Research Article
Assessment of Image Quality in Computed Tomography Using ACR Gammex 464 Phantom in Selected Hospitals in Southwestern Nigeria
1National Institute of Radiation Protection and Research, University of Ibadan, Ibadan, Nigeria.
2Nnamdi Azikiwe University, Anambra State, Nigeria (UNIZIK), Nigeria.
*Corresponding Author: Akerele Olumide Olaife, National Institute of Radiation Protection and Research, University of Ibadan, Ibadan, Nigeria.
Citation: Akerele O. Olaife, Oyeyemi S. Mofolorunso, Nzotta C. Chukwuemeka. (2026). Assessment of Image Quality in Computed Tomography Using ACR Gammex 464 Phantom in Selected Hospitals in Southwestern Nigeria, Journal of Radiology Research and Imaging, BioRes Scientia Publishers. 2(1):1-7. DOI: 10.59657/jrri.brs.26.005
Copyright: © 2026 Akerele Olumide Olaife, this is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: August 21, 2025 | Accepted: September 24, 2025 | Published: January 02, 2026
Abstract
Background: Standardised image-quality (IQ) assurance is essential to preserve diagnostic performance and quantitative accuracy in computed tomography (CT), but IQ data from low- and middle-income settings remain limited.
Objective: To benchmark CT IQ in Southwestern Nigeria against contemporary American College of Radiology (ACR) acceptance criteria using the ACR Gammex 464 phantom and to identify correctable deficits to guide facility QA and national policy.
Methods: In a multicentre cross-sectional audit, 12 CT scanners from 12 centres were evaluated across 20 clinical protocols (10 adult-head, 10 adult-abdomen). For each protocol, one axial acquisition per phantom module was obtained. Image analysis followed ACR “Phantom testing: CT” guidance (revised 9 Nov 2022), assessing slice thickness (±1.5 mm), CT-number accuracy for air, water, polyethylene, acrylic and bone, low-contrast detectability (adult CNR > 1.0), uniformity (edge-centre ≤ 5 HU), and high-contrast resolution (≥ 6 lp/cm head; ≥ 5 lp/cm abdomen).
Results: Slice thickness passed in 20/20 (100%) protocols. HU accuracy was excellent for low-density inserts air 19/20 (95%), water 18/20 (90%), polyethylene 20/20 (100%), acrylic 19/20 (95%) but weaker for bone 14/20 (70%), driven by head protocols (5/10) versus abdomen (9/10). Low-contrast detectability met the adult CNR criterion in 15/20 (75%) (head 6/10, abdomen 9/10). Uniformity passed in 20/20 (100%). High-contrast resolution met benchmarks in 17/20 (85%) (head 7/10, abdomen 10/10); two head protocols resolved 8 lp/cm.
Conclusions: CT systems demonstrated consistently strong geometry, uniform water-phantom response, and adequate high-contrast resolution. Actionable gaps were (i) positive HU bias for bone particularly in head protocols suggestive of residual beam-hardening/energy-response effects, and (ii) sub-threshold CNR in a subset of head studies. These data provide an implementable baseline for protocol-specific optimisation, targeted calibration/servicing, and integration of routine phantom audits within the national QA/accreditation framework.
Keywords: computed tomography; image quality; ACR Gammex 464 phantom; hounsfield unit accuracy; low-contrast detectability; contrast-to-noise ratio; spatial resolution; uniformity; beam hardening; quality assurance; nigeria; LMIC
Introduction
Computed tomography (CT) is central to modern diagnostics, but its population-dose contribution and the risk of suboptimal image quality demand rigorous quality assurance (QA) so that diagnostic benefits consistently outweigh potential harms (International Atomic Energy Agency [IAEA], 2011). The quality of an image and thus its ability to help with diagnosis, depends on task-relevant factors like low-contrast detectability, spatial resolution, noise, uniformity, and the accuracy of Hounsfield Units (HU). Acquisition parameters, reconstruction methods, and equipment calibration all have an effect on these factors. For example, iterative reconstruction can lower the dose while keeping the detectability, whereas inadequate beam-hardening correction can bias HU especially for dense materials like bone which makes CT less useful for quantitative purposes (Schindera et al., 2013; Solomon et al., 2015; Haase et al., 2022; Katsura et al., 2018). Phantom-based testing offers a consistent, patient-independent method for monitoring these parameters and for comparing scanner performance. The ACR Gammex 464 CT accreditation phantom has separate modules for checking HU accuracy, slice thickness, low- and high-contrast resolution, uniformity, and noise with well-define acceptance criteria used globally in clinical QA and accreditation (American College of Radiology, (ACR) 2017; McCollough et al., 2004; Sun Nuclear Corporation, n.d.). Extensive analyses of ACR accreditation data highlight the significance of standardized phantom audits in detecting inter-scanner variability and optimization opportunities (Boutin et al., 2021).
