Research Article
Bioactive Potential of Pistacia Chinensis Leaves: A Study on Antioxidant, Antidiabetic, and Antibacterial Properties
1 Rehman Collage of Allied Health Sciences, Rehman Medical Institute, Peshawar, Pakistan.
2 Department of Biochemistry, Faculty of Chemical and Life Sciences, Abdul Wali Khan University Mardan, Pakistan.
3 Department of Agronomy, Faculty of Crop Production, the University of Agriculture Peshawar, Pakistan.
*Corresponding Author: Sadiq Ullah, Rehman Collage of Allied Health Sciences, Rehman Medical Institute, Peshawar, Pakistan.
Citation: Ullah S., Ullah O., Yousaf S., Akbar I., Begum S., et al. (2026). Bioactive Potential of Pistacia Chinensis Leaves: A Study on Antioxidant, Antidiabetic, and Antibacterial Properties, Journal of BioMed Research and Reports, BioRes Scientia Publishers. 10(1):1-8. DOI: 10.59657/2837-4681.brs.26.223
Copyright: © 2026 Sadiq Ullah, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: December 05, 2025 | Accepted: December 19, 2025 | Published: January 06, 2026
Abstract
Pistacia chinensis is a medicinal plant that has been traditionally utilized in the northern regions of Khyber Pakhtunkhwa for therapeutic purposes since antiquity. A decoction derived from its leaves has been employed in the treatment of various ailments, including headaches, loss of appetite, and diabetes. The present investigation aimed to explore the antioxidant, anti-diabetic (through alpha-amylase inhibition), and antibacterial properties of Pistacia chinensis leaves. For this purpose, the leaf material underwent successive solvent extraction employing solvents of ascending polarity: n-hexane, ethyl acetate, chloroform, methanol, ethanol, and distilled water. The antioxidant capacity of each extract was assessed utilizing the ferric ion reducing antioxidant power (FRAP) assay. Antibacterial activity was evaluated through the agar well diffusion method. The extracts exhibited significant antibacterial efficacy against both gram-positive (Klebsiella pneumoniae) and gram-negative bacteria, including Escherichia coli, Haemophilus influenzae, and Acinetobacter species. Furthermore, extracts of Pistacia chinensis demonstrated remarkable alpha-amylase inhibitory activity, indicating potential anti-hyperglycemic effects. To the best of our knowledge, this study represents the inaugural comprehensive report on the antioxidant, antimicrobial, and alpha-amylase inhibitory activities of Pistacia chinensis leaf extracts in comparison to standard reference compounds. These findings substantiate the traditional use of this plant and suggest its prospective application in the development of therapies for diabetes, oxidative stress-related disorders, and bacterial infections.
Keywords: pistacia chinensis; medicinal plants; solvent extraction; phytochemical screening
Introduction
Diabetes mellitus is a chronic metabolic condition and a syndrome marked by an increased glucose level in the blood (hyperglycemia) due to insulin insufficiency or impaired efficacy, with a strong hereditary base. Medicinal plants are regarded as a major source of medicinal compounds in both traditional and modern therapies. According to the World Health Organization (WHO 2002), around 75-80% of the population relies on plant-based medicines to treat their disease [1-2]. Oxidative stress is described as a disruption in the balance between reactive oxygen species (free radicals) and antioxidant protection. Alcohol use, cigarette smoking, nutrition, gender, geographic location—particularly at high elevations—and occupation are risk factors linked to oxidizing species (OS)-induced diseases [3]. Historically, Pistacia lentiscus Leaves has been used to treat hypertension as a stimulant and diuretic. In order to value this species and create new high-value products that can be utilized in the pharmaceutical and agro-food industries, we were interested in examining the chemical profile of the plant's leaves and it’s in vitro, in vivo, and in silicoantioxidant, antimicrobial anticoagulant, and antidiabetic effects.