptosis. Intrinsic along with extrinsic pathway are detailed in Figure 2.
Figure 2: Courtesy reference no-11The mechanism of the intrinsic and extrinsic pathway of apoptosis. The intrinsic apoptosis pathway is mitochondrial-dependent and cleaves caspase-3 by caspase-9, while the extrinsic apoptosis pathway is mitochondrial-independent and cleaves caspase-3 by caspase-8, and exists in natural killer (NK) cells and CD8-positive cytotoxic T lymphocytes.
Apoptosis Associated Decontrolled Genes Along with Treatments in BC
Apoptosis possesses a significant part in the contingency as well as generation of BC. Variable studies have illustrated that apoptosis is implicated in variable events of breast generation in addition to phenomenon of BC in view of diminished apoptosis, resulting in disequilibrium amongst cell proliferation along with cell demise at the time of generation of BC. Aberrant expression of a plethora of apoptosis associated genes occur in the events of BC.
The expression of Bcl2 family proteins implicated in apoptosis continue to be aberrant invariably in BC. Bcl2 expression takes place in practically 80% of subjects of human BC [70], in addition its overexpression takes place in around 75% of subjects [71]. Thereby Bcl2 expression for hampering apoptosis might be utilized in the form of a parameter for anticipation of prognosis of BC. Alipour et al. [72], fashioned a nanohybrid carrier labelled as MiRGD, that possesses the capability of binding DNA as well as precisely administer it to BC cells in addition to facilitating apoptosis by silencing Bcl2 [72]. Additionally, it is feasible to form a BH3 domain-like structure for hampering Bcl2 via domain crosstalk. Bcl2 might be hampered by the BH3 mimetic ABT-737or 199(venetoclax) in combination with tamoxifen, that has the capability of hampering BC growth with greater efficacy along with decrease drug resistance [73]. Bcl2 further is capable of hampering BC growth by facilitating the expression of Bcl2 hampering proteins. For instance, the expression of Bim, Bax, or Bak might be facilitated by oleander in as well as quercetin, that might facilitate apoptosis of BC cells hampering the expression of Bcl2 for hampering BC growth [74].
Additionally, direct facilitation of caspases further is capable of facilitating apoptosis in BC cells. Considerable expression of caspase-3 might take place in addition to activated by facilitating MOMP followed by hampering anti apoptotic proteins along with activating intrinsic apoptosis, therefore stimulating apoptosis as well as hampering BC proliferation in addition to migration [75]. Procaspase-9/3/7 cleavage along with mitochondrial liberation of CytC possess the capacity of getting facilitated by diallyl trisulfide in BC cells resulting in PARP proteolysis followed by stimulating extrinsic along with intrinsic pathway [76].
Different signaling molecules which stimulate apoptosis further might help in BC therapy. In case of human BC cells apoptosis stimulated by interleukin (IL-4), might be associated with phosphorylation of insulin receptor substrate-1(IRS-1), as well as signal transcription and transducer 6 (STAT 6). STAT 6 is capable of dimerization as well as translocation to the nucleus subsequent to phosphorylation, that activates promoter of particular genes [77]. Moreover, IL-4 possesses the capability of facilitating apoptosis by hampering extracellular signal - regulated kinase (ERK1), therefore facilitating activation of caspase-8 [78]. Interferon-γ (IFN-γ) has the capacity of facilitating the expression of FasL in addition to regulating apoptosis of BC cells through the Fas: Fc fusion protein transmitting signals [79]. Additionally, regulating Omi/ HtrA2 liberation possesses the capability of diminishing TRAIL resistance along with facilitate BC cells apoptosis [80]. TRAIL as well as FasL signaling in BC might be disturbed by miR-519a-3p in addition to stimulating resistance for apoptosis, since miR-519a-3p has the capacity of diminishing the expression of TRAIL-R2 along with caspase-7/8 resulting in evasion of cancer cells demise [81]. Additionally, hampering cyclin D1 as well as mTOR, resulting in upregulation of expression of peroxisome proliferator activated receptorγ (PPARγ) [82], disrupting the PI3K/ AKT [83], NFκB [84], signaling pathway by cannabidiol, possesses the capability of stimulating apoptosis, facilitate cells differentiation in addition to continuation of cell proliferation in BC. Hampering topoisomerases for disrupting replication of DNA through the chemotherapeutic agents for instance quinacrine along with doxorubicin (DOX, or adriamycin) might result in cell cycle arrest, DNA injury as well as stimulating expression of p53, or disturb DNA injury healing pathways [85], therefore resulting in apoptosis of BC cells. The proapoptotic tumor suppressor protein Par4 has the capacity of facilitating tumor regression in addition to diminish BC recurrence, by expression of p53, induction of multinucleation, proliferation along with facilitating the BC cells apoptosis [86].
Pyroptosis
Pyroptosis: A PCD Stimulated by GSDMs
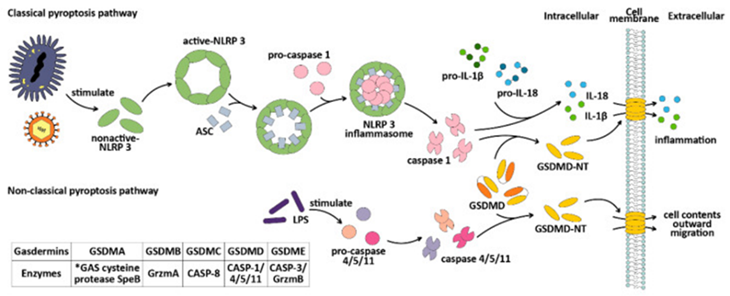
Gasdermin family proteins (GSDMs) are associated with non-syndromic hearing impairment [87], alopecia [88], childhood asthma [89], neurodegenerative diseases [90], breast cancer [91], melanoma along with gastric cancer [92]. Six kinds of GSDMs been have isolated inclusive of GSDMA, GSDMB, GSDMC, GSDMD, GSDME/ DFNA5 along with PJVK/ DFNB59 [93]. Such proteins that basically get expressed in gastrointestinal tract (GIT), as well as skin [93,94], were observed to possess innovative working for resulting in inflammatory cell demise through pyroptosis in 2015[95,96]. The initial detailing of pyroptosis was done in 2001[97], despite at the time specified, the precise mechanistic modes continued to be uncharted, along with the only insight present was that it portrayed an innovative kind of cell demise with its etiological factors were caspase proteins that possessed the capability of resulting in inflammation. Presently we have insight in reference to the mechanistic modes of pyroptosis. It portrays a kind of PCD modulated by GSDMs which liberate interleukins for stimulating inflammation. Pyroptosis that gets stimulated by GSDM might be categorized into Canonical Pathway and Noncanonical Pathway.
The Canonical Pathway: Gets modulated by caspase-1 in addition to is based on GSDMD [49]. Intracellular GSDMs comprised of a C terminal repressor domain (RD) in addition to N terminal pore forming domain (PFD) further labelled as CT along with NT [92,98]. Just the identified NT fragments are capable of binding with membrane lipids leading to perforation taking place. Generally intracellular GSDMs are present in a self-hampering state (other than PJVK) [95]. Nevertheless, in case of bacterial as well as viral invasion (alias pathogen - associated molecular patterns (PAMP) the intracellular pattern recognition (PRR), Nod- receptors (NLR) acknowledge PAMP in addition to aid in NLR3 in binding with the adaptor protein ASC (apoptosis associated speck protein that possesses a CARD. The CARD domain of ASC subsequently results in enrolment of procaspase-1 along with leads to assembly of NLRP3 inflammasome [99], therefore activate caspase-1 as well as results in cleavage of GSDMs for liberation of cytotoxic NT fragment [95], resulting in cell membrane perforations in addition to cell demise. Concomitantly, the cell cargo gets liberated from the cell, stimulating an inflammatory reaction. The activated caspase 1 further result in cleavage of pro IL-1β as well as pro IL-18 [100], into mature caspase 1, activating IL-1β in addition to IL-18 along with liberating them in extracellular area [99], resulting in the accumulation of inflammatory as well as escalating the inflammatory reaction.
The Noncanonical Pathway: Gets modulated by caspase-4/5/11 [43,44]. The basic variation amongst canonical pathway along with noncanonical pathway is non requirement of ASC adaptor protein in the noncanonical pathway [43,101], for aiding in enrolment of caspase-4/5/11 monomers. Furthermore, in case of noncanonical pathway caspase-4/5/11 does not have the capability of cutting pro IL-1β as well as pro IL-18, which restricts its capacity for augmenting inflammatory reaction to pyroptosis. Nevertheless, in case of nonimmune cells caspase-4 possess the capacity of cleavage of pro IL-18 subsequent to configurational alterations following cleavage [102]. Caspase-11 further is capable of facilitating canonical pyroptosis pathway in addition to IL-1β maturation along with liberation aiding in processing of caspase 1 [103]. The CARD domain of caspase-4/5/11[44], has the capacity of recognition as well as binding to the lipid A of the pathogen lipopolysaccharides (LPS) [43,44,103]. Subsequent to the stimulation of LPS, activation of caspase-4/5/11 takes place by oligomerization [44], as well as cleavage of GSDMs for liberation of cytotoxic NT fragments takes place [43], inclusive of triggering pyroptosis. The two canonical along with noncanonical pathway pyroptosis are detailed in Figure 3.
Figure 3: Courtesy reference no-11-The mechanisms of the classical and non-classical pyroptosis pathways. Gasdermins can be cleaved by different enzymes, among which GSDMA can be cleaved by the GAS cysteine protease SpeB [104], GSDMB can be cleaved by granzyme A [105], GSDMC can be cleaved by caspase-8, GSDMD can be cleaved by caspase-1/4/5/11, and GSDME can be cleaved by caspase-3 or granzyme B. *GAS: group A Streptococcus. Grzm: granzyme. CASP: caspase.
Apart from GSDMD, other proteins belonging to GSDM family pyroptosis resulting from GSDM, labelled in the form of apoptotic caspase pathway since caspase-3, that is implicated in cleavage of GSDME, gets further utilized for activating apoptosis. Nevertheless, in case of expression of GSDME, transformation of apoptosis takes place into pyroptosis in view of cleavage of GSDME by caspase-3, for liberation of NT fragments that possess pore generating actions [106,107]. GSDMB has the capacity of cleavage by granyzymeA for liberation of GSDMB- NT as well as results in pyroptosis [108]. Existence of a positive feedback pathway for pyroptosis is there, where the immune microenvironment, activated by pyroptosis, CD8+T cells in addition to NK cells further possesses the capability of facilitating pyroptosis cleavage of GSDME by granyzymeB [7]. Cleavage of GSDMC, might be accomplished by caspase-6, caspase-8 [109,110], TNFα, therefore leading to pyroptosis.
Pyroptosis Associated Decontrolled Genes along with Treatment in BC
Pyroptosis possesses double actions in cancer since it is associated with the liberation of inflammatory factors for instance IL-1β as well as IL-18 in addition to inflammatory factors might be associated with inimical sequelae on the tumor microenvironment (TME) at the time of BC generation. In contrast to normal cells, BC cells possess overexpressed IL-1β invariably [111]. IL-1β as well as IL-18 in the form of inflammatory factors are capable of facilitating the accrual of immune cells. IL-1β further is capable of facilitating growth of blood vessels in tumors [112], inclusive of in BC. NLRP3 inflammasome has been held responsible for infiltration by myeloid cells since it has the capacity of facilitating 1β maturation via activation of caspase 1 [113]. In addition to overexpression of IL-1β in BC cells, NLRP3 inflammasome, further has been observed to be overexpressed in BC cells [114]. IL-1β further has the capacity of facilitating BC cells migration in addition to invasion by activation of ERK1/2 [111], which further is capable of activating MAPK signaling pathway.
GSDME expression quantities are generally lesser in BC cells in contrast to normal cells [107]. GSDME expression get escalated via DNA demethylation, therefore stimulating pyroptosis, escalating the chemosensitivity along with reverting the resistance to chemotherapeutic agent paclitaxel [115]. Caspase-3activation further is capable of facilitating apoptotic caspase pathway of pyroptosis in BC cells, hampering tumor metastasis in an efficacious manner [116]. ROS possess a significant part in the BC cells that received DOX therapy, that has the capacity of stimulating caspase-3/ GSDME modulated pyroptosis via ROS / JNK pathway [117]. STAT3 phosphorylation possesses the capability of facilitating mitochondrial ROS generation, stimulating pyroptosis by facilitating caspase-3/ PARP/ GSDME axis as well as hampering tumor growth in addition to lung metastasis in case of TNBC [118].