Comparing scanner performance to established QA standards shows that Europe has better compliance. This is because the European Society of Radiology (ESR) promotes and implements structured clinical audit frameworks, and the EU Basic Safety Standards are supported by the ESR "Esperanto" audit guide and toolkit. A national evaluation of Kenyan CT facilities, however, indicated an overall QA performance of 50 ± 3%, with image quality conforming to acceptance criteria in approximately 61% of facilities and QA program performance at 37%. This highlights the persistent disparities between high-resource and limited-resource environments (European Society of Radiology, 2019; European Commission, 2010; ESR, 2020; Korir et al., 2012). Health Management Publications Office of the EUPMC PubMed. The increasing use of CT in low- and middle-income countries, particularly in Sub-Saharan Africa, necessitates the need for robust quality assurance program. However, efforts to standardize imaging procedures and guarantee regulatory compliance are hampered by the substantial lack of published data on CT image quality performance in this area. Against this background, we conducted a multi-centre evaluation of CT image quality in selected hospitals in Southwestern Nigeria using the ACR Gammex 464 phantom and American College of Radiology acceptance criteria. Our objectives were to quantify compliance across key image-quality metrics, identify protocol- or equipment-related deficiencies amenable to corrective action, and provide region-specific baseline data to inform facility QA programmes and national policy.
Materials and Methods
Study Design and Setting
This work was designed as a cross-sectional, multi-center evaluation of CT scanner image quality. The study was conducted in selected hospitals located within Southwestern Nigeria. These facilities included a variety of private diagnostic centers with CT scanning capabilities as well as secondary and tertiary healthcare facilities.
ACR Gammex 464 Phantom and Image Acquisition
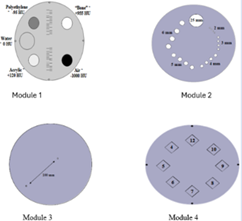
All image quality evaluations were conducted using the ACR Gammex 464 phantom, constructed from solid water-equivalent material. This cylindrical phantom is 20 cm in diameter by 16 cm in length. The ACR CT accreditation phantom consists of four modules designed to evaluate key image quality parameters. Module 1 assesses positioning and alignment, CT number accuracy, and slice thickness, featuring 1-mm steel BBs placed at 3, 6, 9, and 12 o’clock positions (19.9 cm apart), material cylinders representing bone, polyethylene, water, acrylic, and air for CT number accuracy evaluation as well as 0.5-mm ramp wires for slice thickness determination. Module 2 evaluates low-contrast resolution using cylinders of 2–6 mm diameters with 0.6% contrast against a ~90 HU background, along with a 25-mm cylinder to determine the contrast-to-noise ratio. Module 3 comprises a uniform tissue-equivalent material to assess CT number uniformity and includes 0.28-mm BBs for in-plane measurement accuracy and section sensitivity profile assessment. Module 4 measures high-contrast (spatial) resolution with bar patterns ranging from 4 to 12-line pairs per centimeter (lp/cm) within 15 × 15 mm areas, 3.8 cm in depth, and also contains four 1-mm steel beads for positional reference. ACR 2022 contains a detailed description and composition of the design. fig 1 shows each module's schematic representation, as provided by ACR 2022.
Figure 1: Each module's schematic representation, as provided by ACR 2022
Phantom Positioning and Scanning Protocol
The phantom was positioned at the isocenter of the CT gantry using manufacturer-specific alignment markers and immobilized to avoid motion artifacts. Scanning was performed using the axial mode, with a standard head protocol adapted for phantom evaluation. Key acquisition parameters were recorded, including tube voltage (kVp), tube current (mA), slice thickness, rotation time, and reconstruction algorithm. These parameters are specific to individual facility and equipment. Pictures showing the position of the phantom at the isocenter of the CT gantry is given in figure 2.