[4] Several investigations have shown that oxidative stress, mostly caused by the production of free radicals brought on by hyperglycemia, plays a role in the onset, development, and progression of diabetes as well as its consequences. Because to membrane lipid peroxidation, protein glycation, and the concurrent deterioration of antioxidant defense mechanisms, an unusually high concentration of free radicals causes damage to membranes [5]. The ability of bacteria to withstand many of the currently available medicines has made bacterial resistance to antimicrobials a major global concern in recent years. Staphylococcus aureus (Staphylococcus aureus) and Pseudomonas aeruginosa (Pseudomonas aeruginosa) are thought to be the most significant organisms that cause nosocomial infections, which have high rates of morbidity and mortality. The following bacterial strains were used to assess the antibacterial properties of essential oils in vitro: Escherichia coli ATCC 25922, Bacillus subtilis ATCC 3366, Staphylococcus aureus ATCC 29213, and Pseudomonas aeruginosa ATCC 27853. These strains were all kept as stock at -20°C in 20% glycerol [6-7]. Gymnemasylvestre, Trigonella foenum, and Phyllanthus emblica are used to control diabetes. The combination of these plant parts in various ratios may result in synergistic antidiabetic effect. As a result, the current study sought to develop polyherbal formulations containing varying proportions of Gymnemasylvestre (Leaves), Trigonella foenum (Seeds), and Phyllanthus emblica (Fruits) in order to investigate their antidiabetic and antihyperlipidemic activity in Streptozotocin-induced diabetic rats. The pharmacological action of a single plant is deemed smaller than that of a polyherbal composition including a ratio of multiple plants. In-vitro and in vivo study used CCl4 as a source of free radicals to induce damage in the lungs and thyroid gland of a rat model. Biochemical and histological studies were conducted on the lungs and thyroid tissues. The current study examined the preventive effect of an ethanol extract of Pistacia chinensis bark (PCEB) in rats against CCl4-induced lung and thyroid damage. PCEB inhibited the rise of thiobarbituric acid-reactive substances, hydrogen peroxide, nitrite, and protein content and restored the levels of antioxidant enzymes, such as catalase, peroxidase, superoxide dismutase, glutathione-S-transferase, glutathione reductase, glutathione peroxidase, gamma-glutamyltranspeptidase, and quinone reductase, in both lung and thyroid tissues of CCl4-treated rats. A decrease in the number of leukocytes, neutrophils, and hemoglobin, as well as the T3 and T4 content, and an increase in monocytes, eosinophils, the lymphocyte count with CCl4 was recovered to normal levels following PCEB therapy. Histological examination of CCl4-treated rats revealed a variety of lung lesions, including rupture of alveolar walls and bronchioles, fibroblast aggregation, and disordered Clara cells [8-9]. The present study investigated the hypoglycemic potential of a fluid extract derived from Pistacia chinensis (leaves). Preliminary phytochemical analysis revealed the presence of bioactive compounds, including reducing carbohydrates, cardiac glycosides, tannins, saponins, glycosides, flavonoids, and steroids. The water-soluble extract of Pistacia chinensis (leaves) exhibited significant inhibitory activity against α-glucosidase and α-amylase enzymes, indicating its potential role in the management of type II diabetes through the modulation of postprandial glucose levels and to evaluate the extract for antibacterial inhibitory potential of Pistacia chinensis (leaves) against specific bacteria.
Materials and Methods
The Pistacia Chinensis leaves were collected from the District Bunner, which is located about 60 kilometers west of Mardan, Pakistan. A member of the botany department's (Abdul Wali khan University Faculty of life Science) made the botanical identification using the method of Aml O Alhadad [7].
Preparation of sample
Using the method of Aml O Alhadad[7] 80 g of the dried leaves of Pistacia Chinensis leave were macerated separately in each 500 mL of five different bioactive compounds for 48 hours to produce the extract. A rotary evaporator was used to dry off the solvent after the resulting extract had been vacuum-filtered. For later usage, the dried extract was kept at 4 °C.