GSDMB expression that possesses 6 basic protein isoforms [106], get upregulated in approximately 65% HER2 BC cells [119], particularly GSDMB-2. The substantially greater expression of GSDMB-2 has the capacity of stimulating tumor invasion along with aid in metastasis [91,120]. Scientific researchers, illustrated that just GSDMB-3/4 possesses a stable pore generation structure β9-β11hairpin, in addition to the cavity generated by α3-β6-β9 possesses the capability of stimulating pyroptosis, enabling it to be advantageous in BC treatment. Rest of GSDMB- isoforms have the capacity of hamperingGSDMB-3/4 modulated pyroptosis [108]. In case of mammary glands GSDMB-2 facilitates BC by aiding HER2 [121]. Molina Crespo, et al. [119], fashioned an agent labelled as AbGB-NC with the objective of achieving anti- GSDMB activity which is encapsulated in hyaluronic acid nano capsules (NC). AbGB-NCs were capable of repressing maximum cancer metastasis in a mouse HER2 BC model, diminishing the aggressiveness of HER2 BC [119].
GSDMC gets substantially expressed in BC [122], generating double actions akin to that of GSDMB. GSDMC possesses the capacity of pyroptosis induction in BC cells [109,110,122]. In case of BC cells, programmed death ligand1 (PDL1) is capable of converting tumor necrosis factor alpha (TNFα) stimulated apoptosis into pyroptosis by cross talking with p-Stat3 for escalating the transcription of GSDMC [110]. In the meantime, extensive quantities of GSDMC possess the capacity of escalating the sensitivity of cancer cells to PARP hampering agents. PARP hampering agents are capable of facilitating caspase-3/ GSDME modulated pyroptosis, whereas GSDMC have the capacity of escalating the cells sensitivity to chemotherapeutic agents via caspase-6 /8 [109]. In the TNBC approximately 35% of tissues obtained from cases of cancer illustrated extensive expression of GSDMC [109], pointing the plausibility of PARP hampering agents in TNBC therapy. Additionally, Sun et al. [122], illustrated that the LINC00511/ hsa miR -573/ GSDMC axis, possesses the capacity of escalating the GSDMC expression [122], however, such upregulation is correlated with a bad prognosis as well as tumor immune infiltration of BC. Nevertheless, further work is required for getting insight in this association in reference to bad prognosis etc.
Necroptosis
Necroptosis: A PCD Stimulated by RIPK’s along with MLKL
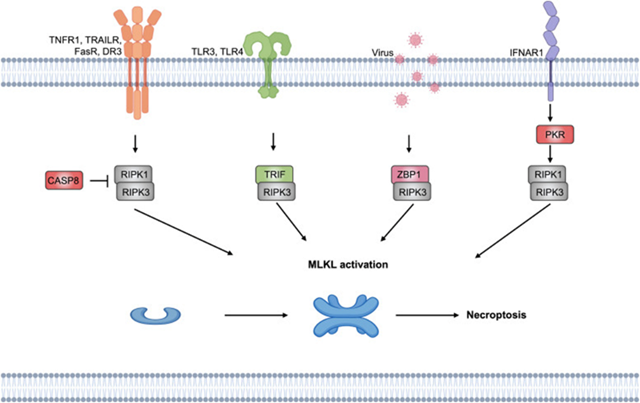
Necroptosis alias Programmed necrosis portrays a kind of cell demise which takes place once apoptosis is hampered. It does not have dependence on cysteine caspases, however rather implicates receptor interacting protein kinase (RIPK’s) for instance RIPK1, RIPK3 along with mixed lineage kinase domain (MLKL) [123]. Variable stimuli possess the capacity of stimulating necroptosis, maximum frequently it gets stimulated by TNFα [124]. Following stimulation by TNFα, phosphorylation of RIPK3 takes place by RIPK1, that results in generation of the necrosome a complex comprised of RIPK1/ RIPK3, with the activated RIPK3 in turn leads to phosphorylation in addition to activation of MLKL leading to escalated permeability of cellular membrane, resulting in rupturing of cell along with liberation of its constituents leading to necroptosis [8]. Necroptosis modes are detailed in Figure 4.
Figure 4: Courtesy reference no-197-Molecular mechanism of necroptosis. Z-DNA binding protein 1 (ZBP1) and cell surface death receptors, such as FasRs, TNFR, IFN receptors, and TLRs, initiate necroptosis, and RHIM-containing downstream proteins bind to RIPK3. The necrosome is then developed, which causes cell lysis.
Necroptosis along with treatments in BC
Necroptosis possesses a significant part in the generation of breast, therefore targeting necrotic associated proteins might aid in BC treatment. Expression of RIP3 is significantly lesser in BC tissues in contrast to normal breast tissues in maximum BC cases [109]. The expositions for this are the transcriptional methylation of RIP3, resulting in suppression of RIP3. Thereby downstream MLKL does not get activated subsequent to its phoshorylation along with necroptosis gets hampered. Furthermore, suppression of RIPK3 aids in generation of higher chemotherapeutic resistance as well as facilitates survival of BC cells. Koo et al. [125], pointed that BC treatment might be attained by addition of agents (5-azacitidine and decitabine) possessing the capacity of hypomethylation for the induction of RIP3 expression [125]. Jiao et al. [126], found a significantly escalated expression of MLKL in addition to determined phoshorylation of MLKL in the necrotic areas of BC breast solid tumors. On knock out (KO) of MLKL, lesser tumors illustrated cell demise along with occasional necrotic areas in the tumors. There was elimination of capacity of tumors to metastasize to the lungs in case of mice. This pointed that the generation of necrotic areas of tumors has a robust association with necroptosis [126]. Stoll et al. [127], observed MLKL along with RIP3 facilitated the expression of IFN-γ as well as IFN-α associated genes along with anti-cancer immunity in a mouse model of BC [127].
Variable agents for instance) cysteine elimination [128], quercetin [129], Smac mimetic LCL-161 [128], in addition to non benzaquinone analogs of geldanamycin DHQ3 [130], have been illustrated to facilitate BC cell demise by stimulating the initiation of necroptosis. Numerous scientists are evaluating greater variety of methodologies for stimulating induction of necroptosis for BC treatments for instance induction of expression of RIPK3 [125], along with metallic analogs for stimulating induction of necroptosis for getting over chemotherapeutic resistance in BC [131].
Cell Demise Manipulated by Metallic Ions in Microenvironment
The cellular microenvironment possesses the capability of significantly affecting the longevity of cells, specifically the quantities of existent metallic ions. A plethora of metallic ions work in the form of active regions for enzymes, aiding in their working in an efficacious manner. Nevertheless, escalated quantities of metallic ions might result in cellular toxicity. The mechanistic modes of cell demise controlling which have been charted as well as their insight has been attained are ferroptosis in addition to cuproptosis.
Ferroptosis
Ferroptosis: A PCD Stimulated by Fe (II) along with ROS
Iron ions possess a significant part in organisms. An iron ion constitutes the active centre of a plethora of proteins. In the form of a crucial protein in reference to metabolism, the iron in addition to sulfur (Fe-S) cluster take part in the electron transport chain (ETC), along with it acts in the form of a co-enzyme factor of CytC as well as variable enzymes. Iron ions further represent significant constituents of heme in addition to an oxygen carrier.
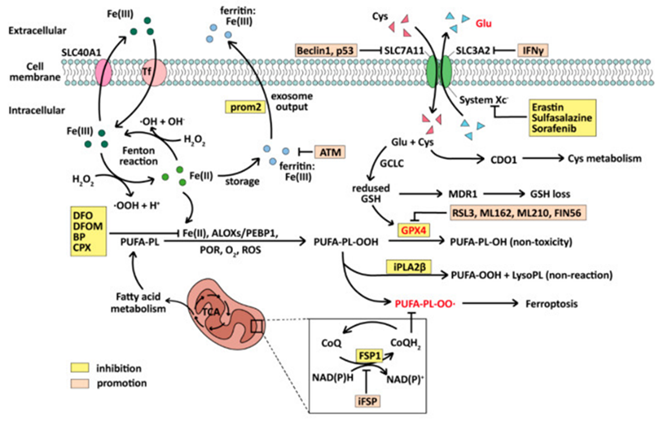
Ferroptosis portrays a kind of cell demise, where ROS along with lipid peroxides get generated by iron metabolism as well as their accrual for generating fatal toxicity in view of cells are not capable of metabolizing them in a smooth manner. ROS constitutes a significant factor resulting in generation of lipid peroxides in the cells that possess robust oxidation actions for instance Fe (II). Ferroptosis basically is controlled by glutathione (GSH) in addition to glutathione peroxidase4 (GPx4) [132], an enzyme that possesses the capability of breaking down lipid peroxides in the cells along with coenzyme Q10 (CoQ10) CoQH2 systems. CoQH2 systems is based on ferroptosis suppressor protein 1 (FSP1) [a flavoprotein CoQ oxidoreductase] [133]. Subsequent to its initial invention in 2012 [9], ferroptosis has been isolated in different diseases, particularly in the treatment of cancer. Following ferroptosis taking place, unique alterations take place in the mitochondrial morphology, inclusive of diminished cristae, rupturing of outer membrane, an escalated quantity of intracellular ROS, which get followed by rupturing of cell membrane resulting in cell demise. Concomitantly biochemical pointers of iron ions are significantly escalated [134]. The crucial aspect of ferroptosis is in the generation as well as accrual of lipid peroxides correlated with fatal toxicity. Basically, it implicates polyunsaturated fatty acids (PUFAs)- phospholipid (PL) -hydroperoxides (PUFA- PL-OOH), whereas Fe (II), ALOXs/PEBP, POR, O2 in addition to ROS possess the capacity of converting PUFA- PL into PUFA- PL-OOH [132], resulting in cell demise.
Accrual of PUFA- PL-OOH is based in the actions of GPx4 along with FSP1. GPx4 possesses the capability of diminishing the PUFA- PL-OOH quantities by converting it to PUFA- PL-OH which has no toxicity [132], whereas CoQH2 formed by FSP1 is capable of capturing lipophilic free radicals as well as hampering the generation PUFA- PL-OO.[133]. In case of hampering of GPx4 along with FSP1 actions, accrual of lipid peroxides along with their diffusion takes place in view of enzymes capable of their breakdown as well as CoQH2, that possesses the capability of avoidance of their transportation are eliminated [133]. Following that a cascade of reactions subsequent to iron in addition to hydrogen peroxide that form hydroxyl radicals gets labelled as Fenton reactions [135]. With the existence of Fe 2+, the intracellular Fenton reactions would persistently form ROS [136], which further leads to lipid peroxidation. In such manner, accrual of considerable quantities of lipid peroxides take place in the cell, damaging the initial enzyme system, resulting in poisoning in addition to breakdown of cell.
Ferroptosis controlling takes place in a myriad of manners-i) the cysteine import system Xc-( cysteine- glutamate transporter receptor) upstream of GPx4 gets hampered along with System Xc gets controlled by hampering agents for instance imidazole ketone erastin (IKE), Sulfasalanazine as well as Sorafenib [137]. System Xc is comprised of a dimer of solute carrier family generation7 members 11 (SLC7A11), along with SLC3A2 that has placement on the cell membrane, which has the basic working of replacement of intracellular glutamate with extracellular cysteine [138], that aid in cysteine entry into the cell. In the form of a cofactor of GPx4, GSH gets formed from glutamate by the of GCLC (glutamate cysteine ligase catalytic subunit) [132]. In case of blockade of cysteine import, a decrease In intracellular GSH generation ensues, leading to diminished GPx4 actions in addition to ferroptosis.