Figure 2: Pictures showing the position of the phantom at the CT gantry
Image Quality Parameters and Evaluation Criteria
The acquired images were transferred to a DICOM compatible image analysis workstation. Evaluation was performed using ACR Phantom Image‑Quality Tests and Acceptance Criteria (updated per ACR, 2022).
Table 1: ACR Phantom Image‑Quality Tests and Acceptance Criteria
| Parameter | How it’s tested on the ACR 464 phantom | ACR acceptance / pass–fail criterion |
| Alignment / positioning (Modules 1 & 4) | Single axial slice (≤ 2 mm) with DFOV ≈ 21 cm. Verify all four 1‑mm BBs are visible and wire‑ramp symmetry (±1 wire). | Pass if all four BBs are seen on one slice and long ramp wires are centered within ±1 wire. |
| CT‑number (HU) accuracy (Module 1) | Adult abdomen protocol preferred. Place ~200 mm² ROIs and WW 400, WL 0 in polyethylene, water, acrylic, bone, and air inserts; record mean HU. | Polyethylene −107 to −84 HU; Water −7 to +7 HU; Acrylic +110 to +135 HU; Bone 850 to 970 HU; Air −1005 to −970 HU. |
| Slice‑thickness accuracy (Modules 1 & 4) | Use wire‑ramp; count 0.5‑mm steps to compute measured slice width. | Measured slice width within ±1.5 mm of the prescribed value. |
| Low‑contrast detectability (Module 2) | Window/level: WW 100, WL 100. ROIs (~100 mm²) inside 25‑mm rod and background; compute CNR = |A − B| / SD. | Adult head & abdomen: CNR > 1.0 (pediatric thresholds differ). |
| Uniformity (Module 3) | WW 100, WL 0. Five ROIs (~400 mm²): center + four edges (one ROI diameter in from periphery); record mean HU. | Each edge–center difference ≤ 5 HU (5–7 HU minor deficiency; > 7 HU major deficiency). |
| High‑contrast (spatial) resolution (Module 4) | Visually determine highest resolvable bar pattern (4–12 lp/cm) with protocol‑appropriate windows. | Adult abdomen: resolve ≥ 5 lp/cm; high‑resolution protocols (e.g., head HR): resolve ≥ 6 lp/cm. |
Notes. Acceptance criteria and procedures follow ACR Accreditation Support “Phantom testing: CT” (revised Nov 9, 2022). Slice‑thickness and high‑contrast bar thresholds align with the ACR/AAPM Medical Physics description of the accreditation phantom tests.
Data Analysis
The raw data obtained from the phantom measurements are provided in tabular format, processed to derive meaningful statistics and compared to ACR 2022 acceptance criteria. Descriptive statistics were used to summarize performance across all facilities. Compliance rates were determined for each image quality metric, and scanners falling outside ACR thresholds were noted.
Ethical Considerations
Ethical approval for this study was obtained from the institutional review boards of participating hospitals. As the study involved only phantom imaging and did not include patient data, informed consent was not required
Results
Twenty CT scanners (10 adult-head, 10 adult-abdomen protocols) were audited against the ACR “Phantom testing: CT” acceptance criteria (revised 9 November 2022). One axial image per phantom module was acquired per protocol.
Slice thickness. All scanners met tolerance (measured width within ±1.5 mm of the prescribed value): 10/10 head and 10/10 abdomen (100%).