Crude extracts preparation
The following experiments using the method of Wajid khan [10]. The plant's bark was dried in the shadow at room temperature. The dehydrated plant material was ground to a fine powder using an electric grinder. 50ml of each extraction solvent (methanol, ethyl acetate, ethanol, Distal water and n-hexane) was used to dissolve 5 grams of plant powder for each bioactive compound. The solution was kept in a shaking incubator for 48 hours. After shaking, the slurry was filtered using Whatman filter paper No. 1. The filtrate was then dried in a rotary evaporator at 40°C. After full drying, the extract was weighed and then kept in sterilized glass vials for further analysis using disc diffusion dilution assay.
Extract Preparation for Antimicrobial Activity
Antibacterial and antifungal tests were conducted to assess the plant extracts' antimicrobial properties. According to the resources available, four bacteria (Easchercoli, Klebsiella, Hemophilus, Acetobacter) were chosen for this purpose. Pistacia Chinensis leave extract and fraction were tested for antibacterial properties against strains of bacteria in vitro. After preparation and autoclaving, the medium was transferred into petri dishes. The appropriate bacterial strains were sown into each petri dish. Using a micropipette, 15mg/ml of the sample was added to each disc. A positive reference for bacterial strains was streptomycin (5 mg/mL). Following the loading of samples, cultures were left to grow for 24 hours at 37 °C for bacterial strains in incubator. Following the specified period, the microorganisms' activity was measured in millimeters, and the zone of inhibition and average activity were obtained [11].
Culture preparation for different microbial strains
Antimicrobial activity was evaluated against E.coli, S.aureus, P.Aeruginosa, K. Pneumonia, Acetobacters and (H. Influenza) (pure samples of bacterial strains were purchased from a SIGMA life science) by using agar well diffusion method Inoculums of all microbes were prepared in sterilized Lauria-Bertini media gl” (SIGMA life science) in separate test tubes which were then placed in shaker incubator at 37°C for 24 hours to contain approximately 10 cfu/ml.
Agar-well diffusion assay for antimicrobial activity
Antimicrobial activity was evaluated by agar well diffusion method. Lauria-Bertin aga media was prepared and autoclaved at 121°c for 15 minutes which was then cooled and poured in Petri plates under sterilized conditions of laminar flow hood. The wells of 9 mm were bored in each plate and the plates were inoculated with 75µl of inoculum. Each of sample 100µl was pipetted in each well and plates were incubated at 37°c for 24 hours. After 24 hours zones of inhibition were measured and expressed in millimeters.
Antioxidant activity
First the sample was diluted 3-4 times with the same solvent in which it was extracted. Then five different concentrations were prepared by serial dilution while the volume of sample was kept constant to 750µl. Each sample (750µl) was mixed with an equal amount of phosphate buffer (0.2 M. pH 6.6) and 1% potassium ferricyanide (a source of ferric ions). The mixture was incubated at 50°C for 20 minutes followed by addition of (750µl) Trichloroacetic acid (10%) to stop the reaction and was then centrifuged at 3000 rpm for 10 minutes. The upper layer (1.5 ml) was separated and mixed with an equal amount of distilled water and 0.1 ml FeCl3 solution (0.1%). A blank was also prepared by using different concentrations of ascorbic acid as standard by same procedure and the absorbance was measured at 700 nm.
Alpha-amylase inhibitory assay
The effect of plant extracts on α-amylase was assessed in accordance. In short, 10 µL of amylase enzyme solution (0.5 mg/mL), 100 µL of the extracts, and the same volume of sodium phosphate (0.02 M, pH: 6.9) were incubated in a test tube at room temperature for 10 minutes. Next, 100 µL of 1% starch in 0.02 M (pH 6.9) sodium phosphate buffer was added. Additionally, the reaction mixtures were incubated for ten more minutes. After adding 100 µL of dinitro salicylic acid colorant to quench the mixture, it was heated in a water bath until a yellowish orange hue formed. Five milliliters of distilled water were added to the mixture after it had cooled at 540 nm, the absorbance was using a spectrophotometer to measure? whereas is the sample, A0 is the blank solution (solvent only), and Ac is the control (acarbose). This is how the inhibition percentage (%) was calculated.[12] Inhibition (%) ∶1-(A_s∕A (C)) × 100. We used the method for alpha-amylase assay; the final reaction volume was 220 µl in each well of ELISA 96 well plate. Each well contained 220 µl of reaction mixtures (20 µl DMSO +20 ul sample +10 μl enzyme + 50 µl phosphate buffer+20 µl starch+100 μl DNS). Starch was used as a substrate. 20 µl of DMSO was added to each well and 20 µl of each sample was diluted seven times in DMSO then 50 µl of phosphate buffer was added to each well whose PH was (6.9). 20 μl of starch was added to each well and incubate it for 20 min at 37°C. After incubation 10 μl of the enzyme were added to each well. Then finally 100 µl of DNS was added to each well and incubate it for 30 minutes at 37°C. Absorbance was determined at 540 nm using ELISA. Acarbose was used as a standard. All tests were performed in duplicate.