Additionally, ferroptosis induction is further feasible by direct controlling of the reduction of GSH, along with GPx4. GPx4 controlling might be by hampering agents for instance, RSL3, ML162, ML-210, as well as FIN56, whereas GSH generation gets hampered by butylthionine sulphoamine (BSO) [139]. The crucial factor of ferroptosis, Fe (II) might be hampered in a plethora of manners for instance by different hampering agents-i) desferoxamine (DFO), desferoxamine mesylate (DFOM), 2,2’bipyridine (BP) in addition to ciclopirox (CPX) [140]. ii)Alternately its storage might be performed as Fe (III) by ferritin along with exported to the extracellular space through prominin2 prom2) as exosomes [141]. iii) Furthermore, ferroptosis induction might be attained by facilitating ROS generation in view of robust oxidation of ROS.iv) The deactivation of lipid peroxides that are toxic might further influence ferroptosis taking place. iPhosphoLipaseAβ(iPLAβ) believed to be a lipid antidote, possesses the capability of converting PUFA-PL-OOH into non-reactive PUFA-OOH [142]. The event of ferroptosis along with its hampering as well as facilitating factors are displayed in Figure 5.
Figure 5: Courtesy reference no-11The mechanism of ferroptosis and its inhibitors. The whole process revolves around lipid peroxidation and depends primarily on Fe (II) involvement in the activation of the Fenton reaction and the inactivation of GPX4.
Ferroptosis Associated Decontrolled Genes along with Treatments in BC
Tumor infiltrating neutrophils (TINs), that hamper tumor propagation has been labelled as immunrepressive TINs [143]. In reference to BC immunrepressive TINs possesses the capability of upregulating acotinate decarboxylase 1(Acod1) in addition to the generation of it acotinate (ITA). ITA is capable of activating nuclear factor erythroid-2-related factor-2((Nrf2) based antioxidants, hampering the generation of ROS therefore aid tumor cells to escape ferroptosis along with facilitating lung metastasis of BC cells [143]. Acod1 portrays a plausible target for treatment as well as avoidance of metastasis in addition to hampering of Acod1 might facilitate the ferroptosis sensitivity of immunrepressive TINs along with decrease tumor metastasis. Acyl-CoA synthetase long-chain family member 4 (Acsl4) is substantially expressed in endoplasmic reticulum (ER) negative BC cells as well as might embrace cancer cells growth [144]. In case of TNBC oleic acid liberated from mammary fat cells has the capability of hampering lipid peroxidation in addition to subsequent hampering of ferroptosis [145]. Acsl4 is capable of utilizing adenosine triphosphate (ATP) esterification of CoA into free fatty acids (FFA) [146]. Acsl4 further the capability of affecting ferroptosis by affecting the generation of long chain PUFA [[146]. In such case esterification reactions implicated in Acsl4 might be driven in a trajectory, Acsl4 further has the capability of influencing the phenomenon facilitative of ferroptosis, it might be facilitative for demise of ER negative BC cells. Additionally, upregulation of GPx4 took place in the luminal androgen receptor (LAR) sub kind of TNBC. The hampering of GPx4 is capable of stimulating ferroptosis along with tumor immunity in LAR. The combination of GPx4 hampering agents with α-PD-1 immunotherapy escalates the effectiveness of immunotherapy [147].
Other signaling pathways correlated with ferroptosis might aid in BC treatment. i) Ferroptosis is based on the transportation of iron ions as the well as hampering of cysteine import. Hampering ferroportin-1(FPN1) has the capability of disturbing iron transportation, facilitating ferroptosis of BC cells [161]. In case of recalcitrant HER2 positive BC Fibroblast growth factor 4(FGF4) augments cysteine uptake in addition to Fe 2+, actions, through the β-catenin/TCF-4 SLC7A11/ FPN1axis along with hampers ferroptosis [148], pointing that it might be a plausible target for treatment in BC. Ii)Furthermore glutathione cytoplasmic degrading enzyme (CHAC1) is capable of elevating ferroptosis resulting from cysteine starvation via the general controlled non repressed kinase (GCN2- inhibitor eukaryotic initiation factor 2 α (GCN2- eIF-2α- ATG4) pathway in TNBC [149]. iii) Additionally, hepatic leukemia factor (HLF) that is an oncoprotein in TNBC, controls the liberation of transforming growth factor beta1(TGF-β1), by tumor associated macrophages (TAM), by facilitating IL-6. TGF-β1 in turn facilitates HLF for activating gamma glutamyl transferase (GGT), facilitates chemoresistance to tumor cells along with TNBC cells proliferation as well as metastasis by facilitating ferroptosis [150].
Cuproptosis
Cuproptosis: A PCD Stimulated by Cu(I)
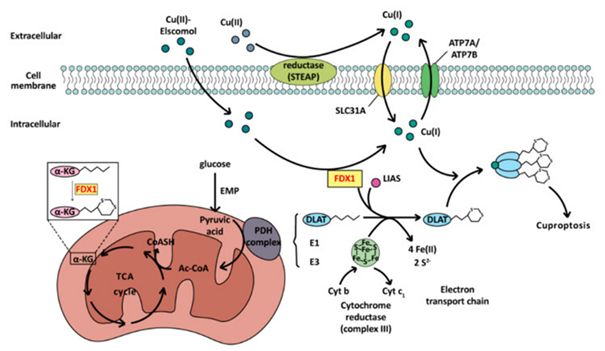
To start with detailing of cuproptosis was done in 2022[10]. Such kind of cell demise varies from PCD in reference to its eventually deadly compound. Here the etiological factor is the escalated accrual of Copper ions (Cu2+) in cells resulting in aberrant oligomerization as well as elimination of actions of lipoylated proteins in addition to eventually cell demise [10,151]. In this case toxicity takes place secondary to copper ions directly, whereas in case of others for instance ferroptosis-toxicity takes place due to lipid peroxides, or apoptosis from caspases.
Copper ions possess a significant part in organisms. They constitute the active centre of a plethora of significant enzymes in cell metabolism for instance CytC oxidases, superoxide dismutase (SOD), ceruloplasmin, lysine oxidase etc. [152,153]. In case of substantially lesser intracellular copper quantities a plethora of metabolic enzymes have depleted functionality. Both Cu+ along with Cu2+ have concomitant presence in the cytoplasm. In the form of a reducing coenzyme factor Cu2+, have the capacity of generating ROS via the Fenton reactions, for instance Fe 2+, In case of substantially greater intracellular quantities [152]. ROS are capable of triggering mitochondrial stress status as well as leading to injury, facilitating mitochondrial autophagy’s [151]. Additionally, copper ions in the form of active centre of SOD, possess the capacity of escalating the enzymatic actions of SOD in addition to forage ROS [153], whereas GSH is capable of diminishing toxicity in view of possesses the capability of binding Cu2+ [154].
Cuproptosis is based on the crucial gene ferredoxin1 (FDX1) along with thiocapryl synthase (LIAS) [15]. The toxicity stems from these reasons that Cu2+ ions possess the capacity of binding fatty acylated pyruvate dehydrogenase system which stimulates them to generate oligomeric proteins, therefore depleting their actions [10,155], along with blockade of generation of acetylCoA. FDX1 is capable of facilitating the fatty acylation of pyruvate dehydrogenase (PDH) as well as α ketoglutarate in addition to further possesses the capability of reducing Cu2+ into Cu+, augmenting the toxicity of copper ions. LIAS has the capacity of generating PDH co- factor lipoic acid (LA) along with is capable of total fatty acylation of modifications by the Fe-S cluster subsequent to binding with FDX1 [156], that constitutes the eventual accomplishment cuproptosis. At the time of such events, substantially greater amounts of Fe-S clusters get utilized leading to damage of the electron transport chain (ETC) [10]. Concurrently, in view of blockade of transformation of pyruvate into acetylCoA, the tricarboxylic acid cycle (TCA alias Kreb’s cycle), gets hampered leading to robust mitochondrial impairment as well as resulting in cell poisoning in addition to final demise. The event of total cuproptosis is displayed in Figure 6.
Figure 6: Courtesy reference no-11The mechanism of cuproptosis. As a cell death mode caused by exogenous factors, excessive accumulation of copper ions mainly affects the TCA cycle during cuproptosis. Dihydrolipoamide transacetylase (E2) of the PDH complex is oligomerized, regulated by ferredoxin FDX1 and the influence of copper ions, resulting in cell death.
In view of presence of copper ions might further results in autophagy along with facilitating ferroptosis[151], currently the morphological properties of cuproptosis continue to be uncharted .It has been recognized that once cuproptosis takes place in view of hampering of TCA cycle , biochemical pointers in cells would go through significant alterations inclusive of ,not restricted to accrual of copper ions, pyruvate, αketoglutarate as well as succinyl CoA[10].They portray a significant index for isolation of cuproptosis.
Cuproptosis Associated Decontrolled Genes along with Treatments in BC
In view of proliferation of cancer cells metabolic enzymes associated with proliferation in addition to respiration generally possess robust actions in contrast to normal cells. In the form of a co-enzyme factor intricately associated with metabolism, copper ions possess greater quantities in cancer tissues in contrast to normal cells [157]. Copper ions r are capable of facilitating angiogenesis [158], endorse cell metabolism along with respiration, contributes to gene transcription as well as expression [157,159]. Thereby copper insufficiency treatments have been formed [159], whose aim is repressing cancer by diminishing the quantities of copper ions in the TME, via the utilization of copper chelators. In case of stage III/IV TNBC, tetrathiomolybdate (TM- a copper chelator) has displayed better effectiveness as well as practically negligible toxicity [160].
Copper ions further are capable of facilitating PD-L-1 expression on the tumor cell membrane surface [160,161], that is exceptional. for the placement of anti-cancer agents targeting PD-L-1. Greater quantities of copper ions facilitate the ubiquitination breakdown of PD-L-1 in addition to facilitate the activation along with proliferation of tumor antigen particular CD8+T cells as well as NK cells [162]. Under such circumstances cancer dependent nano agents possess pronounced benefits. Avoidance of direct entry of copper ions takes place in the body on utilization of nanogel for its administration as well as controlling modifications of its liberation in addition to diminishing toxicity. Lu, et al. [163], fashioned a copper dependent nano medicine labelled as Cu ETNP’s which work against GSH for escalating the actions of copper ions, aiding in reverting the cisplatin chemotherapy resistance along with hampering tumor proliferation by activating cuproptosis [163]. NP@ESC, a nano agent fashioned by Guo et al. [164], ROS sensitive polymers as well as elescomol in addition to copper ions are capable of aiding α-PD-1 in cancer by facilitating PD-L-1 expression on the tumor cell surface along with causing improvement of the effectiveness of immunotherapy [164].
Apparently, there is enrichment of metallic ions transportation in the TNBC tissue. In case of escalating the quantities of zinc as well as copper ions in the TNBC cells, the copper homeostasis would breakdown in the tumor cells, activating cuproptosis of tumor cells, enhanced chemical sensitivity in addition to hamper proliferation of tumor cells [165]. Cuproptosis related gene (CRG) illustrate greater than mean expression in TNBC [165,166]. SLC31A1, a copper transporter possesses significantly greater expression in BC cells in contrast to normal cells [167,168]. Patients with little lesser quantities of expression of SLC31A1 possess favourable prognosis [167], since lesser absorption of copper ions by tumor cells for proliferation in addition to angiogenesis, ease of enabling regulation of tumor cells. Nevertheless, in cases of greater expression of SLC31A1, advantage might be taken of such circumstances to fashion copper enriched nano agents, since greater absorption of nano agents takes place by cancer cells in contrast to normal cells with ease, causing improvement of cancer therapy by activating cuproptosis. Additionally, SLC31A1 is positively associated with immune checkpointsCD274, CTLA4 (CTL associated antigen 4) CTLA4/ CD152 that get controlled by LINC 00614 / miR 204-5-p /SLC31A1 controlling axis [168], pointing that SLC31A1 might be plausible target for adjuvant immunotherapy. Furthermore, anticipation of cancer in adjuvant immunotherapy might get utilized in TNBC patients in promoting therapy by utilizing CRG [166].
Future perspectives along with Directions
Existence of plethora of cell demise methodologies are there inclusive of autophagy, apoptosis, pyroptosis, necroptosis, ferroptosis along with cuproptosis, that possess variable controlling signaling pathways as well as separate mechanistic modes in various cancers in addition to variable agent therapies. Recently subsequent to the invention of PANoptosis, that portrays cross controlling of mechanistic modes of PCD’s, parts of variable demise modes on which work has been conducted at variable times earlier have been recently clubbed together, pointing that controlling of cell demise has the requirement of a holistic approach.