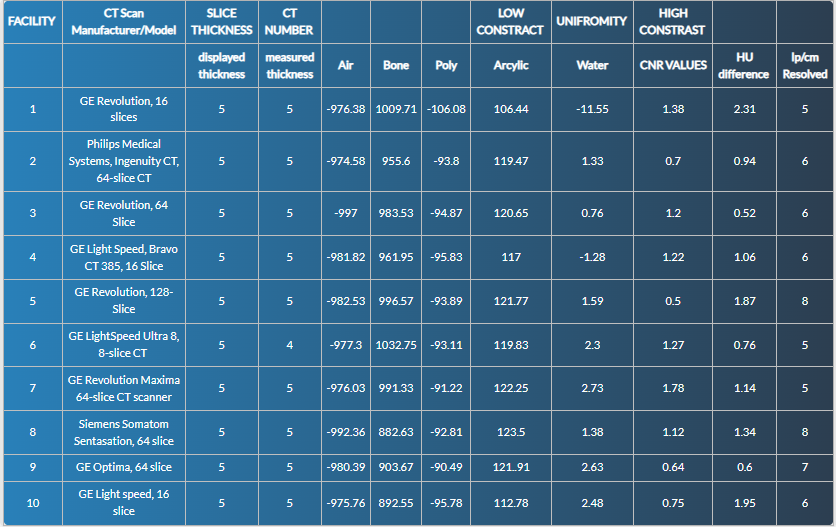
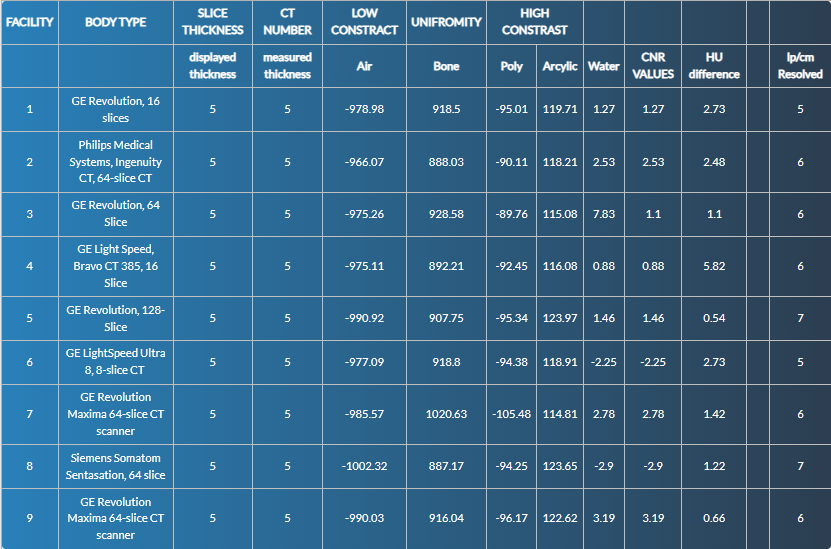
CT-number (HU) accuracy (Module 1). Performance was uniformly high for low-density inserts, with pass rates of 19/20 (95%) for air, 18/20 (90%) for water, 20/20 (100%) for polyethylene, and 19/20 (95%) for acrylic. High-density performance was weaker: bone passed in 14/20 (70%) overall, driven by a lower pass rate in head protocols (5/10) compared with abdomen (9/10).
Low-contrast detectability (Module 2). Using the adult criterion CNR > 1.0, 15/20 scanners (75%) passed: 6/10 head and 9/10 abdomen. Failing head studies showed CNR values of 0.50–0.75; the single failing abdomen study had CNR 0.88.
Uniformity (Module 3). Edge-to-centre HU differences satisfied the ≤ 5 HU threshold in all scanners (20/20, 100%).
High-contrast (spatial) resolution (Module 4). Using program benchmarks (≥ 6 lp cm⁻¹ for head; ≥ 5 lp cm⁻¹ for abdomen), 17/20 scanners (85%) passed overall: 7/10 head and 10/10 abdomen. Two head scanners resolved 8 lp cm⁻¹.
Table 2: Detailed pass-rate summary by protocol
| Parameter / Criterion | Head (n =10) | Abdomen (n=10) | Combined (n=20) |
| Slice thickness (±1.5 mm) | 10/10 (100%) | 10/10 (100%) | 20/20 (100%) |
| High-contrast resolution (lp/cm threshold) | 7/10 (70%) | 10/10 (100%) | 17/20 (85%) |
| Low-contrast detectability (CNR > 1.0) | 6/10 (60%) | 7/10 (70%) | 13/20 (65%) |
| Uniformity (edge-centre ≥ 5 HU) | 10/10 (100%) | 9/10 (90%) | 19/20 (95%) |
| HU accuracy: Air (-1005 to - 970) | 10/10 (100%) | 9/10 (90%) | 19/20 (95%) |
| HU accuracy: Water (-7 to +7) | 9/10 (90%) | 9/10 (90%) | 18/20 (90%) |
| HU accuracy: Polyethylene (-107 to -84) | 10/10 (100%) | 9/9 (100%) | 19/19 (100%) |
| HU accuracy: Acrylic (+110 to +135) | 7/9 (78%) | 10/10 (100%) | 17/19 (89%) |
| HU accuracy: Bone (+850 to +970) | 5/10 (50%) | 9/10 (90%) | 14/20 (70%) |
Discussion
This multi-centre phantom audit provides a robust baseline for CT image quality in Southwestern Nigeria and shows that most systems meet core technical standards when benchmarked against the ACR “Phantom testing: CT” criteria (revised 9 Nov 2022). All scanners satisfied the slice-thickness and uniformity tolerance (edge–centre ≤5 HU across sites), indicating stable system geometry and detector/beam conditioning (American College of Radiology [ACR], 2017; McCollough, Bruesewitz, & Kofler, 2004). High-contrast spatial resolution met program benchmarks in the majority of scanners (overall 85%), with two head protocols resolving 8 lp/cm, underscoring adequate modulation transfer for line-pair tasks of routine clinical relevance.