Results and Discussion
We employed a eutectic solvent in our investigation, and the entire procedure was founded on the ideas of green chemistry followed [12].
Anti-oxidant activity
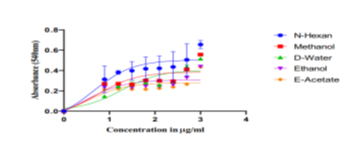
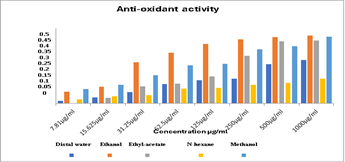
Since antioxidants compete to lower free radicals, no one approach is thought to be enough to account for the different ways that antioxidants work. As a result, this study included many forms of antioxidant capacity assessment [12]. This technique is a popular assay for determining the amount of lipid peroxidation in biological systems and membranes. As can be seen in Fig. 1, CFL and PBF inhibited lipid peroxidation well but not statistically significantly (72.80± 9.67%) and 81.32± 1.27%), respectively (P =0.4169). Free radicals initiate the oxidative chain process known as "lipid peroxidation," in which lipid molecules undergo maximal oxidation one after the other to produce lipid peroxide. When the substrate is exhausted, this chain reaction usually comes to a stop. Additional requirements include two radicals reacting to produce a non-radical product or two radicals reacting with antioxidants to provide readily denotable hydrogen for peroxyl radical abstraction. When Fe2+ combines with H2O2, hydroxyl radicals are produced, which can then take part in lipid peroxidation. The primary cause of this process is ROS; the free radical chain reaction starts in the phosphatide of the biological membrane and starts propagation reactions that damage erythrocytes and induce hemolysis.[12]. Our study investigates in vitro antioxidant activity of several solvent extracts of Pistacia chinensis leaves, including n-hexane, ethyl acetate, ethanol, methanol, and distilled water, was compared to that of normal ascorbic.
Figure 1
Anti-diabetic properties were assessed in vitro using the α-amylase inhibition assay
Regarding the inhibition of α-amylase, the in vitro antidiabetic activity of Pistacia lentiscus leaf extract produced an intriguing finding. Figure 4 and Table 3 demonstrate the inhibitory findings, which were 17.75%, 41.11%, 46.64%, and 68.20% for the concentrations of 0.05 mg/mL, 0.11 mg/mL, 0.23 mg/mL, and 0.45 mg/mL, respectively, with an IC50 value of 0.266 ± 0.01 mg/ml [13].The findings of the investigation by Foddai et al. showed that Pistacia lentiscus extract had a high ability to chelate the enzyme α-amylase[13]. However, there were some phenomena in the inhibitory activity of flavonoids with similar chemical structures, such as myricetin being 40 times more effective than dihydromyricetin. These were explained by the results of molecular docking, which suggested that the inhibitory activity may be influenced by different orientations in the α-amylase active center. α-amylase activity was significantly reduced in a dose-dependent manner by all callus and leaf extracts. The ethyl acetate extract of callus exhibited the strongest inhibition of the three extracts (IC50 value: 328.6 ± 1.99 µg/ml), followed by the methanolic extract (IC50 value: 498.8 ± 2.44 µg/ml) and the chloroform extract (IC50 value: 657.3 ± 1.52 µg/ml) [13,14]. The maximum enzyme inhibition was demonstrated by the leaf methanolic extract (IC50 value 371.5 ± 1.40 µg/ml), which was followed by the