Other than PANoptosis, Rest of interactions of cell demise in BC
The interactions of variable cell demise designs take place in separate backgrounds, apart from in reference to apoptosis, pyroptosis, necroptosis, further amongst ferroptosis, apoptosis along with autophagy. Autophagy modulated by ATG5 possesses the capability of breaking down ferritin inclusive of ferritin light polypeptide (FTL1) as well as ferritin heavy polypeptide (FTH1) in addition to facilitate ferroptosis [169]. In case of BC extensive quantities of iron modulated ROS generation are capable of stimulating autophagy without the existence of ferroptosis [170]. Additionally, the lncRNA LINC 00618 is capable of diminishing the expression of SLC31A1, hampering ferroptosis, along with facilitate the expression of BAX, therefore facilitating apoptosis [171]. Furthermore, erastin is capable of stimulating ferroptosis/ apoptosis, whereas based on the activation of PUMA. In case of activation of PUMA by erastin, apoptosis gets stimulated, whereas on the inactivation of PUMA, ferroptosis gets stimulated [172].
ROS has invariably been at the centre stage with regards to variable cell demise modes. ROS possess the capacity of facilitating AMPK pathway as well as ATG4 oxidation for activating autophagy [173]. ROS has the capacity of facilitating signaling pathways for instance NFκB in addition to activator protein 1(AP1) [174], stimulating inflammation, facilitating MOMP along with CytC [175], for facilitating apoptosis. ROS further is capable of activating pyroptosis through the ROS -JNK- caspase-3-GSDME [176]. Mitochondrial ROS has the capacity of auto phosphorylating RIP1, aid RIP 3 enrolment into necrosome [51], as well as facilitate necroptosis. ROS further is capable of facilitating ferroptosis via lipid peroxidation [177]. with regards to cuproptosis, copper ions possess the capacity of facilitating ROS generation in addition to stimulating OS, escalating the injury to the cells [178]. Additionally, ROS might be hampered by NFS1, that facilitates Fe-S clusters, therefore influencing apoptosis, pyroptosis, necroptosis, along with ferroptosis [179]. Cu (II) possesses the capability of facilitating ROS as well as enhance utilization of GSH via the Fenton reactions, facilitating ferroptosis [180]. Additionally, the enhanced Cu (II) is capable of utilizing Fe-S clusters, to facilitate cuproptosis [10]. The interactions amongst cell demise might be used in a greater advantageous manner in the treatment of BC, particularly with the generation of associated agents. In future scientific researchers for sure would concentrate greater on the interactions of plethora of modes of cell demise.
The Complicated Nature of Modes of Cell Demise: Mutations Along with Chemotherapeutic Resistance
The mechanistic modes of cell demise possess a crucial part in the generation of BC, in addition to genetic mutations possess specifically important influence on such events. The PI3K / phosphatase as well as tension homolog (PTEN) pathway correlated with autophagy, possesses a significant part in BC cells. PI3K possesses the capability of facilitating the generation of PIP3, therefore causing activation of downstream effector molecules PI3K / Akt. The activation of Akt apart from influencing autophagy, further controls apoptosis [181]. PTEN represents a phosphatase implicated in dephosphorylating PIP3 to PIP2 working in the form of negative controller of the PI3K pathway, therefore weakening the PI3K / Akt signaling stepwise patterns. Mutations along with elimination of working of PTEN have been isolated in variable cancers [182], causing activation of PI3K / AKT pathway, that facilitate proliferation, survival as well as metastasis of tumor cells. Furthermore, mutations take place at the time of pyroptosis in BC cells. Mutations of GSDME at the cleavage region of caspase-3 in addition to mutations at the N terminal takes place more commonly. Such mutations significantly decrease spontaneous / agents stimulated pyroptosis along with facilitate survival of BC cells [183].
Apart from genetic mutations demise of BC cells further gets impacted by mutations of factors for instance human epidermal growth factor receptor (EGFR) along with ER mutations. On the binding of growth factor to EGFR causing activation of downstream numerous signaling pathways for instance MAPK/ERK pathway as well as PI3K/ AKT pathway, which are implicated in the controlling of proliferation, differentiation, migration as well as survival [184]. Overexpression/ mutations of EGFR are capable of causing unregulated proliferation in addition to survival aiding in propagation of cancer [185]. Additionally, ER mutations are significant factors aiding in BC. ER mutations are intricately associated with resistance to endocrine treatments in BC [186]. The invention of such molecular mechanistic modes is crucial for the generation of innovative approaches. Scientific researchers have observed that lapatinib possesses the capability of diminishing the phosphorylation of EGFR along with HER2, blockade of Akt, MAPK as well as mTOR signaling pathways, offer resistance to hampering actions of insulin like growth factor1(IGF1) signal, facilitate apoptosis of BC cells in addition to reverting the resistance of HER2 positive BC to trastuzumab [187]. Additionally, lapatinib was illustrated to have just the inimical sequelae of canonical chemotherapeutic agents for instance rash, diarrhoea, nausea, along with fatigue [187], as well as its combination with trastuzumab does not possess extensive toxicity.
Resistance to chemotherapeutic agents has been an area of trouble in reference to cancer treatment in addition to resistance of cancer cells is frequently escalated /dampened by the expression of some genes with the influence of treatment with chemotherapeutic agents, leading to evasion of cell demise [188,189]. For instance, in HER2 positive BC patients getting neoadjuvant chemotherapy, frequently overexpression of DNAJC12 takes place to offer resistance to DOX via the DNAJC12-HSP70- Akt signaling, axis hampering ferroptosis along with apoptosis resulting in resistance to chemotherapeutic agents [190]. Additionally, in ER positive BC patients subsequent to tamoxifen (Tax) treatment expression of transcriptional regulator nuclear protein (NUPR1) was significantly upregulated as well as resistance formed. The exposition for same is upregulation of NUPR1 leading to escalated autophagy that in turn stimulates autophagy cryoprotection mechanistic mode, therefore causing survival of cancer cells [191]. In case of TNBC overactivation of Src homology containing phosphatase 2(SHP2) results in resistance to pyroptosis via the JNK-/ NFκB/ caspase-1/-GSDMD pathway. combination of SHP2 hampering with antiPD-L-1 has illustrated greater effectiveness of treatment [191].
Noticeably, one single BC treatment might result in variable modes of cell demise in various BC cells. For instance, cisplatin a frequently employed chemotherapeutic agent is capable of stimulating variable modes of cell demise. In case of MCF7 cells (ER positive) as well as MDA-MB231 cells (triple negative) BC cell lines cisplatin has the capacity of stimulating autophagy in addition to apoptosis [192]. Nevertheless, the reactions of such 2cell kinds are not completely akin. with the idea of utilizing cisplatin for stimulating pyroptosis, MDA-MB231 cells possess greater susceptibility to pyroptosis, whereas MCF7 cells do not go through pyroptosis with use of cisplatin [193].
Conclusion
BC portrays the commonest cancer globally associated with maximum mortality with escalating incidence along with demise. The mechanistic modes correlated with the variable subkinds of BC as well as complicated carcinogenic factors inclusive of role of dysregulated cholesterol metabolism including oxysterols in the pathophysiology of BC & Syndecans-the signaling controllers possessing prognostic targetable therapeutic significance in BC continue to be not completely charted [194,195]. Since chemotherapeutic resistance in addition to inimical sequelae of radiotherapy to the human body along with benefits of targeted treatment have been emphasized. Kong et al [196], generated an innovative nanodrug with the idea of sensitization of photothermal chemotherapy for BC in vitro. They generated polylactic acid-based drug nanoparticles (PLA@DOX/GA/ICG) possessing greater photothermal transformation capacity by encasing the water-soluble anticancer drug doxorubicin (DOX), photothermal transformation agent indocyanine green (ICG) and liposoluble drug gambogic acid (GA) using a double emulsion methodology. Gambogic acid entrapped in PLA@DOX/GA/ICG nanoparticles might work in the form of HSP90 protein hamper or to acquire bidirectional sensitization to chemotherapy along with photothermal therapy under 808 nm laser irradiation for the first time, which attenuated efficaciously BC cells in vitro. Such nanodrug was anticipated to get utilized for the efficacious BC therapy [196]. Greater such combinations were explored recently by Fu et al. [197], in reference to using cell demise in the form of hampering metastasis in addition to improvement of prognosis of BC by utilizing necroptosis, ferroptosis along with pyroptosis with canonical therapies including depleting cancer stem cell (CSC) [197].
Overall combinations of such cell demise mechanistic modes inclusive of autophagy, apoptosis, pyroptosis, necroptosis, ferroptosis along with cuproptosis, with the variable treatment modes for instance chemotherapeutic agents, radiotherapy, endocrine therapy, etc. would pave the path for achieving greater efficient treatment of BC.
References
- Siegel, R. L., Miller, K. D., Wagle, N. S., & Jemal, A. (2023). Cancer statistics, 2023. CA: A Cancer Journal for Clinicians, 73(1):17–48.
Publisher | Google Scholor - Fuentes, J. D. B., Morgan, E., Aguilar, A. D. L., Mafra, A., Shah, R., Guisti, F., et al. (2024). Global stage distribution of breast cancer at diagnosis. JAMA Oncology, 10(1):71–78.
Publisher | Google Scholor - Lucasiewicz, S., Czeczelewski, M., Forma, A., Baj, J., Sitarz, R., & Stale, A. (2021). Breast cancer – Epidemiology, risk factors, classification, prognostic and current treatment strategies: A review. Cancers (Basel), 13(17):4287.
Publisher | Google Scholor - Prat, A., Cheang, M. C. U., Martin, M., Parker, J. S., Carrasco, F., Caballero, R., et al. (2013). Prognostic significance of progesterone receptor tumor cells within immunohistochemically defined luminal A breast cancer. Journal of Clinical Oncology, 31(2):203–209.
Publisher | Google Scholor - Ding, Y., Chen, X., Liu, C., Ge, W., Wang, Q., Hao, X., et al. (2021). Identification of a small molecule as inducer of ferroptosis and apoptosis through ubiquitination of GPX4 in triple-negative breast cancer cells. Journal of Hematology & Oncology, 14:1–21.
Publisher | Google Scholor - D’Arcy, M. S. (2019). Cell death: A review of the major forms of apoptosis, necroptosis and autophagy. Cell Biology International, 43(6):582–592.
Publisher | Google Scholor - Tang, R., Xu, J., Zhang, B., Liu, J., Liang, C., Hua, J., et al. (2020). Ferroptosis, necroptosis and pyroptosis in anticancer immunity. Journal of Hematology & Oncology, 13:110.
Publisher | Google Scholor - Zhu, M., Liu, D., Liu, G., Zhang, M., & Pan, F. (2023). Caspase-linked programmed cell death in prostate cancer: Apoptosis, necroptosis and pyroptosis to PANoptosis. Biomolecules, 13(12):1715.
Publisher | Google Scholor - Dixon, S. J., Lemberg, K. M., Lamprecht, M. R., Skouta, R., Zaitsev, E. M., Gleason, C. E., et al. (2012). Ferroptosis, an iron-dependent nonapoptotic form of cell death. Cell, 149(5):1060–1072.
Publisher | Google Scholor - Tsvetkov, P. T., Coy, S., Petrova, B., Dreishpoon, M., Verma, A., Abdusamad, M., et al. (2022). Copper induces cell death by targeting lipoylated TCA cycle proteins. Science, 375(6586):1254–1261.
Publisher | Google Scholor - Qian, J. Y., Zhao, L., Xu, L., Zhao, J., Tang, Y., Yu, M., et al. (2024). Cell death: Mechanisms and potential targets in breast cancer therapy. International Journal of Molecular Sciences, 25(18):9703.
Publisher | Google Scholor - Deter, R. L., & de Duve, C. (1967). Influence of glucagon, an inducer of cellular autophagy, on some physical properties of rat liver lysosomes. Journal of Cell Biology, 33(2):169–174.
Publisher | Google Scholor - Takeshige, K., Baba, M., Tsuboi, S., Noda, T., & Ohsumi, Y. (1992). Autophagy in yeast demonstrated with proteinase-deficient mutants and conditions for its induction. Journal of Cell Biology, 119(2):301–311.
Publisher | Google Scholor - Tsukada, M., & Ohsumi, Y. (1993). Isolation and characterization of autophagy-defective mutants of Saccharomyces cerevisiae. FEBS Letters, 333(1–2):169–174.