Two addressable deficiencies emerged. The accuracy of bone-insert HU was the most prevalent quantitative deficiency, with an overall pass rate of 70% (head 5/10 versus abdomen 9/10). This aligns with residual beam hardening and energy-response calibration effects that disproportionately affect dense materials, particularly on older platforms or when correction models are inadequate (Haase et al., 2022; Katsura et al., 2018). Biased HU can misclassify bone mineral density and disrupt electron-density maps utilized in radiotherapy planning, leading to potential dose misallocation. Secondly, low-contrast detectability was inferior in head protocols (CNR >1.0 in 6/10 head versus 9/10 abdomen), indicating potential for protocol optimization, thinner slices, task-specific kernels, and iterative reconstruction demonstrated to enhance detectability at constant or diminished dose (Schindera et al., 2013; Solomon et al., 2015). Collectively, these findings advocate for a transition from uniform QA to protocol-specific optimization, where kVp, mAs, kernel, and slice width collaboratively influence noise texture, beam-hardening characteristics, and quantitative precision.
The inter-scanner variability reported aligns with extensive accreditation datasets that reveal significant disparities in phantom performance, even with seemingly equivalent methods, underscoring the importance of standardized phantom audits for early fault discovery and ongoing enhancement (Boutin et al., 2021). At the systems level, regions that incorporate structured clinical audits under ESR guidance and the EU Basic Safety Standards demonstrate enhanced and more consistent compliance; in contrast, the Kenyan national baseline indicated an overall QA performance of approximately 50% (image-quality acceptance around 61%; program performance at 37%), underscoring enduring disparities between high- and low-resource environments (Council of the European Union, 2014; European Society of Radiology, 2019; Howlett, Brady, Ebdon-Jackson, & Hierath, 2023; Korir et al., 2012). In Nigeria, integrating routine phantom auditing with formal clinical audit and national licensure requirements would provide the governance necessary to sustain the predominantly good performance documented here (IAEA, 2011; ACR, 2017).
Limitations
This study was limited to a purposive sample of CT scanners in Southwestern Nigeria, potentially restricting the generalizability of our findings to other regions. Using only one phantom model (ACR Gammex 464) makes it hard to find possible performance problems that other QA tools could show better. Incomplete scanner metadata, including precise model year, usage workload, and comprehensive maintenance logs, impeded the accurate correlation of observed failures to specific equipment age or service history. Finally, our study only looked at phantom-based image quality metrics. We didn't look at how they affected clinical diagnostic accuracy or patient dose measurements. This means that we don't know how these technical problems will affect real life.
Conclusion
This multi-centre phantom audit establishes the baseline of CT image quality in Southwestern Nigeria in accordance with present ACR criteria. Slice thickness and uniformity were consistently exceptional, and high-contrast spatial resolution satisfied program requirements in the majority of systems. However, two amendable deficiencies were prominent: (i) a systematic positive bias in bone-equivalent Hounsfield Units, particularly in head protocols, indicative of residual beam-hardening and energy-calibration effects; and (ii) inadequate low-contrast detectability in a subset of head examinations. These deficiencies jeopardize quantitative activities (e.g., bone evaluation, radiation planning) and the identification of minor soft-tissue pathology. This audit identifies pattern-specific deficiencies and their probable technical causes, establishing a practical baseline for guiding protocol optimization, focused maintenance, and regulatory enhancement.