chloroform extract (IC50 826.8 ± 1.10 µg/ml) and the ethyl acetate extract (IC50 524.6 ± 1.18 µg/ml). Both the methanolic extract of the leaf and the ethyl acetate extract of the callus demonstrated α-amylase inhibition that was equivalent to that of regular Acarbose [14]. Our study analysis to demonstrate the solvent extracts from Pistacia chinensis leaves were tested for their ability to inhibit α-amylase activity at 4 mg/mL concentration. Pistacia chinensis leaf extracts in n-hexane, ethyl acetate, ethanol, methanol, and distal water were tested for their ability to inhibit α-amylase activity. N-hexane had the strongest α-amylase inhibitory activity (95%), compared to other solvent extracts. At a maximum concentration of 4 mg/ml, the methanol extracts inhibited α-amylase by 88%, while the distal water extract of the leaves inhibited it by 80%. The ethanol extract of the leaves inhibited α-amylase by 75% at a dosage of 4 mg/ml, whereas ethyl acetate inhibited it by 55%. Based on these findings, it is possible to deduce that the usage of these plant extracts will be extremely advantageous in slowing the pace of carbohydrate digestion and absorption, hence contributing to effective diabetes control by lowering hyperglycemia levels. Future research will give insight into the molecular processes by which this plant and its active chemicals regulate glucose homeostasis The inhibition of α-amylase by Pistacia Chinensis leaves in various solvents at varying concentrations. Various solvent extracts (0.2-4mg/ml) were tested for α-amylase inhibitory activity. The aqueous extract demonstrated the highest α-amylase% inhibition. The graph depicted concentration on the X-axis and inhibition on the Y-axis as indicate in figure 2.
Figure 2
Antimicrobial activity analysis
Another set of tests was conducted to investigate the therapeutic potential of Pistacia chinensis against certain bacterial strains. Zones of inhibition were examined and compared to common antibiotics such as ampicillin. In the current investigation, the antibacterial activity of solvent extracts of Pistacia chinensis leaves, including n-hexane, ethanol, methanol, and aqueous extracts, was assessed against chosen bacteria.
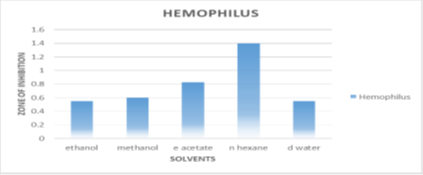
Sensitivity against Haemophilus bacterium
According to conventional streptomycin inhibition, n-hexane inhibits haemophile bacteria the most (1.4±0.141mm). Ethyl acetate inhibits at 0.825±0.106 mm, Ethanol, methanol, and de-ionized water inhibit by 0.55±0.141mm, 0.825±0.106mm, and 0.825±0.106mm, respectively as seen in Figure 3.
Figure 3
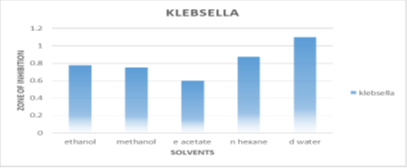
Resistance to Klebsiella influenza pneumonia
The most significant inhibition of distilled water extract against Klebsiella bacteria is 1.1±0.141 mm. In compared to normal ampicillin, n-hexane exhibits 0.875±0.106 mm of inhibition, but ethanol, ethyl acetate, and methanol exhibit lower inhibition of 0.775±0.247 mm, 0.6±0.070 mm, and 0.75±0.212 mm. Figure 4 illustrates.
Figure 4
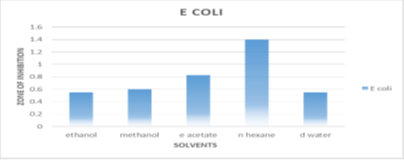
Activity against Escherichia coli
N-hexane inhibits Escherichiacoli bacteria at a maximum of 1.4±0.07 mm, whereas ethanol, methanol, and d water exhibit comparably low inhibition of 0.45±0mm, 0.45±0.14mm, and 0.45±0.070mm.