Publisher | Google Scholor - Kaushik, S., & Cuervo, A. M. (2012). The coming of age of chaperone-mediated autophagy. Nature Reviews Molecular Cell Biology, 12(6):263–276.
Publisher | Google Scholor - Yamamoto, H., & Matsui, T. (2024). Molecular mechanisms of macroautophagy, microautophagy, and chaperone-mediated autophagy. Journal of Nippon Medical School, 91(1):2–9.
Publisher | Google Scholor - Nakatogawa, H., Suzuki, K., Kamada, Y., & Ohsumi, Y. (2009). Dynamics and diversity in autophagy mechanisms: Lessons from yeast. Nature Reviews Molecular Cell Biology, 10(7):458–467.
Publisher | Google Scholor - Cocco, S., Leone, A., Piezzo, M., Caputo, R., Di Lauro, V., Di Rella, F., et al. (2020). Targeting autophagy in breast cancer. International Journal of Molecular Sciences, 21(21):7836.
Publisher | Google Scholor - Huang, R., Xu, Y., Wan, W., Shou, X., Qian, J., et al. (2015). Deacetylation of nuclear LC3 drives autophagy initiation under starvation. Molecular Cell, 57(3):456–466.
Publisher | Google Scholor - Ichimura, Y., Kirisako, T., Takao, T., Satomi, Y., Shimonishi, Y., Ichihara, N., et al. (2000). A ubiquitin system mediates protein lipidation. Nature, 408(6811):488–492.
Publisher | Google Scholor - Kimmelman, A. C., & White, E. (2017). Autophagy and tumor metabolism. Cell Metabolism, 25(5):1037–1043.
Publisher | Google Scholor - Yu, T., Guo, F., Yu, Y., Sun, T., Ma, D., Han, J., et al. (2017). Fusobacterium nucleatum promotes chemoresistance to colorectal cancer by modulating autophagy. Cell, 170(3):548–563.
Publisher | Google Scholor - Hayashi, M. T., Cesare, A. J., Rivera, T., & Karlseder, J. (2015). Cell death during crisis is mediated by telomere deprotection. Nature, 522(7557):492–496.
Publisher | Google Scholor - Nassour, J., Radford, R., Correia, A., Fusté, J. M., Schoell, B., Jauch, A., et al. (2019). Autophagic cell death restricts chromosomal instability during replicative crisis. Nature, 565(7741):659–663.
Publisher | Google Scholor - Lam, M. F. I., & de Michael, K. M. (2022). Caloric restrictions and breast cancer treatment: A mini review. Journal of Molecular Medicine, 100(8):1095–1109.
Publisher | Google Scholor - Levy, J. M. M., Towers, C. G., & Thorburn, A. (2017). Targeting autophagy in cancer. Nature Reviews Cancer, 17(9):528–542.
Publisher | Google Scholor - Li, Z., Chen, B., Wu, A. G., Huang, Y. Z., Shao, G. L., Ji, S. F., et al. (2010). Genetic and epigenetic silencing of the beclin 1 gene in sporadic breast tumors. BMC Cancer, 10:98.
Publisher | Google Scholor - Wang, M. C., Wu, A. G., Huang, Y. Z., Shao, G. L., Ji, S. F., Wang, R. W., et al. (2015). Autophagic regulation of cell growth by altered expression of beclin 1 in triple-negative breast cancer. International Journal of Clinical and Experimental Medicine, 8(5):7049–7058.
Publisher | Google Scholor - Wijshake, T., Chen, B., Zou, Z., Zhong, L., Xiao, G., Xie, Y., et al. (2021). Tumor-suppressor function of beclin 1 in breast cancer cells requires E-cadherin. Proceedings of the National Academy of Sciences of the United States of America, 118(7):e2020478118.
Publisher | Google Scholor - Liang, X. H., Jackson, S., Seaman, M., Brown, K., Kempkes, B., Hibshoosh, H., & Levine, B. (1999). Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature, 402(6762):672–676.
Publisher | Google Scholor - Lin, S., & Gregory, R. I. (2015). MicroRNA biogenesis pathways in cancer. Nature Reviews Cancer, 15(6):321–333.
Publisher | Google Scholor - Esparza-Garrido, R. R., Torres-Marquez, M. E., Rodrigues, V., Velasquez-Wong, A. C., Gomez, F. S., et al. (2016). Breast cancer cell line MDA-MB-231 miRNA expression profiles after BIK interference: BIK involvement in autophagy. Tumor Biology, 37(5):6749–6759.
Publisher | Google Scholor - Soni, M., Patel, Y., Markoutsa, E., Jie, C., Liu, S., Xu, P., et al. (2018). Autophagy, cell viability and chemoresistance regulated by miR-489 in breast cancer. Molecular Cancer Research, 16(9):1348–1360.
Publisher | Google Scholor - Ueda, S., Suda, K., Takanashi, M., Kanekura, K., & Kuroda, M. (2020). miR-27a ameliorates chemoresistance of breast cancer cells by disruption of reactive oxygen species and impairment of autophagy. Modern Pathology, 33(5):863–873.
Publisher | Google Scholor - Tian, W., Alsaadi, R., Guo, Z., Kalinina, A., Carrier, M., Tremblay, M. E., et al. (2019). An antibody for analysis of autophagy induction. Nature Metabolism, 1(3):232–239.
Publisher | Google Scholor - Han, X., Mo, J., Yang, Y., Wang, Y., & Lu, H. (2022). Crucial role of lncRNAs-mediated autophagy in breast cancer. International Journal of Medical Sciences, 19(7):1082–1092.
Publisher | Google Scholor - Ferrè, F., Colantoni, A., & Helmer-Citterich, M. (2016). Revealing protein–lncRNA interactions. Briefings in Bioinformatics, 17(1):106–116.
Publisher | Google Scholor - Xue, X., Yang, A., Zhang, A., Fong, K. W., Kim, J., Song, B., et al. (2016). LncRNA HOTAIR enhances ER signaling and confers tamoxifen resistance. Oncogene, 35(21):2746–2755.
Publisher | Google Scholor - Li, Z., Qian, J., Li, J., & Zhu, C. (2019). Knockdown of lncRNA HOTAIR downregulates the drug resistance of breast cancer cells to doxorubicin via the PI3K/AKT/mTOR signaling pathway. Experimental and Therapeutic Medicine, 18(1):435–442.
Publisher | Google Scholor - Zhang, H., Zhang, N., Liu, Y., Su, P., Liang, Y., Li, Y., et al. (2019). Epigenetic regulation of NAMPT by NAMPT-AS drives progression of triple-negative breast cancer. Cancer Research, 79(13):3347–3359.
Publisher | Google Scholor - Zhang, X. H., Li, B. F., Ding, J., Shi, L., Ren, H. M., Liu, K., et al. (2020). LncRNA DANCR–miR-758-3p–PAX6 molecular networks regulate apoptosis and autophagy in breast cancer. Cancer Management and Research, 12:4073–4084.
Publisher | Google Scholor - Malireddi, R. K. S., Kesavardhana, S., & Kanneganti, T.-D. (2019). ZBP1 and TAK1: Master regulators of NLRP3 inflammasome/pyroptosis, apoptosis, and necroptosis (PANoptosis). Frontiers in Cellular and Infection Microbiology, 9:406.
Publisher | Google Scholor - Yang, J., Zhao, Y., & Shao, F. (2015). Non-canonical activation of inflammatory caspases by cytosolic LPS in innate immunity. Current Opinion in Immunology, 32:78–83.
Publisher | Google Scholor - Shi, J., Zhao, Y., Wang, Y., Gao, W., Ding, J., Li, P., et al. (2014). Inflammatory caspases are innate immune receptors. Nature, 514(7521):187–192.
Publisher | Google Scholor - Kantari, C., & Walczak, H. (2011). Caspase-8 and Bid: Caught in the act between death receptors and mitochondria. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research, 1813(4):558–563.
Publisher | Google Scholor - Cain, K., Bratton, S. B., & Cohen, G. M. (2002). The Apaf-1 apoptosome: A large caspase-activating complex. Biochimie, 84(2-3):203–214.
Publisher | Google Scholor - Hu, L., Chen, M., Chen, X., Zhao, C., Fang, Z., Wang, H., et al. (2020). Chemotherapy-induced pyroptosis is mediated by BAK/BAX–caspase-3–GSDME pathway and inhibited by bromopalmitate. Cell Death & Disease, 11:1–17.
Publisher | Google Scholor - Yee, Y. H., Chong, S. J. F., Kong, L. R., Goh, B. C., & Pervaiz, S. (2020). Sustained IKKβ phosphorylation and NF-κB activation by superoxide-induced peroxynitrite-mediated nitrotyrosine modification of B56γ3 and PP2A inactivation. Redox Biology, 41:101834.
Publisher | Google Scholor - Karin, M., & Lin, A. (2002). NF-κB at the crossroads of life and death. Nature Immunology, 3(3):221–227.
Publisher | Google Scholor - Abais, J. M., Xia, M., Zhang, Y., Boini, K. M., & Li, P. L. (2015). Redox regulation of NLRP3 inflammasome: ROS as trigger or effector? Antioxidants & Redox Signaling, 22(13):1111–1129.
Publisher | Google Scholor - Zhang, Y., Su, S. S., Zhao, S., Yang, Z., Zhong, C. Q., Chen, X., et al. (2017). RIP1 autophosphorylation is promoted by mitochondrial ROS and is essential for RIP3 recruitment into necrosome. Nature Communications, 8:14329.
Publisher | Google Scholor - Kerr, J. F., Wyllie, A. H., & Currie, A. R. (1972). Apoptosis: A basic biological phenomenon with wide-ranging tissue kinetics. British Journal of Cancer, 26(4):239–257.
Publisher | Google Scholor - Nissing, C., & Ryan, K. M. (2023). Fifty years on and still alive: Apoptosis, a biological phenomenon with wide-ranging implications in tissue kinetics. British Journal of Cancer, 128(3):426–431.
Publisher | Google Scholor - Yoon, J. H., & Gores, J. (2002). Death receptor-mediated apoptosis in the liver. Journal of Hepatology, 37(3):400–410.
Publisher | Google Scholor - Sokolowski, J. D., Gamage, K. K., Heffron, D. S., LeBlanc, A. C., Deppmann, C. D., & Mandell, J. W. (2014). Caspase-mediated cleavage of actin and tubulin is a common and sensitive marker for axonal degeneration in neural development and injury. Acta Neuropathologica Communications, 2:16.
Publisher | Google Scholor - Michels, J., Johnson, P. W., & Packham, G. (2005). Mcl-1. International Journal of Biochemistry & Cell Biology, 37(2):267–271.
Publisher | Google Scholor - Tahzib, N., Ransom, N., Reitsamer, H., & McKinnon, S. (2003). Alpha-fodrin is cleaved by caspase-3 in chronic ocular hypertensive rat model of glaucoma. Brain Research Bulletin, 62(6):491–495.
Publisher | Google Scholor - Wang, L. G., Liu, X. M., Kreis, W., & Budman, D. R. (1999). The effect of antimicrotubule agents on signal transduction pathways of apoptosis: A review. Cancer Chemotherapy and Pharmacology, 44(5):355–361.
Publisher | Google Scholor - Rosen, A., & Casciola-Rosen, L. (1997). Macromolecular substrates for the ICE proteases during apoptosis. Journal of Cellular Biochemistry, 64(1):50–54.
Publisher | Google Scholor - Enari, M., Sakahira, H., Yokoyama, Y. H., Okawa, K., Iwamatsu, A., & Nagata, S. (1998). A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature, 391(6662):43–50.
Publisher | Google Scholor - O’Neill, K. L., Huang, K., Zhang, J., Chen, Y., & Luo, X. (2016). Inactivation of prosurvival Bcl-2 proteins activates Bax/Bak through the outer mitochondrial membrane. Genes & Development, 30(9):973–988.
Publisher | Google Scholor - Kaur, K. K., Allahbadia, G. N., & Singh, M. (2022). Mode of actions of bile acids in avoidance of colorectal cancer development and therapeutic applications in treatment of cancers: A narrative review. Journal of Pharmacy and Nutrition Sciences, 12(1):35–53.
Publisher | Google Scholor - Xiong, S., Mu, T., Wang, G., & Jiang, X. (2014). Mitochondria-mediated apoptosis in mammals. Protein & Cell, 5(10):737–749.