Recommendation
To translate these findings into durable quality gains, we recommend: (1) immediate technical remediation on underperforming scanners beam-hardening recalibration, verification of bow-tie filters and AEC settings etc; (2) protocol-specific QA cycles with regular constancy checks and annual full ACR-phantom audits, supported by calibrated QA tools and a designated site QA lead with trend reporting; (3) targeted upskilling of radiographers and medical physicists in advanced phantom measurement, reconstruction-kernel and slice-width optimisation, and noise/CNR management; (4) regional maintenance hubs staffed by certified engineers to deliver preventive servicing and reliable access to genuine parts; (5) centralised QA data registry to track pass/fail rates, flag outliers, and benchmark facilities; and (6) national policy alignment, whereby the NNRA adopts ACR-equivalent acceptance thresholds into licensing, couples them to periodic accreditation audits, and harmonises practice with structured clinical-audit guidance. Implemented together, these measures will enhance diagnostic reliability, protect patients, and align CT services with international best practice.
Summary
Summary of Image Quality Results Parameters for Adult Head Scans
Summary of Image Quality Results Parameters for Adult Abdomen Scans
References
- American College of Radiology. (2025). The 2017 CT QC manual. ACR Accreditation Support.
Publisher | Google Scholor - American College of Radiology. (2022). Phantom testing: CT. ACR Accreditation Support.
Publisher | Google Scholor - Boutin, R. D., Hernandez, A. M., Lenchik, L., Graffy, P. M., Pickhardt, P. J., & Yoo, D. C. (2021). CT phantom evaluation of 67,392 ACR accreditation examinations: Implications for opportunistic screening of osteoporosis using CT. AJR American Journal of Roentgenology, 216(2):447–452.
Publisher | Google Scholor - European Commission. (2009/2010). European Commission guidelines on clinical audit for medical radiological practices (diagnostic radiology, nuclear medicine and radiotherapy) (Radiation Protection No. 159). Luxembourg: Publications Office of the EU.
Publisher | Google Scholor - European Society of Radiology (ESR). (2018). The ESR audit tool (Esperanto): Genesis, contents and pilot. Insights into Imaging, 9(6):899–903.
Publisher | Google Scholor - European Society of Radiology (ESR). (2019). Esperanto: ESR guide to clinical audit in radiology and the ESR clinical audit tool (2nd ed.). Vienna: ESR.
Publisher | Google Scholor - Haase, V., Unberath, M., Maier, A., et al. (2022). Single-material beam-hardening correction via an analytical energy response model for diagnostic CT. Medical Physics, 49(10):6763–6779.
Publisher | Google Scholor - Howlett, D. C., Brady, A. P., Ebdon-Jackson, S., & Hierath, M. (2023). Clinical audit in European radiology: Current status and recommendations for improvement (endorsed by the ESR). Insights into Imaging, 14:116.
Publisher | Google Scholor - International Atomic Energy Agency. (2012). Quality assurance programme for computed tomography: Diagnostic and therapy applications (Human Health Series No. 19). Vienna: IAEA.
Publisher | Google Scholor - Katsura, M., Sato, J., Akahane, M., Kunimatsu, A., Abe, O., & Ohtomo, K. (2018). Current and novel techniques for metal artifact reduction at CT: Practical guide for radiologists. RadioGraphics, 38(2):450–461.
Publisher | Google Scholor - Korir, G. K., Wambani, J. S., Korir, I. K., Tries, M. A., & Mulama, M. M. (2012). Establishing the quality management baseline for diagnostic radiology in Kenya. Journal of Applied Clinical Medical Physics, 13(5):3764.
Publisher | Google Scholor - McCollough, C. H., Bruesewitz, M. R., & Kofler, J. M. (2004). The phantom portion of the ACR CT accreditation program: The tests and the criteria. Medical Physics, 31(9):2423–2441.
Publisher | Google Scholor - Malone, J., Guleria, R., Craven, C., Horton, P., Järvinen, H., Mayo, J., et al. (2012). Justification of diagnostic medical exposures: Some practical issues. Report of an International Atomic Energy Agency consultation. The British Journal of Radiology, 85(1013):523–538.
Publisher | Google Scholor - Schindera, S. T., Odedra, D., Raza, S. A., et al. (2013). Iterative reconstruction algorithm for CT: Can radiation dose be decreased while low-contrast detectability is preserved? Radiology, 269(2):511–518.
Publisher | Google Scholor - Solomon, J., Wilson, J., Grant, K. L., et al. (2015). Potential for radiation dose reduction in a multireader study of low-contrast detectability using advanced modeled iterative reconstruction. Radiology, 275(3), 735–745.
Publisher | Google Scholor - Sun Nuclear Corporation. (n.d.). ACR CT Accreditation Phantom (Gammex 464) datasheet. (Manufacturer’s product sheet).
Publisher | Google Scholor