Figure 5
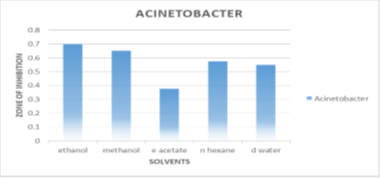
Activity against Acinetobacter haumanni
The extracts with the greatest degree of inhibition against Acinetobacter were ethanol (0.7±0.212mm), methanol (0.65±0.06mm), n-hexane (0.575±0.17 mm), and distal water (0.55±0.070mm), respectively. N-hexane has the highest overall inhibition against all bacteria. Shown in Figure 6.
Zone of Inhibition
Various fractions derived from the crude extract of Pistacia chinensis in comparison with conventional Streptomycin and Ampicillin as shown in Table 1.
Figure 6
Table 1
| Standards | E. coli | Klebsella | Haemeophilus | Acitobacter |
| G–ve | G–ve | G–ve | G –ve | |
| Streptomycin | 15mm | 16mm | 15mm | 16mm |
| Ampiciline | 18mm | _ | 20mm | _ |
| Diameter zone of (Mean±SD) | ||||
| Ethanol | 0.45±0 | 0.775±0.247 | 0.55±0.141 | 0.7±0.212 |
| Ethyl acetate | 0.62±0.10 | 0.6±0.070 | 0.825±0.106 | 0.375±0.035 |
| Methanol | 0.45±0.141 | 0.75±0.212 | 0.6±0 | 0.65±0.061 |
| n-Hexane | 1.4±0.07 | 0.875±0.106 | 1.4±0.141 | 0.575±0.17 |
| Distal water | 0.45±0.070 | 1.1±0.141 | 0.825±0.106 | 0.55±0.070 |
Tannins are currently gaining popularity as both a bioactive component of foods and biological antioxidant. Tannins are a unique class of water-soluble phenolic metabolites with a relatively high molecular weight that may form powerful complexes with carbohydrates and proteins. Tannins had previously been regarded as one of the plant-derived antinutrients due to their tendency to precipitate proteins, impede digestive enzymes, and reduce vitamin and mineral absorption. However, various health advantages have lately been linked to tannin use, and certain epidemiological correlations with lower rates of chronic illness have been identified. Several studies have shown that tannins may have major biological impacts. Tannins are found in many plant-based foods, including legume seeds, cereal grains, fruits, vegetables, and drinks including wine, tea, chocolate, and cider. They are a vital element of human diets and have positive implications for both diet and health. Multiple investigations have shown that tannins have possibly substantial biological effects, such as antioxidant or radical scavenging action, as well as in vitro inhibition of lipid peroxidation and lipoxygenases. Tannins' antioxidant effect is derived from their ability to scavenge free radicals and reactive oxygen species, as well as the chelation of transition metal ions that initiate the oxidation process. Because of their insulin-enhancing capability, antioxidants have been shown to give synergistic advantages in the treatment of diabetes [15]. The variance in effectiveness between a-glucosidase and a-amylase inhibitory activity may be attributed to phytochemicals present in plants that affect one enzyme more than the other, as plants contain a variety of phytochemicals such as flavonoids, tocopherols, carotenoids, and ascorbic acids. A-glucosidase is an enzyme found near the brush edge of the microvillus in the small intestine. This regulating enzyme catalyzes the last phase of carbohydrate metabolism, which transforms oligosaccharides to monosaccharides prior to their absorption into the blood circulation [16]. The antioxidant activity of flavonoids and tannins isolated from Persicaria hydro-piper was measured in vitro using ascorbic acid as a reference. In the assay, the FeCl/K,Fe (CN) complex was used as a source of ferric ions, which might reduce to ferrous ions in the presence of phytochemical substances. This produces a green color complex, the intensity of which is determined spectrophotometrically. Thus, increased absorption indicates more antioxidant activity. The antioxidant activity of plant extracts was evaluated utilizing a ferric ion reducing agent and a free radical scavenging test. The plant's leaves exhibited varying antioxidant activity. It is owing to the existence of a higher concentration of bioactive compounds in the plant's leaves than in its stem. In today's results, the ethanol and methanol extracts of the leaves demonstrated excellent antioxidant activity. It demonstrates that