Publisher | Google Scholor - Chin, H. S., Li, M. X., Tan, I. K. L., Ninnis, R. L., Reljic, B., Scicluna, K., et al. (2018). VDAC2 enables BAX to mediate apoptosis and limit tumor development. Nature Communications, 9:4976.
Publisher | Google Scholor - Tait, S. W. G., & Green, D. R. (2010). Mitochondria and cell death: Outer membrane permeabilization and beyond. Nature Reviews Molecular Cell Biology, 11(9):621–632.
Publisher | Google Scholor - Singh, G., Guibao, C. D., Seetharaman, J., Agarwal, A., Grace, C. R., McNamara, D. E., et al. (2022). Structural basis of BAK activation in mitochondrial apoptosis initiation. Nature Communications, 13:1–15.
Publisher | Google Scholor - Nakano, K., & Vousden, K. H. (2001). PUMA, a novel proapoptotic gene, is induced by p53. Molecular Cell, 7(3):683–694.
Publisher | Google Scholor - Kashyap, D., Garg, V. K., & Goel, N. (2021). Intrinsic and extrinsic pathways of apoptosis: Role in cancer development and prognosis. In R. Donev (Ed.): Advances in Protein Chemistry and Structural Biology, Academic Press, 25:73–120.
Publisher | Google Scholor - Yang, Q. H., Church-Hajduk, R., Ren, J., Newton, M. L., & Du, C. (2003). Omi/HtrA2 catalytic cleavage of inhibitor of apoptosis (IAP) irreversibly inactivates IAPs and facilitates caspase activity in apoptosis. Genes & Development, 17(12):1487–1496.
Publisher | Google Scholor - Parton, M., Dowsett, M., & Smith, I. (2001). Studies of apoptosis in breast cancer. British Medical Journal, 322(7291):1528.
Publisher | Google Scholor - Merino, D., Lok, S. W., Visvader, J. E., & Lindman, G. J. (2015). Targeting BCL-2 to enhance vulnerability to therapy in estrogen receptor-positive breast cancer. Oncogene, 35(16):1877–1887.
Publisher | Google Scholor - Alipour, M., Shekhnejad, R., Fouani, M. H., Bardania, H., & Hosseinkhani, S. (2023). DNAi–peptide nanohybrid small particles target BCL-2 oncogene and induce apoptosis in breast cancer cells. Biomedicine & Pharmacotherapy, 166:115299.
Publisher | Google Scholor - Vaillant, F., Merino, D., Lee, L., Breslin, K., Pal, B., Ritchie, M. E., et al. (2013). Targeting BCL-2 with the BH3 mimetic ABT-199 in estrogen receptor-positive breast cancer. Cancer Cell, 24(1):120–129.
Publisher | Google Scholor - Li, X. X., Wang, D. Q., Sui, C. G., Meng, F. D., Sun, S. L., Zheng, J., et al. (2020). Oleandrin induces apoptosis via activating endoplasmic reticulum stress in breast cancer cells. Biomedicine & Pharmacotherapy, 124:109852.
Publisher | Google Scholor - Ding, W., Chen, C., Li, J., Geng, X., Zhang, H., & Sun, Y. (2022). Quercus acutissima Carruth root extract triggers apoptosis, autophagy, and inhibits cell viability in breast cancer cells. Journal of Ethnopharmacology, 289:115039.
Publisher | Google Scholor - Stan, S. D., & Abtahi, M. (2022). Diallyl trisulfide induces apoptosis in breast ductal carcinoma in situ–derived minimally invasive breast cancer. Nutrients, 14(7):1455.
Publisher | Google Scholor - Gooch, J. L., Christy, B., & Yee, D. (2002). STAT6 mediates interleukin-4 growth inhibition in human breast cancer cells. Neoplasia, 4(4):324–331.
Publisher | Google Scholor - Doseff, A. L., Baker, J. H., Bourgeois, T. A., & Wewers, M. D. (2003). Interleukin-4-induced apoptosis entails caspase activation and suppression of extracellular signal–regulated kinase phosphorylation. American Journal of Respiratory Cell and Molecular Biology, 29(3):367–374.
Publisher | Google Scholor - Kolben, T., Jeschke, U., Reimer, T., Kolben, T., Schmoeckel, E., Semmlinger, A., et al. (2018). Induction of apoptosis in breast cancer cells in vitro by Fas ligand reverse signaling. Journal of Cancer Research and Clinical Oncology, 144(2):249–256.
Publisher | Google Scholor - Wang, S., Xu, Y., Li, C., Tao, H., Wang, A., Sun, C., et al. (2018). Gambogic acid sensitizes breast cancer cells to TRAIL-induced apoptosis by promoting the crosstalk of extrinsic and intrinsic apoptotic signaling. Food and Chemical Toxicology, 117, 334–341.
Publisher | Google Scholor - Breunig, C., Pel, J., Küblbeck, M., Miller, M., Antonelli, D., Erdem, N., et al. (2017). MicroRNA-519a-3p mediates apoptosis resistance in breast cancer cells and their escape from recognition by natural killer cells. Cell Death & Disease, 8(7):e2973.
Publisher | Google Scholor - Sultan, A. S., Marie, M. A., & Sheweita, S. A. (2018). Novel mechanism of cannabidiol-induced apoptosis in breast cancer cell lines. Breast, 41:34–41.
Publisher | Google Scholor - Campbell, M. J., Carlberg, C., & Koeffler, H. P. (2008). A role for the PPARγ in cancer therapy. PPAR Research, 2008:314974.
Publisher | Google Scholor - Elstner, E., Müller, C., Koshizuka, K., Williamson, E. A., Park, D., Asou, H., et al. (1998). Ligands for peroxisome proliferator–activated receptor γ and retinoic acid receptor inhibit growth and induce apoptosis of human breast cancer cells in vitro and in BNX mice. Proceedings of the National Academy of Sciences of the United States of America, 95(15):8806–8811.
Publisher | Google Scholor - Preet, R., Sidharth, S., Saraswat, S. R., Das, S., Nayak, A., Das, D., et al. (2016). Chk inhibitor synergizes quinacrine-mediated apoptosis in breast cancer cells by compromising base excision repair cascade. Biochemical Pharmacology, 105:23–33.
Publisher | Google Scholor - Alvarez, J. V., Pan, T. C., Ruth, J., Feng, Y., & Zhou, A. (2013). Par-4 downregulation promotes breast cancer recurrence by preventing multinucleation targeted therapy. Cancer Cell, 24(1):30–44.
Publisher | Google Scholor - Van Laer, L., Huizing, E. H., Verstreken, M., Van Zuijlen, D., Wauters, J. G., Bossuyt, P. J., et al. (1998). Nonsyndromic hearing impairment is associated with a mutation in DFNA5. Nature Genetics, 20(2):194–197.
Publisher | Google Scholor - Runkel, F., Marquardt, A., Stoeger, C., Kochmann, E., Simon, D., Kohnke, B., et al. (2004). The dominant alopecia phenotypes Bareskin, Rex denuded, and reduced coat 2 are caused by mutations in gasdermin 3. Genomics, 84(5):824–835.
Publisher | Google Scholor - Yu, J., Kang, M., Kim, B., Kwon, J., Song, Y., Choi, Y., et al. (2011). Polymorphisms in GSDMB are associated with asthma susceptibility, atopy, and BHR. Pediatric Pulmonology, 46(7):701–708.
Publisher | Google Scholor - Li, T., Zheng, G., Li, B., & Tang, L. (2021). Pyroptosis: A promising therapeutic target for noninfectious diseases. Cell Proliferation, 54(6):e13137.
Publisher | Google Scholor - Hergueta-Redondo, R. M., Sarrió, D., Megías, D., Mota, A., Rojo-Sebastián, A., et al. (2014). Gasdermin-B promotes invasion and metastasis in breast cancer cells. PLoS ONE, 9(3):e90099.
Publisher | Google Scholor - Saeki, N., Usui, T., Aoyagi, K., Kim, D. H., Sato, M., Mabuchi, T., et al. (2009). Distinctive expression and function of four GSDM genes (GSDMA–D) in normal and malignant upper gastrointestinal epithelium. Genes, Chromosomes & Cancer, 48(3):261–271.
Publisher | Google Scholor - Zou, J., Zheng, Y., Huang, Y., Tang, D., Kang, R., & Chen, R. (2021). The versatile gasdermin family: Their function and roles in diseases. Frontiers in Immunology, 12:751533.
Publisher | Google Scholor - Tamura, M., Tanaka, S., Fujii, T., Komiyama, H., & Ek, A. (2007). Members of a novel gene family, Gsdm, are expressed in epithelium of skin and gastrointestinal tract in a highly tissue-specific manner. Genomics, 89(5):618–629.
Publisher | Google Scholor - Shi, J., Zhao, Y., Wang, K., Shi, X., Wang, Y., Huang, H., et al. (2015). Cleavage of GSDMs by inflammatory caspases determines proapoptotic cell death. Nature, 526(7575):660–665.
Publisher | Google Scholor - Broz, P. (2015). Caspase target drives pyroptosis. Nature, 526(7575):642–643.
Publisher | Google Scholor - Cookson, B. T., & Brennan, M. A. (2001). Proinflammatory programmed cell death. Trends in Microbiology, 9(3):113–114.
Publisher | Google Scholor - Johnson, A. G., Wein, T., Duncan-Lowey, B., Yirmiya, E., Oppenheimer-Shaanan, Y., et al. (2022). Bacterial gasdermins reveal an ancient mechanism of cell death. Science, 375(6577):221–225.
Publisher | Google Scholor - Wang, S., Yuan, Y. H., Chen, N. H., & Wang, H. B. (2018). Mechanism of NLRP3 inflammasome/pyroptosis activation and their role in Parkinson’s disease. International Immunopharmacology, 67:458–464.
Publisher | Google Scholor - Gu, Y., Kuida, K., Tsutsui, H., Ku, G., Hsiao, K., Fleming, M. A., et al. (1997). Activation of interferon-γ-inducing factor mediated by interleukin-1β-converting enzyme. Science, 275(5297):206–209.
Publisher | Google Scholor - Kayagaki, N., Warming, S., Lamkanfi, M., Vande Walle, L., Louie, S., Dong, J., et al. (2011). Non-canonical inflammasome activation targets caspase-11. Nature, 479(7371):117–121.
Publisher | Google Scholor - Shi, X., Sun, Q., Hou, Y., Zeng, H., Cao, Y., Dong, M., et al. (2023). Recognition and maturation of IL-18 by caspase-4 noncanonical inflammasome. Nature, 624(25):442–450.
Publisher | Google Scholor - Hagar, J. A., Powell, D. A., Aachoui, Y., Ernst, R. K., & Miao, E. A. (2013). Cytoplasmic LPS activates caspase-11: Implications in TLR4-independent endotoxic shock. Science, 341(6151):1250–1253.
Publisher | Google Scholor - Deng, W., Bai, Y., Deng, F., Pan, Y., Mei, S., et al. (2022). Streptococcal pyrogenic exotoxin B cleaves GSDMA and triggers pyroptosis. Nature, 602(7897):496–502.
Publisher | Google Scholor - Zhou, Z., He, H., Wang, K., Shi, X., Wang, Y., Su, Y., et al. (2020). Granzyme A from cytotoxic lymphocytes cleaves GSDMB to trigger pyroptosis in target cells. Science, 368(6494):eaaz7548.
Publisher | Google Scholor - Rogers, C., Fernandes-Alnemri, T., Mayes, L., Alnemri, D., & Alnemri, E. S. (2017). Cleavage of DFNA5 by caspase-3 during apoptosis mediates progression to secondary necrotic/pyroptotic cell death. Nature Communications, 8:14128.
Publisher | Google Scholor - Wang, Y., Gao, W., Shi, X., Ding, J., Liu, W., He, H., et al. (2017). Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of gasdermin E. Nature, 547(7661):99–103.
Publisher | Google Scholor - Kong, Q., Xia, S., Pan, X., Ye, K., Li, Z., Li, H., et al. (2023). Alternative splicing of GSDMB modulates killer lymphocyte–triggered pyroptosis. Science Immunology, 8(84):eadg3196.
Publisher | Google Scholor - Wang, S., Chang, C. W., Huang, J., Zeng, S., Zhang, X., Hung, M. C., & Hou, J. (2024). Gasdermin C sensitizes tumor cells to PARP inhibitor therapy in cancer models. Journal of Clinical Investigation, 134(3):e166841.
Publisher | Google Scholor - Hou, J., Zhao, R., Xia, W., Chang, C. W., You, Y., Hsu, J., et al. (2020). PD-L1–mediated gasdermin C switch from apoptosis to pyroptosis in cancer cells facilitates tumor necrosis. Nature Cell Biology, 22(10):1264–1275.
Publisher | Google Scholor - Ma, L., Lan, F., Zheng, Z., Xie, F., Wang, L., Liu, W., et al. (2012). Epidermal growth factor and interleukin-1β synergistically promote ERK1/2-mediated invasive breast ductal cancer cell migration and invasion. Molecular Cancer, 11:79.
Publisher | Google Scholor - Voronov, E., Shouval, D. S., Krelin, Y., Cagnano, E., Benharroch, D., Iwakura, Y., et al. (2003). IL-1 is required for tumor invasiveness and angiogenesis. Proceedings of the National Academy of Sciences of the United States of America, 100(5):2645–2650.
Publisher | Google Scholor - Kantono, M., & Guo, B. (2017). Inflammasome and cancer: The dynamic role of the inflammasome in tumor development. Frontiers in Immunology, 8:1132.
Publisher | Google Scholor - Ji, X., Huang, X., Li, C., Guan, N., Pan, T., Dong, J., & Li, L. (2023). Effect of tumor macrophages on the pyroptosis of breast cancer tumor cells. Cell Communication and Signaling, 21:197.
Publisher | Google Scholor - Gong, W., Fang, P., Leng, M., & Shi, Y. (2023). Promoting GSDME through DNA demethylation increases chemosensitivity of breast cancer MCF-7/Taxol cells. PLoS ONE, 18(3):e0282244.
Publisher | Google Scholor - Zhong, C., Li, Y., Li, W., Lian, S., Li, Y., Wu, C., et al. (2023). Ganoderma lucidum extract promotes tumor cell pyroptosis and inhibits metastasis in breast cancer. Food and Chemical Toxicology, 174:113654.
Publisher | Google Scholor - Zhang, Z., Zhang, H., Li, D., Zhou, X., Qin, Q., & Zhang, Q. (2021). Caspase-3-mediated GSDME-induced pyroptosis in breast cancer cells through ROS/JNK signaling pathway. Journal of Cellular and Molecular Medicine, 25(18):8159–8168.
Publisher | Google Scholor - An, H., Heo, U. S., Kim, P., Lian, Z., Lee, S., Park, J., et al. (2021). Tetraarsenic hexoxide enhances generation of mitochondrial ROS to promote pyroptosis by activating caspase-3/GSDME in triple-negative breast cancer. Cell Death & Disease, 12:159.
Publisher | Google Scholor - Molina-Crespo, A., Cadete, A., Sarrió, D., Gámez-Chiachio, C. M., Martínez, L., Chao, K., et al. (2019). Intracellular delivery of an antibody targeting gasdermin B reduces HER2 breast cancer aggressiveness. Clinical Cancer Research, 25(16):4846–4858.
Publisher | Google Scholor - Oltra, S. S., Colombo, S., Sin, L., Pérez-López, M., Molina-Crespo, A., et al. (2023). Distinct GSDMB protein isoforms and protease cleavage processes differentially drive pyroptotic cell death and mitochondrial damage in cancer cells. Cell Death & Differentiation, 30(6):1366–1381.
Publisher | Google Scholor - Sarrió, D., Rojo-Sebastián, A., Pérez-López, M., Díaz-Martín, E., Martínez, L., et al. (2022). Gasdermin B protumor function in a novel knock-in mouse model depends on the in vivo biological context. Frontiers in Cell and Developmental Biology, 10:813929.
Publisher | Google Scholor - Sun, K., Chen, R. X., Li, J. Z., & Luo, J. X. (2022). LINC00511/miR-573 axis–mediated high expression of gasdermin C associates with poor prognosis and tumor immune infiltration in breast cancer. Scientific Reports, 12:15120.
Publisher | Google Scholor - Liu, X., Miao, M., Sun, J., Wu, J., & Qin, X. (2023). PANoptosis: A potential new target for programmed cell death in breast cancer treatment and prognosis. Apoptosis, 29(3):277–288.
Publisher | Google Scholor - Zhu, F., Zhang, W., Yang, T., & He, S. D. (2019). Complex roles of necroptosis in cancer. Journal of Zhejiang University-SCIENCE B, 20(5):399–413.
Publisher | Google Scholor - Koo, G. B., Morgan, M. J., Lee, D. G., Kim, W. J., Yoon, J. S., Koo, J. S., et al. (2015). Methylation-dependent loss of RIPK3 expression in cancer represses programmed necrosis in response to chemotherapeutics. Cell Research, 20(6):707–725.
Publisher | Google Scholor - Jiao, D., Cai, Z., Choksi, S., Ma, D., Choe, M., Kwon, H. J., et al. (2018). Necroptosis of tumor cells leads to tumor necrosis and promotes tumor metastasis. Cell Research, 28(8):868–870.
Publisher | Google Scholor - Stoll, G., Ma, Y., Yang, H., Kepp, O., Zitvogel, L., Kroemer, G., et al. (2017). Pro-necrotic molecules impact local immunosurveillance in human breast cancer. OncoImmunology, 6(7):e1299302.
Publisher | Google Scholor - Jin, G., Lan, Y., Han, F., Sun, Y., Liu, Z., Zhang, M., et al. (2015). Smac mimetic–induced caspase-independent necroptosis requires RIP1 in breast cancer. Molecular Medicine Reports, 13(4):3159–3166.
Publisher | Google Scholor - Khorsandi, L., Orazizadeh, M., Niazvand, F., Abbaspour, M. R., Mansouri, E., & Khodadi, A. (2017). Quercetin induces apoptosis and necroptosis in MCF-7 breast cancer cells. Bratislavské Lekárske Listy, 118(2):123–128.
Publisher | Google Scholor - Zhang, Z., Li, H. M., Zhou, C., Li, Q., Ma, L., Zhang, Z., et al. (2016). Non-benzoquinone geldanamycin analogs trigger a novel form of death in human breast cancer cells. Journal of Experimental & Clinical Cancer Research, 35:1–13.
Publisher | Google Scholor - Famme, M., Cressey, P. B., Lu, C., Bruno, P. M., Eskandri, A., Hemmann, M. T., et al. (2017). Induction of necroptosis in cancer stem cells using a nickel(II)-dithiocarbamate phenanthroline complex. Chemistry – A European Journal, 23(40):9674–9682.
Publisher | Google Scholor - Stockwell, B. R. (2022). Ferroptosis turns 10: Emerging molecular mechanisms, physiological functions and therapeutic applications. Cell, 185(14):2401–2422.
Publisher | Google Scholor - Doll, S., Freitas, F. P., Shah, R., Aldrovandi, M., da Silva, M. C., Ingold, I., et al. (2019). FSP1 is a glutathione-independent ferroptosis suppressor. Nature, 575(7784):688–692.
Publisher | Google Scholor - Tang, Z., Ju, Y., Dai, X., Ni, N., Liu, Y., Zhang, D., et al. (2021). HO-1–mediated ferroptosis as a target for protection against retinal pigment epithelium degeneration. Redox Biology, 43:101971.
Publisher | Google Scholor - Reyhani, A., McKenzie, T. G., Fu, Q., & Qiao, G. G. (2019). Fenton-chemistry-mediated radical polymerization. Macromolecular Rapid Communications, 40(11):e19004220.
Publisher | Google Scholor - Ma, S., Henson, E. S., Chen, Y., & Gibson, S. B. (2016). Ferroptosis is induced following siramesine and lapatinib treatment of breast cancer cells. Cell Death & Disease, 7, e2307.
Publisher | Google Scholor - Liu, M. R., Zhu, W. T., & Pei, D. S. (2021). System Xc-: A regulatory target of ferroptosis in cancer. Investigational New Drugs, 39:1123–1131.
Publisher | Google Scholor - Koppula, P., Zhuang, L., & Gan, B. (2020). Cysteine transporter SLC7A11/xCT in cancer: Ferroptosis, nutrient dependency, and cancer. Protein & Cell, 14(5):599–620.
Publisher | Google Scholor - Tang, D., Chen, X., Kang, R., & Kroemer, G. (2021). Ferroptosis: Mechanisms and health implications. Cell Research, 31:107–125.
Publisher | Google Scholor - Scarpellini, C., Klejborowska, G., Lanthier, L. C., Hassannia, B., Berghe, T. V., & Augustyns, K. (2023). Beyond ferrostatin: A comprehensive review of ferroptosis inhibitors. Trends in Pharmacological Sciences, 44:902–916.
Publisher | Google Scholor - Brown, C. W., Amante, J. J., Chhoy, P., Elaimy, A. L., Liu, H., Zhu, L. J., et al. (2019). Prominin2 drives ferroptosis resistance by stimulating iron export. Developmental Cell, 51:575–586.
Publisher | Google Scholor - Chen, D., Chu, B., Yang, X., Liu, Z., Jin, Y., Kon, N., et al. (2021). iPLAβ-mediated lipid detoxification controls p53-driven ferroptosis independent of GPx4. Nature Communications, 12:1–15.
Publisher | Google Scholor - Zhao, Y., Liu, Z., Liu, G., Zhuang, Y., Liu, S., Gan, D., et al. (2019). Neutrophils resist ferroptosis and promote breast cancer metastasis through aconitate decarboxylase 1. Cell Metabolism, 30:434–446.
Publisher | Google Scholor - Monaco, M. E., Creighton, C. J., Lee, P., Zou, X., Topham, M. K., & Stafforini, D. M. (2010). Expression of long-chain fatty acyl-CoA synthetase in breast and prostate cancers is associated with sex steroid hormone receptor negativity. Translational Oncology, 3:91–98.
Publisher | Google Scholor - Xie, Y., Wang, B., Zhao, Y., Tao, Z., Wang, Y., Chen, G., & Hu, X. (2022). Mammary adipocytes protect triple-negative breast cancer cells from ferroptosis. Journal of Hematology & Oncology, 15:72.
Publisher | Google Scholor - Doll, S., Proneth, B., Tyurina, Y. Y., Panzilius, E., Kobayashi, S., Ingold, I., et al. (2016). ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nature Cell Biology, 18(10):1137–1148.
Publisher | Google Scholor - Yang, F., Xiao, Y., Ding, J. H., Jin, X., Ma, D., Li, D. Q., et al. (2023). Ferroptosis heterogeneity in triple-negative breast cancer reveals an innovative immunotherapy combination strategy. Cell Metabolism, 35:84–100.e8.
Publisher | Google Scholor - Zou, G., Zheng, S., Xie, X., Ye, F., Hu, X., Tian, Z., et al. (2022). N6-methyladenosine-regulated FGFR4 attenuates ferroptotic cell death in recalcitrant HER2-positive breast cancer. Nature Communications, 13:1–18.
Publisher | Google Scholor - Chen, M. S., Wang, S. F., Hsu, C. Y., Yin, P. H., Yeh, T. S., Lee, H. C., & Tseng, L. M. (2017). CHAC1 degradation of glutathione under cysteine starvation induces necroptosis and ferroptosis in human triple-negative breast cancer cells via the GCN2–eIF2α–ATG4 pathway. Oncotarget, 8:114588–114602.
Publisher | Google Scholor - Li, H., Yang, P., Wang, J., Zhang, J., Ma, Q., Jiang, Y., et al. (2022). HLF regulates ferroptosis development and chemoresistance in triple-negative breast cancer by activating tumor cell–macrophage crosstalk. Journal of Hematology & Oncology, 15:1–6.
Publisher | Google Scholor - Xue, Q., Yan, D., Li, X., Kang, R., Klionsky, D. J., Kroemer, G., et al. (2023). Copper-dependent autophagic degradation of GPx drives ferroptosis. Autophagy, 19:1982–1996.
Publisher | Google Scholor - Balamurugan, K., & Schaffner, W. (2006). Copper homeostasis in eukaryotes: Teetering on a tight rope. Biochimica et Biophysica Acta (Molecular Cell Research), 1763:737–746.