these extracts contain large levels of antioxidant component, which corresponds to significant antioxidant activity. The N-hexane extract of leaves had relatively poor antioxidant activity, indicating that it contains less anti-oxidant. An amylase inhibition experiment was used to assess the anti-diabetic effects of Pistacia Chinensis in vitro. Different solvent extracts of Pistacia Chinensis leaf parts were tested for their capacity to inhibit methanol, while water extracts of Pistacia Chinensis leaf parts were separately assessed for inhibition of a-amylase activity. The ethanol extract had the highest inhibitory effect against α-amlyase when compared to other solvents. Among all solvent extracts of Pi chinensis leaves, n-hexane. The results demonstrated that using these plant extracts will be extremely advantageous in slowing the pace of carbohydrate digestion and absorption, hence contributing to effective diabetes control by lowering postprandial hyperglycemia. Future research will give insight into the molecular processes by which this plant and its active chemicals regulate glucose homeostasis.
Plant-based vaccines have demonstrated great therapeutic potential for respiratory disorders in recent years. This demonstrates that in the future, plant-based remedies will have a significant impact on the discovery of new medicinal products and therapies. Plant-based medicines have limitations as the mechanism of action for various pharmacological activities is not yet fully understood. A caution to the usage of medicinal plants is that around 15,000 species are under threat of becoming extinct owing to habitat loss and overharvesting, underscoring the urgent need for conservation measures. The use of biotechnological techniques such as tissue culture, micro-propagation, synthetic seed technology, and molecular-marker-based approaches can increase medicinal plant output and potency. These developments can be supplemented by conservation initiatives to guarantee the sustainable use of these substances. The plant compounds responsible for the pharmacological impacts, as well as their mode of action with safety precautions, should be researched further, follow by clinical studies. This will lead to the creation of medically acceptable plant-based therapies [17]. Various studies have shown indications that medicinal plants might be potential sources of novel antibacterial medicines, even against a variety of antibiotic-resistant bacteria. In this study, the antibacterial activity of acetone and ethanol extracts of the plant's leaves was assessed against Escherichia coli, Klebsiella pneumonia, Haemophilus influenzae, and Acinetobacter haumanni. The n-hexane extract of Pistacia Chinensis has the highest resistance to Escherichia colibacteria (1.4±0.07mm) and klebsiella bacteria (0.875±0.106mm) compared to standard antibiotics (Ampicillin) as shown in table 1 above.
Conclusion
The medicinal plant used in this study has excellent antioxidant and amylase inhibitory values, as well as powerful bioactivities that demonstrated resistance to certain microorganisms. These findings may give scientific proof for the traditional applications of this plant. However, further extensive investigations on bioactive compound separation and identification, as well as in vivo pharmacological activity evaluation, might be carried out in the future.
References
- Kumar, C., Kumar, R., & Nehar, S. (2013). Phytochemical properties and total antioxidant status of acetone and methanol extract of Terminalia arjuna Roxb. bark and its hypoglycemic effect on type II diabetic albino rats. Journal of Pharmacognosy and Phytochemistry, 2(1):199–208.
Publisher | Google Scholor - Sattar, S., Khan, M. R., Shah, N. A., Noureen, F., & Naz, K. (1970). Nephroprotective potential of Pistacia chinensis bark extract against induced toxicity in rats. Nusantara Bioscience, 8(2):192–200.
Publisher | Google Scholor - Voronkova, Y. S., Voronkova, O. S., Gorban, V. A., & Holoborodko, K. K. (2018). Oxidative stress, reactive oxygen species, antioxidants: A review. Ecology and Noospherology, 29(1):52–55.