Publisher | Google Scholor - Uriu-Adams, J. Y., & Keen, C. L. (2005). Copper, oxidative stress, and human health. Molecular Aspects of Medicine, 26:268–298.
Publisher | Google Scholor - Saporito-Magrina, C. M., Musacco Sebio, R. N., Andrieux, G., Kook, L., Orrego, M. T., Tuttolomondo, M. V., et al. (2018). Copper-induced cell death and the protective role of glutathione: The implications of impaired protein folding rather than oxidative stress. Metallomics, 10(11):1743–1754.
Publisher | Google Scholor - Wang, Y., Zhang, L., & Zhou, F. (2022). Cuproptosis: A new form of programmed cell death. Cellular & Molecular Immunology, 19:867–868.
Publisher | Google Scholor - Dreishpoon, M. B., Petrova, B., Ward, D. M., & Kaplan, J. (2023). FDX1 regulates cellular protein lipoylation through direct binding to LIAS. Journal of Biological Chemistry, 299:105046.
Publisher | Google Scholor - Blockhuys, S., Celauro, E., Hildesjö, C., Feizi, A., Stål, O., Fierro Gonzalez, J. C., et al. (2017). Defining the human copper proteome and analysis of its expression variation in cancers. Metallomics, 9:112–123.
Publisher | Google Scholor - Michalczyk, K., & Cymbaluk-Ploska, A. (2020). The role of zinc and copper in gynecological malignancies. Nutrients, 12: 3732.
Publisher | Google Scholor - Brady, D. C., Crowe, M. S., Turski, M. L., Hobbs, G. A., Yao, X., & Chiurchiù, A., et al. (2014). Copper is required for oncogenic BRAF signaling and tumorigenesis. Nature, 509:492–496.
Publisher | Google Scholor - Redman, B. G., Esper, P., Pan, Q., Dunn, R. L., Hussain, H. K., & Cooney, K. A., et al. (2003). Phase II trial of tetrathiomolybdate in patients with advanced cancer. Clinical Cancer Research, 9:1666–1672.
Publisher | Google Scholor - Chan, N., Wills, A., Kornhauser, N., Ward, M. M., Lee, S. B., Nackos, E., et al. (2017). Influencing the tumor microenvironment: A phase II study of copper depletion using tetrathiomolybdate in patients with breast cancer at high risk of recurrence and in preclinical models of lung metastasis. Clinical Cancer Research, 23:666–676.
Publisher | Google Scholor - Voli, F., Valli, E., Lerra, L., Kimpton, K., Saletta, F., Giorgi, F. M., et al. (2020). Intratumoral copper modulates PD-L1 expression and influences tumor immune evasion. Cancer Research, 80:4129–4142.
Publisher | Google Scholor - Lu, Y., Pan, Q., Gao, W., Pu, Y., & He, S. (2022). Reversal of cisplatin chemotherapy resistance by glutathione-resistant copper-based nanomedicine inducing cuproptosis. Journal of Materials Chemistry B, 10:6295–6306.
Publisher | Google Scholor - Guo, B., Yang, F., Zhang, L., Zhao, Q., Wang, W., Yin, L., et al. (2023). Cuproptosis induced by ROS-responsive nanoparticles with elesclomol and copper combined with α-PD-1 for enhanced cancer immunotherapy. Advanced Materials, 35:e2212167.
Publisher | Google Scholor - Yang, X., Deng, L., Diao, X., Yang, S., Zou, L., Yang, Q., et al. (2023). Targeting cuproptosis by zinc pyrithione in triple-negative breast cancer. iScience, 26:108218.
Publisher | Google Scholor - Sha, S., Si, L., Wu, X., Chen, Y., & Xiong, H. (2022). Prognostic analysis of cuproptosis-related genes in triple-negative breast cancer. Frontiers in Immunology, 13:922780.
Publisher | Google Scholor - Li, L., Li, L., & Sun, Q. (2022). High expression of cuproptosis-related SLC31A1 gene in triple-negative breast cancer in relation to unfavorable outcomes and deregulated immune infiltration: An analysis of public data. BMC Bioinformatics, 23:350.
Publisher | Google Scholor - Wu, J. H., Cheng, T. C., Zhu, B., Gao, H. Y., Zheng, L., & Chen, W. X. (2023). Identification of cuproptosis-related gene SLC31A1 and upstream lncRNA–miRNA regulatory axis in breast cancer. Scientific Reports, 13:1–15.
Publisher | Google Scholor - Hou, W., Xie, Y., Song, X., Sun, X., Lotze, M. T., Zeh, H. J., et al. (2016). Autophagy promotes ferroptosis by degradation of ferritin. Autophagy, 12:1425–1428.
Publisher | Google Scholor - Ma, S., Dielschneider, R. F., Henson, E. S., Xiao, W., Choquette, T. R., Blackstein, A. R., et al. (2017). Ferroptosis and autophagy-induced cell death occur independently after siramesine and lapatinib treatment in breast cancer cells. PLoS ONE, 12:e0182921.
Publisher | Google Scholor - Wang, Z., Chen, X., Liu, N., Shi, Y., Li, Y., & Ouyang, Z., et al. (2021). Nuclear long noncoding RNA LINC00618 accelerates ferroptosis in a manner dependent on apoptosis. Molecular Therapy, 29:263–274.
Publisher | Google Scholor - Hong, S. H., Lee, D. H., Lee, Y. S., Jo, M. J., & Jeong, S. Y., et al. (2017). Molecular crosstalk between ferroptosis and apoptosis: Emerging role of ER stress-dependent PUMA expression. Oncotarget, 8, 115164–115178.
Publisher | Google Scholor - Filomeni, G., De Zio, D., & Cecconi, F. (2015). Oxidative stress and autophagy: The clash between damage and metabolic needs. Cell Death & Differentiation, 22:377–388.
Publisher | Google Scholor - Glorieux, C., Liu, S., Trachootham, D., & Huang, P. (2024). Targeting ROS in cancer: Rationale and strategies. Nature Reviews Drug Discovery, 23:583–606.
Publisher | Google Scholor - Chipuk, J. E., Bouchier-Hayes, L., & Green, D. R. (2006). Mitochondrial outer membrane permeabilization during apoptosis: The innocent bystander scenario. Cell Death & Differentiation, 13:1396–1402.
Publisher | Google Scholor - Shen, X., Wang, H., Wang, C., Jiang, H., & Chen, J. (2021). Caspase-3/GSDME-dependent pyroptosis contributes to chemotherapy-induced nephropathy. Cell Death & Disease, 12:1–16.
Publisher | Google Scholor - Merkel, M., Goebel, B., Boll, M., Adhikari, A., Maurer, V., & Steinhilber, D., et al. (2023). Mitochondrial reactive oxygen species formation determines ACSL4/LPCAT3-mediated ferroptosis. Antioxidants, 12:1590.
Publisher | Google Scholor - Springer, C., Humayun, S., & Skouta, R. (2024). Cuproptosis: Unraveling the mechanisms of copper-induced cell death and implications in cancer therapy. Cancers, 16:647.
Publisher | Google Scholor - Lin, J. F., Hu, P. S., Wang, Y. Y., Tan, Y. T., Yu, K., & Liao, K., et al. (2022). Phosphorylated NFS1 weakens oxaliplatin-based chemosensitivity of colorectal cancer by preventing PANoptosis. Signal Transduction and Targeted Therapy, 7:1–16.
Publisher | Google Scholor - Wang, Z., Zhou, P., Li, Y., Zhang, D., Chu, F., Yuan, F., et al. (2023). Bimetallic polymerization network increases labile iron pool and robust activation of cGAS/STING to induce ferroptosis-based tumor immunotherapy. Small, 20:e2308397.
Publisher | Google Scholor - Yu, X., Long, Y. C., & Shen, H. (2016). Differential regulatory functions of three classes of phosphatidylinositol and phosphoinositide 3-kinases in autophagy. Autophagy, 12:1723–1736.
Publisher | Google Scholor - Alvarez-Garcia, V., Tawil, Y., Wise, H. M., & Leslie, N. R. (2019). Mechanisms of PTEN loss in cancer: It’s all about diversity. Seminars in Cancer Biology, 59:66–79.
Publisher | Google Scholor - Jiang, W., Wang, X., Zhang, C., Xue, L., & Yang, L. (2020). Expression and clinical significance of MAPK and EGFR in triple-negative breast cancer. Oncology Letters, 19:1842–1848.
Publisher | Google Scholor - El Hejjioui, B., Lamrabet, S., Joutei, S. A., Senhaji, N., Bouhafa, T., Malhouf, M. A., et al. (2023). New biomarkers and treatment advances in triple-negative breast cancer. Diagnostics, 13:1949.
Publisher | Google Scholor - Nahta, R., Yuan, L. X., Du, Y., & Esteva, F. J. (2007). Lapatinib induces apoptosis in trastuzumab-resistant breast cancer cells: Effects of insulin-like growth factor-1 signaling. Molecular Cancer Therapeutics, 6:667–674.
Publisher | Google Scholor - Burris, H. A. (2004). Dual kinase inhibition in the treatment of breast cancer: Initial experience with the EGFR/ErbB2 inhibitor lapatinib. The Oncologist, 9:10–15.
Publisher | Google Scholor - Kaur, K. K., Allahbadia, G. N., & Singh, M. (2021). An update on the screening, diagnosis, and management of breast cancer: A review with considerations for future fertility. International Journal of Medical and All Body Health Research, 4(2):40–53.
Publisher | Google Scholor - Kaur, K. K., Allahbadia, G. N., & Singh, M. (2019). Novel ways of targeting triple-negative breast cancer (TNBC) with the latest research: Will it improve prognoses? Journal of Gynaecology, 8(2):45–53.
Publisher | Google Scholor - Shen, M., Cao, S., Long, X., Xiao, L., Yang, L., & Zhang, P., et al. (2024). DNAJC12 causes breast cancer chemotherapy resistance by repressing doxorubicin-induced ferroptosis and apoptosis via activation of AKT. Redox Biology, 70:103035.
Publisher | Google Scholor - Wang, L., Sun, J., Yin, Y., Sun, Y., Ma, J., Zhou, R., et al. (2021). Transcriptional co-regulator NUPR1 maintains tamoxifen resistance in breast cancer cells. Cell Death & Disease, 12:1–16.
Publisher | Google Scholor - Chen, C., Cheng, Y., Lie, H., Feng, X., Zhang, H., Qi, L., et al. (2023). SHP2 potentiates anti-PD-L1 effectiveness by intervening in cell pyroptosis resistance in triple-negative breast cancer cells. Biomedicine & Pharmacotherapy, 168:115797.
Publisher | Google Scholor - Li, C., He, Z., Yao, F., Liao, S., Sun, K., & Sun, S., et al. (2023). Role of escin in breast cancer: Potential mechanisms for inducing ferroptosis and synergistic antitumor activity with cisplatin. Apoptosis, 28:1154–1167.
Publisher | Google Scholor - Yan, H., Luo, B., Wu, X., Guan, F., Yu, X., Zhao, L., et al. (2021). Cisplatin induces pyroptosis via activation of MEG3/NLRP3/caspase-1/GSDMD pathway in triple-negative breast cancer. International Journal of Biological Sciences
Publisher | Google Scholor - Kaur, K. K., Allahbadia, G. N., & Singh, M. (2023). Dysregulated cholesterol metabolism including oxysterols in the pathophysiology of breast cancer: Therapeutic implications – A narrative review. Acta Scientific Cancer Biology, 7(1):7–14.
Publisher | Google Scholor - Kaur, K. K., Allahbadia, G. N., & Singh, M. (2023). Syndecans – The signaling controllers possessing prognostic targetable therapeutic significance in breast cancer for innovative breast cancer therapy. Macromolecules: An Indian Journal, 16(1):132.
Publisher | Google Scholor - Kong, J. C., Zhou, F., Shi, L., Wie, Y., & Wu, C. (2024). A novel nanodrug for the sensitization of photothermal chemotherapy for breast cancer in vitro. RSC Advances, 14(30):21292–21299.
Publisher | Google Scholor - Fu, B., Lou, Y. M., Wu, P., Lu, X., & Xu, C. (2024). Emerging role of necroptosis, pyroptosis, and ferroptosis in breast cancer: New dawn for overcoming therapy resistance. Neoplasia, 55:101017.
Publisher | Google Scholor