Publisher | Google Scholor - Drioiche, A., et al. (2023). Analysis of the chemical composition and evaluation of the antioxidant, antimicrobial, anticoagulant, and antidiabetic properties of Pistacia lentiscus from Boulemane as a natural nutraceutical preservative. Biomedicines, 11(9):Article 2372.
Publisher | Google Scholor - Boukhris, M., Bouaziz, M., Feki, I., Jemai, H., El Feki, A., & Sayadi, S. (2012). Hypoglycemic and antioxidant effects of leaf essential oil of Pelargonium graveolens L’Hér. in alloxan-induced diabetic rats. Lipids in Health and Disease, 11, Article 81.
Publisher | Google Scholor - Haloui, T., et al. (2015). Bacteriostatic and bactericidal profile of leaves and twigs essential oils of Moroccan Pistacia lentiscus L. Journal of Applied Pharmaceutical Science, 5(6):50–53.
Publisher | Google Scholor - Alhadad, A. O., Elmhdwi, M. F., Elshareef, S. M., & Salem, G. S. (2021). Evaluation of the in vitro antibacterial activity of acetone leaf extracts from Pistacia lentiscus against multidrug-resistant Pseudomonas aeruginosa and Staphylococcus aureus. Journal of Medicinal Plants Studies, 9(4):100–105.
Publisher | Google Scholor - Shah, S. S., Manigauha, A., & Dubey, B. (2019). Formulation and evaluation of antidiabetic and antihyperlipidemic activities of polyherbal formulation in streptozotocin-induced diabetic rats. Pharmaceutical Biosciences Journal, 7(1):26–30.
Publisher | Google Scholor - Naz, K., Khan, M. R., Shah, N. A., Sattar, S., Noureen, F., & Awan, M. L. (2014). Pistacia chinensis: A potent ameliorator of CCl₄-induced lung and thyroid toxicity in a rat model. BioMed Research International, 2014, Article 192906.
Publisher | Google Scholor - Khan, W., et al. (2023). Effect of extraction solvent system on the antimicrobial, antioxidant and total polyphenol content of the bark of Pistacia chinensis. Iheringia, Série Botânica, 78:Article e2023022.
Publisher | Google Scholor - Aldughaylibi, F. S., et al. (2022). Extraction of bioactive compounds for antioxidant, antimicrobial, and antidiabetic applications. Molecules, 27(18):Article 5935.
Publisher | Google Scholor - Tebbi, S. O., Trapali, M., & Letsiou, S. (2024). Exploring the antidiabetic, antioxidant and antimicrobial properties of Clematis flammula L. leaves and Pistacia lentiscus L. fruits using choline chloride-based deep eutectic solvent. Waste and Biomass Valorization, 15(5):2869–2879.
Publisher | Google Scholor - Bouakline, H., et al. (2024). The phenolic content of Pistacia lentiscus leaf extract and its antioxidant and antidiabetic properties. The Scientific World Journal, 2024, Article 1998870.
Publisher | Google Scholor - Rajan, M., Chandran, V., Shahena, S., Anie, Y., & Mathew, L. (2022). In vitro and in silico inhibition of α-amylase, α-glucosidase, and aldose reductase by the leaf and callus extracts of Vernonia anthelmintica (L.) Willd. Advances in Traditional Medicine, 22(1):125–139.
Publisher | Google Scholor - Kunyanga, C. N., Imungi, J. K., Okoth, M., Momanyi, C., Biesalski, H. K., & Vadivel, V. (2011). Antioxidant and antidiabetic properties of condensed tannins in acetonic extract of selected raw and processed indigenous food ingredients from Kenya. Journal of Food Science, 76(4):C560–C567.
Publisher | Google Scholor - El-Beshbishy, H. A., & Bahashwan, S. A. (2012). Hypoglycemic effect of basil (Ocimum basilicum) aqueous extract is mediated through inhibition of α-glucosidase and α-amylase activities: An in vitro study. Toxicology and Industrial Health, 28(1):42–50.
Publisher | Google Scholor - Zanzabil, K. Z., & Hossain, S. (2023). Diabetes mellitus management: An extensive review of 37 medicinal plants (186–234).
Publisher | Google Scholor